- Department of Neurosurgery, Beneficência Portuguesa, Ribeirão Preto, Brazil.
Correspondence Address:
Victoria Rodrigues Durand, Department of Neurosurgery, Beneficência Portuguesa, Ribeirão Preto, Brazil.
DOI:10.25259/SNI_1024_2023
Copyright: © 2024 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Breno Nery, Victoria Rodrigues Durand, Rafael De Almeida Rabello, Anna Carolyne Mendes De Oliveira, Eduardo Quaggio, Manoela Marques Ortega, Bruno Camporezi, José Alencar De Sousa Segundo. Occipital dermal sinus associated with infectious teratoma in an adult patient affected by Klippel–Feil syndrome: Rare case report and literature review. 22-Mar-2024;15:94
How to cite this URL: Breno Nery, Victoria Rodrigues Durand, Rafael De Almeida Rabello, Anna Carolyne Mendes De Oliveira, Eduardo Quaggio, Manoela Marques Ortega, Bruno Camporezi, José Alencar De Sousa Segundo. Occipital dermal sinus associated with infectious teratoma in an adult patient affected by Klippel–Feil syndrome: Rare case report and literature review. 22-Mar-2024;15:94. Available from: https://surgicalneurologyint.com/surgicalint-articles/12821/
Abstract
Background: The Klippel–Feil syndrome (KFS) is a rare congenital anomaly characterized by the fusion of cervical vertebrae, which may be associated with other malformations, such as dermoid tumors and teratoma. Some theories explain the embryology of these associations. Another condition that may be present is the dermal sinus (DS), communication between intracranial tumors and the subcutaneous tissue, and predisposing infections. This case report aims to describe an association between these three pathologies as well as correlate them from the literature. This report was based on medical records retrospectively reviewed associated with the systematic bibliographical consultation using indexed databases based on inclusion and exclusion methods.
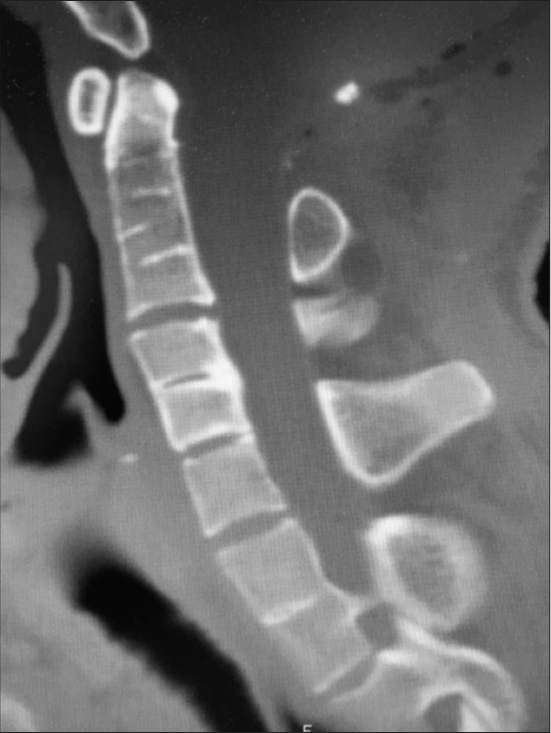
Case Description: An adult male patient, 24 years old, was admitted to our service, presenting fever and meningeal irritation as initial symptoms. In the patient’s clinical history, he was diagnosed with an occipital DS in his childhood, which was previously instructed to be operated on by another neurosurgical team, but the patient chose not to perform the procedure. The magnetic resonance imaging investigation showed a DS associated with a cerebellar infected mass with 2 cm on its main diameter. The patient was treated with preoperative antibiotic therapy and underwent gross total surgical resection of the tumor as well as DS correction, confirmed in the histopathological examination as a teratoma. After surgery, further computed tomography scan analysis showed the presence of cervical vertebrae fusion, compatible with KFS diagnosis.
