- Department of Neuroscience, Winthrop Neuroscience, Winthrop University Hospital, Mineola, New York, USA
Correspondence Address:
Nancy E. Epstein
Department of Neuroscience, Winthrop Neuroscience, Winthrop University Hospital, Mineola, New York, USA
DOI:10.4103/2152-7806.191071
Copyright: © 2016 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Epstein NE. Non-neurological major complications of extreme lateral and related lumbar interbody fusion techniques. Surg Neurol Int 22-Sep-2016;7:
How to cite this URL: Epstein NE. Non-neurological major complications of extreme lateral and related lumbar interbody fusion techniques. Surg Neurol Int 22-Sep-2016;7:. Available from: http://surgicalneurologyint.com/surgicalint_articles/non%e2%80%91neurological-major-complications-extreme-lateral-related-lumbar-interbody-fusion-techniques/
Abstract
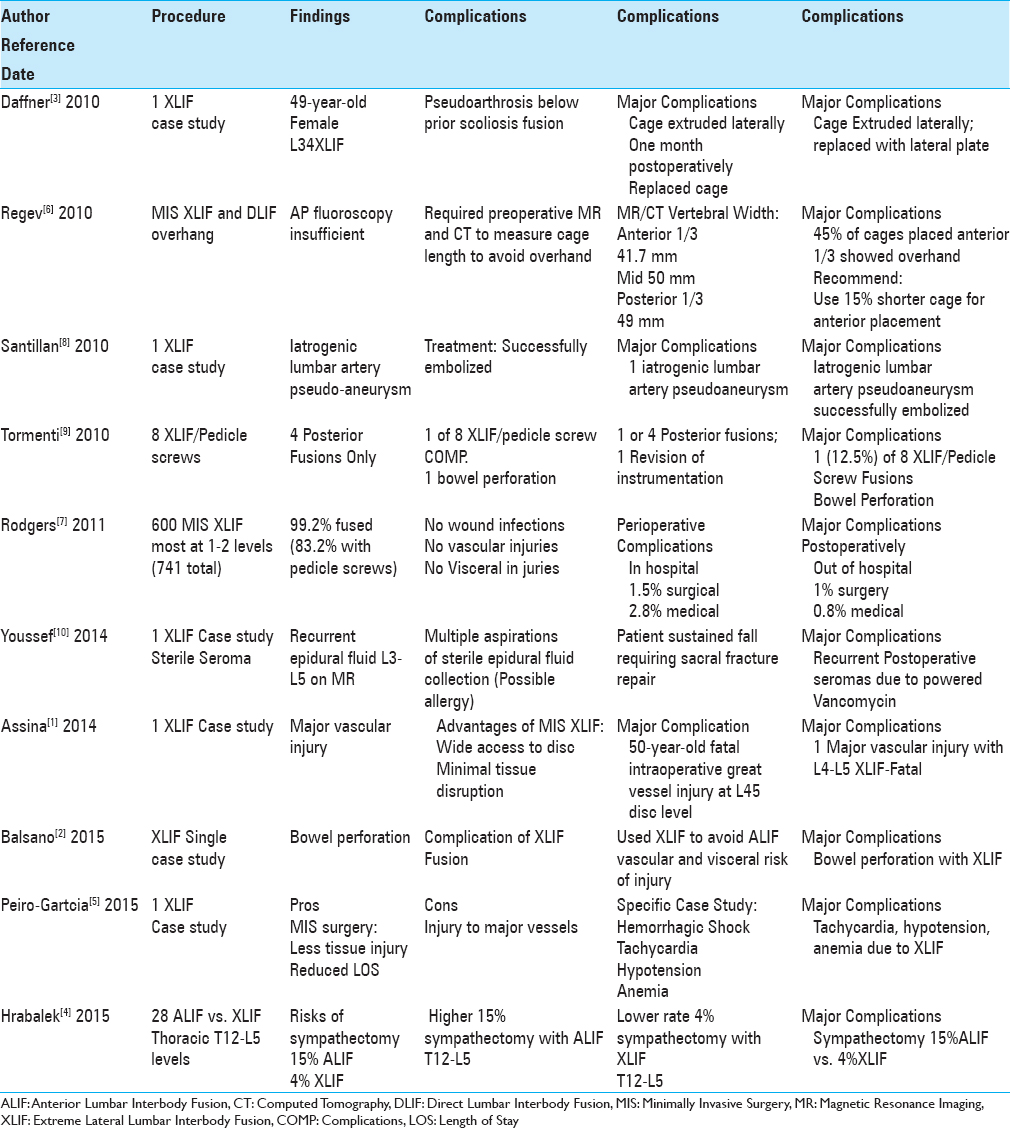
Background:Complications exclusive of new neurological deficits/injuries that follow extreme lateral interbody fusion (XLIF) and related lateral lumbar interbody techniques should be better recognized to determine the safety of these procedures. Unfortunately, a review of the XLIF literature did not accurately reflect the frequency of these “other complications” as few US surgeons publish such adverse events that may lead to medicolegal suits.
Methods:Major complications occurring with XLIF included sympathectomy, major vascular injuries, bowel perforations, sterile seromas, and instrumentation failures.
Results:The frequency of sympathectomy was 4% for XLIF vs. 15% for anterior lumbar interbody fusion (ALIF). There were three major vascular injuries for XLIF; one fatal intraoperative event, one life-threatening retroperitoneal hematoma, and one iatrogenic lumbar artery pseudoaneurysm that was successfully embolized. Two bowel perforations were reported, whereas a third was a “direct communication.” One patient developed a sterile recurrent seroma due to vancomycin powder utilized for an XLIF. One study cited malpositioning of an XLIF cage resulting in a lateral L3–L4 extrusion, whereas the second series looked at the 45% risk of cage-overhang when XLIF devices were placed in the anterior one-third of the vertebral body.
Conclusion:Excluding new neurological deficits, XLIF techniques resulted in multiple other major complications. However, these small numbers likely reflect just the tip of the iceberg (e.g., 10%) and the remaining 90% may never be known as many US-based spine surgeons fail to publish such adverse events as they are discoverable in a court of law and may lead to medicolegal suits.
Keywords: Bowel perforation, extreme lateral interbody fusion, lateral cage extrusion, major vessel injury, non-neurological complications, persistent seroma, retroperitoneal hematoma, sympathectomy
INTRODUCTION
Excluding new neurological deficits, other major complications following extreme lateral interbody fusions (XLIF) and related lateral lumbar interbody techniques should be better recognized to determine the safety of these procedures. The initial list of complications included sympathectomy, major vascular injuries, bowel perforations, seromas, and instrumentation failures. Unfortunately, this list does not likely accurately reflect the real frequency of these “other complications” as few US-based surgeons publish their personal/series-based adverse events due to medicolegal exposure (e.g., “discoverable” in a court of law and potentially leading to a suit). Therefore, the list of major XLIF complications likely constitutes just the tip of the iceberg (e.g., 10%), while the remaining 90% is yet to be published, leaving us wonder just how “safe” XLIF are.
EXCLUDING NEW NEUROLOGICAL DEFICITS, COMPLICATIONS OF MINIMALLY INVASIVE EXTREME LATERAL INTERBODY FUSION AND RELATED TECHNIQUES
Risk of sympathectomy with anterior lumbar interbody fusion vs. extreme lateral interbody fusion
Hrabalek et al. in 2015 retrospectively analyzed the risk of sympathectomy (utilizing thermography) performing anterior lumbar interbody fusion (ALIF) vs. XLIF procedures between the T12–L5 levels [
Three major vascular injuries attributed to extreme lateral interbody fusion
Three authors cited major vascular injuries attributed to XLIF. Santillan et al. in 2010 observed an iatrogenic lumbar artery pseudoaneurysm that followed a L4–L5 XLIF; it was successfully endovascularly embolized.[
Rare bowel perforations attributed to extreme lateral interbody fusion
Two studies documented bowel perforations resulting from XLIF procedures, whereas a third case was a personal communication to the author.[
Through a personal communication, the author was also told of a patient who within 12 hours of XLIF surgery was draining feces.
Case of sterile seroma after multilevel extreme lateral interbody fusion due to vancomycin powder
Youssef et al. in 2014 cited a 59-year-old female who developed a recurrent sterile postoperative seroma following the application of 2 g of vancomycin powder to the XLIF wound; 1 g/bone graft/fusion and 1 g placed in the soft tissues [
Malpositioning of extreme lateral interbody fusion cages: Migration and overhang
Malpositioning and overhang are two technical surgical complications that must be considered when performing XLIF interbody cage procedures.[
Medical vs. surgical complications of extreme lateral interbody fusion
Rodgers et al. in 2011 evaluated intraoperative/postoperative complications of 600 predominantly 1–2 level MIS XLIF (741 levels) that included a 99.2% instrumentation rate (83.2% pedicle screws).[
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Assina R, Majmundar NJ, Herschman Y, Heary RF. First report of major vascular injury due to lateral transpsoas approach leading to fatality. J Neurosurg Spine. 2014. 21: 794-8
2. Balsano M, Carlucci S, Ose M, Boriani L. A case report of a rare complication of bowel perforation in extreme lateral interbody fusion. Eur Spine J. 2015. 24: S405-8
3. Daffner SD, Wang JC. Migrated XLIF cage: Case report and discussion of surgical technique. Orthopedics. 2010. 33: 518-
4. Hrabalek L, Sternbersky J, Adamus M. Risk of sympathectomy after anterior and lateral lumbar interbody fusion procedures. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2015. 159: 318-26
5. Peiró-García A, Domínguez-Esteban I, Alía-Benítez J. Retroperitoneal hematoma after using the extreme lateral interbody fusion (XLIF) approach: Presentation of a case and a review of the literature. Rev Esp Cir Ortop Traumatol. 2015. p.
6. Regev GJ, Haloman S, Chen L, Dhawan M, Lee YP, Garfin SR. Incidence and prevention of intervertebral cage overhang with minimally invasive lateral approach fusions. Spine. 2010. 35: 1406-11
7. Rodgers WB, Gerber EJ, Patterson J. Intraoperative and early postoperative complications in extreme lateral interbody fusion: An analysis of 600 cases. Spine. 2011. 36: 26-32
8. Santillan A, Patsalides A, Gobin YP. Endovascular embolization of iatrogenic lumbar artery pseudoaneurysm following extreme lateral interbody fusion (XLIF). Vasc Endovascular Surg. 2010. 44: 601-3
9. Tormenti MJ, Maserati MB, Bonfield CM, Okonkwo DO, Kanter AS. Complications and radiographic correction in adult scoliosis following combined transpsoas extreme lateral interbody fusion and posterior pedicle screw instrumentation. Neurosurg Focus. 2010. 28: E7-
10. Youssef JA, Orndorff DG, Scott MA, Ebner RE, Knewitz AP. Sterile Seroma Resulting from Multilevel XLIF Procedure as Possible Adverse Effect of Prophylactic Vancomycin Powder: A Case Report. Evid Based Spine Care J. 2014. 5: 127-33