Conclusion: The association between KFS, cerebellar teratoma, and DS has not yet been described in the literature, with only the association of the first two being extremely rare.
Keywords: Dermal sinus, Klippel–Feil syndrome, Teratoma
INTRODUCTION
Klippel–Feil syndrome (KFS) is characterized by the classic triad consisting of limitation of movement, low scalp implantation, and short neck.[
The dermal sinus (DS) is defined as an epithelium-lined tract that results from incomplete separation of the cutaneous ectoderm from the underlying neuroectoderm. Although DS is an easily identifiable and curable lesion, its pathological importance resides in the risk of intracranial infection[
Teratomas originate from failures in the differentiation of embryonic cells, thus being developed during prenatal life. They usually have a good prognosis, but it depends on several clinical factors such as location, gender, age at diagnosis, and histology. They comprise between 0.5% and 1.5% of all intracranial tumors, with a preference for locations close to midline structures, such as the pineal gland and neurohypophysis. Cerebellar teratomas are extremely rare, with few cases described in the literature so far.
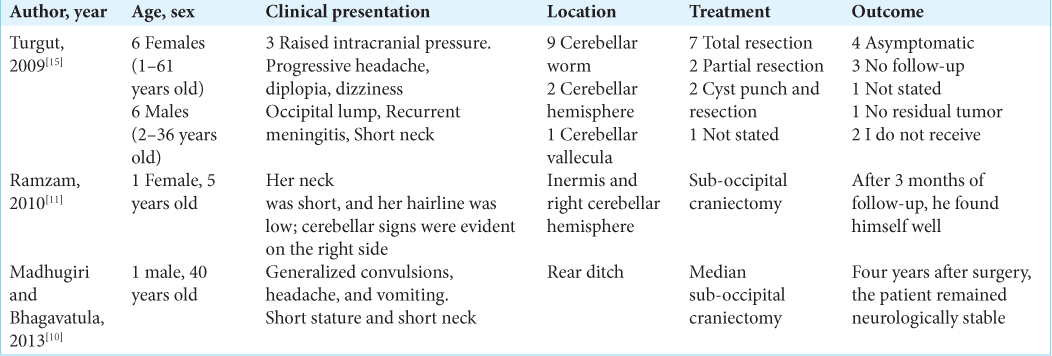
KFS has an incidence of 1:40,000–42,000 births, and DS affects <1% of the neonatal population. In the literature, there are already descriptions of cases of dermoid associated with KFS, as well as KFS and teratomas, despite few identified cases. The association of DS, infected teratoma, and KFS with initial symptoms in adulthood has not yet been described in the literature.[
CASUISTIC AND METHODS
Bibliographic research
A bibliographic search was carried out in the MEDLINE, LILACS, and PubMed-indexed databases, with the following descriptors: “Dermal Sinus,” “Teratoma,” and “Klippel– Feil Syndrome,” using the Boolean operator “AND.” The descriptors were interspersed during the database search to increase the number of identified cases. Language was selected as a selection criterion, preferably choosing articles in Portuguese, Spanish, or English and only articles based on studies with human beings. No year limitation was added during the search. Fifty-five articles were found, and 15 were selected for literature review.
Histological study
The surgical samples were fixed with 10% formaldehyde and were cut in the central region for the process of paraffin embedding after 24 h. The paraffin blocks were cut lengthwise with a thickness of 4 μm for mounting slides that the Hematoxylin stained–Eosin technique aiming to analyze the histological changes of the specimens. The histological analysis objected to determining the presence of tumor, necrosis, pleomorphism, mitosis, fibrosis, and characterization of cells.
Immunohistochemical analysis
The specimens embedded in paraffin also underwent to immunohistochemical staining technique using the avidinbiotin peroxidase complex method (Dako, Glostrup, Denmark) with diaminobenzidine as the chromogen and the primary antibodies: epithelial membrane antigen, synaptophysin, glial fibrillary acidic protein (GFAP), cytokeratin (CAM 5.2), S-100 protein, and Ki-67. Finally, the slides were mounted with Permount (Fisher Scientific, Pittsburgh, USA).
CASE REPORT
Clinical history and neurological examination
An adult patient, a male 24 years old, was admitted to our service, presenting fever as an initial symptom. On neurological examination, we showed the presence of meningeal irritation associated with phlogistic signs and continuous solution of skin in the occipital region while no other clinical abnormality was detected. In the patient’s past clinical history, he was diagnosed with an occipital DS in his childhood, which was previously instructed to be operated on by another neurosurgical team. However, the patient refused to perform the procedure. A lumbar puncture of the cerebrospinal fluid showed suggestive bacterial meningitis (protein elevated, polymorphonuclear leukocytes, and low glucose) in the biochemical and cytological analysis.
Neuroimaging investigation
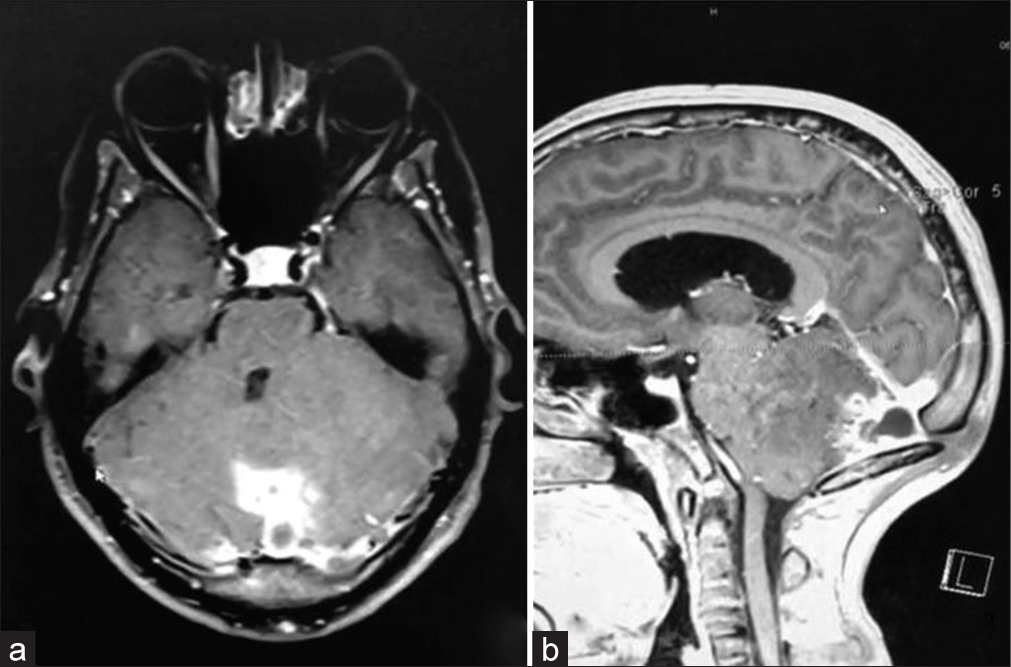
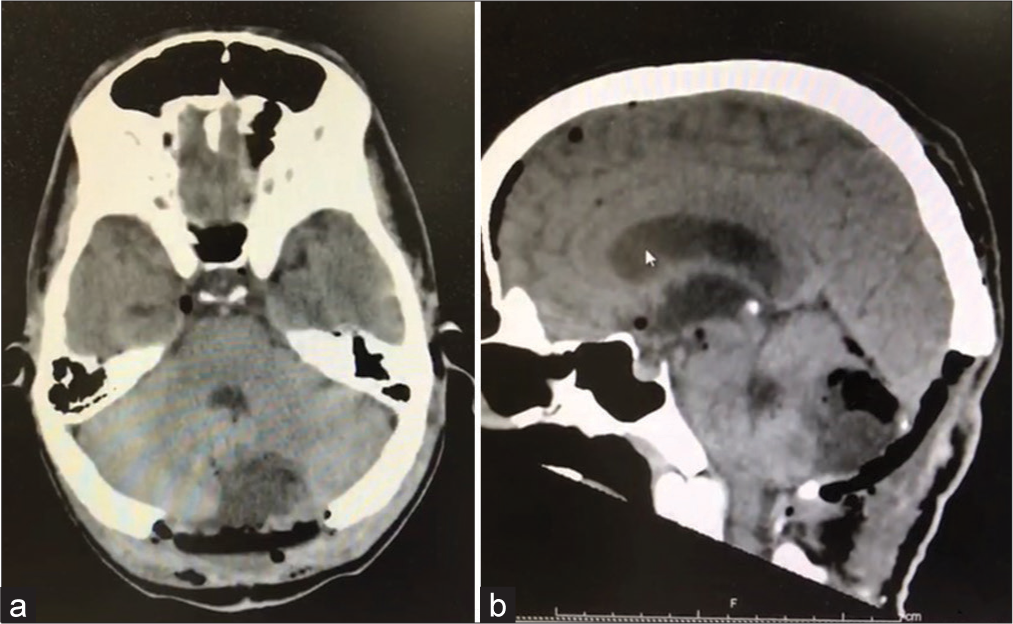
The magnetic resonance imaging (MRI) investigation showed the previously diagnosed DS associated with a cerebellar mass with 2.0 cm on its main diameter. The cerebellar mass presented as an enhanced, heterogeneous, partially hyperintense posterior fossa lesion with moderate mass effect on axial and sagittal contrast-enhanced T1-weighted MRI [
Surgery
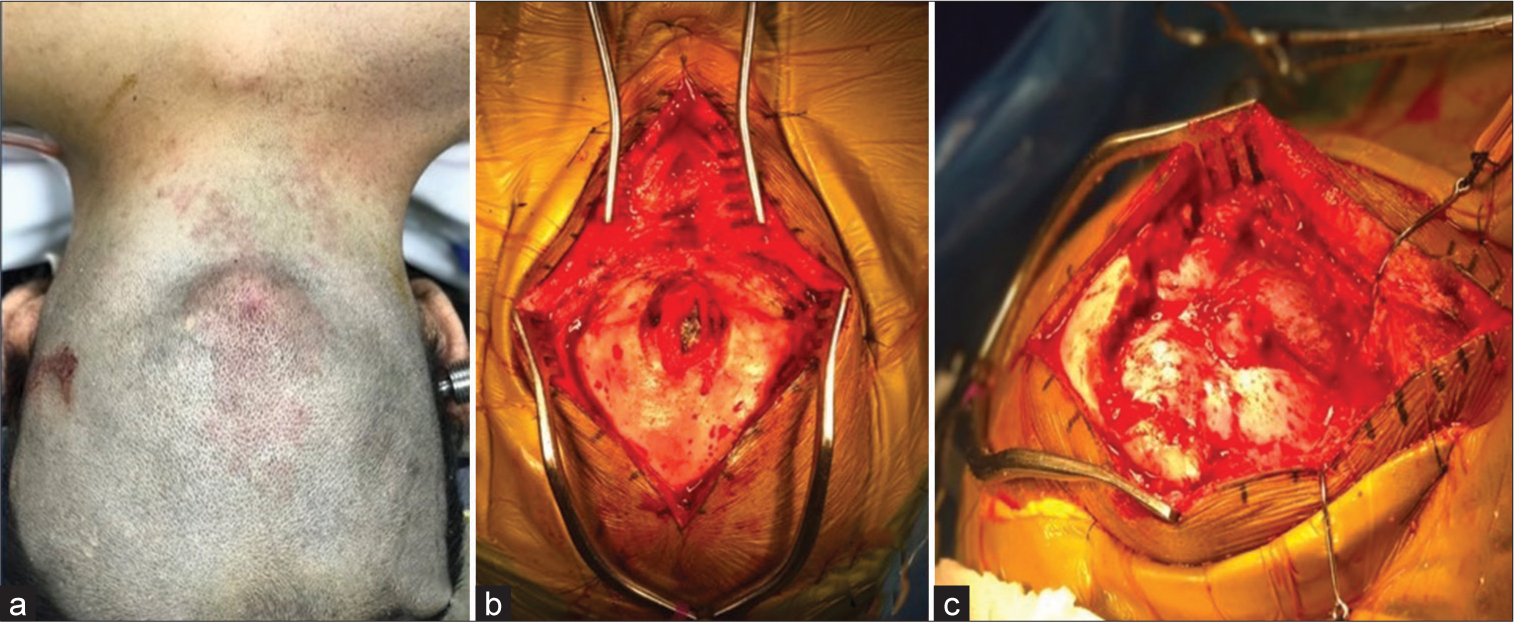
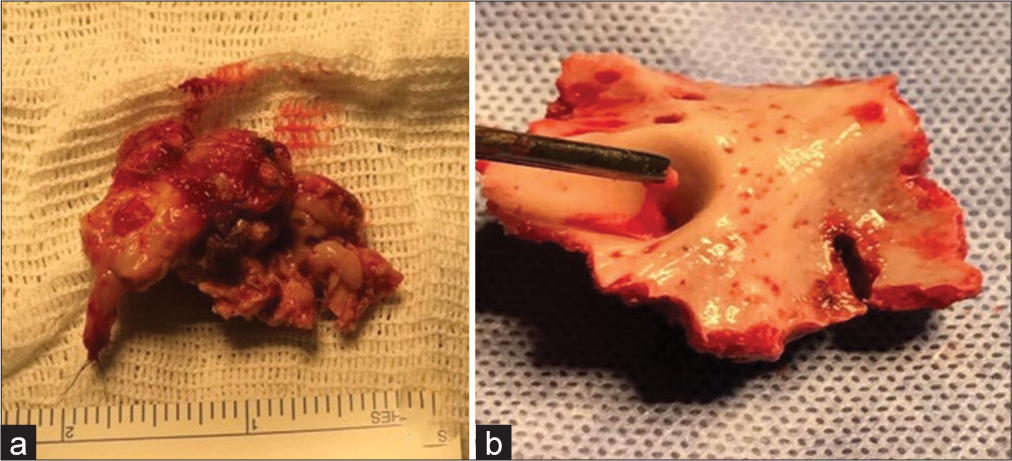
The patient was treated with antibiotic therapy for ten days preoperatively. After the preoperative trichotomy, it is possible to see the DS skin defect [
Pathological examination
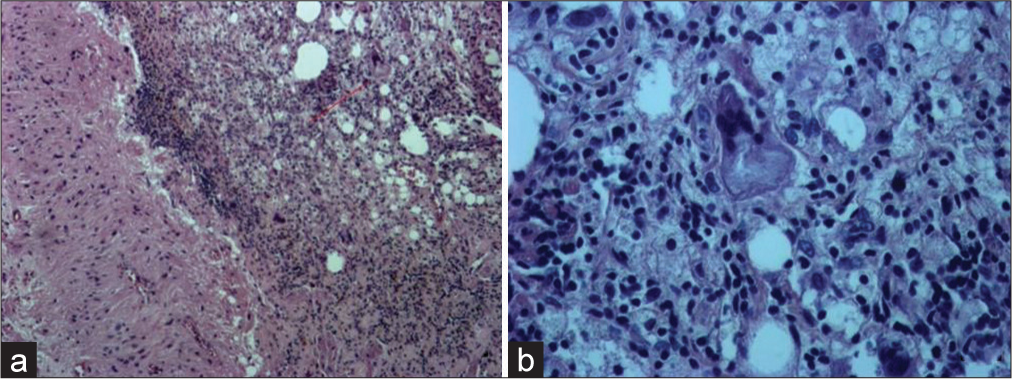
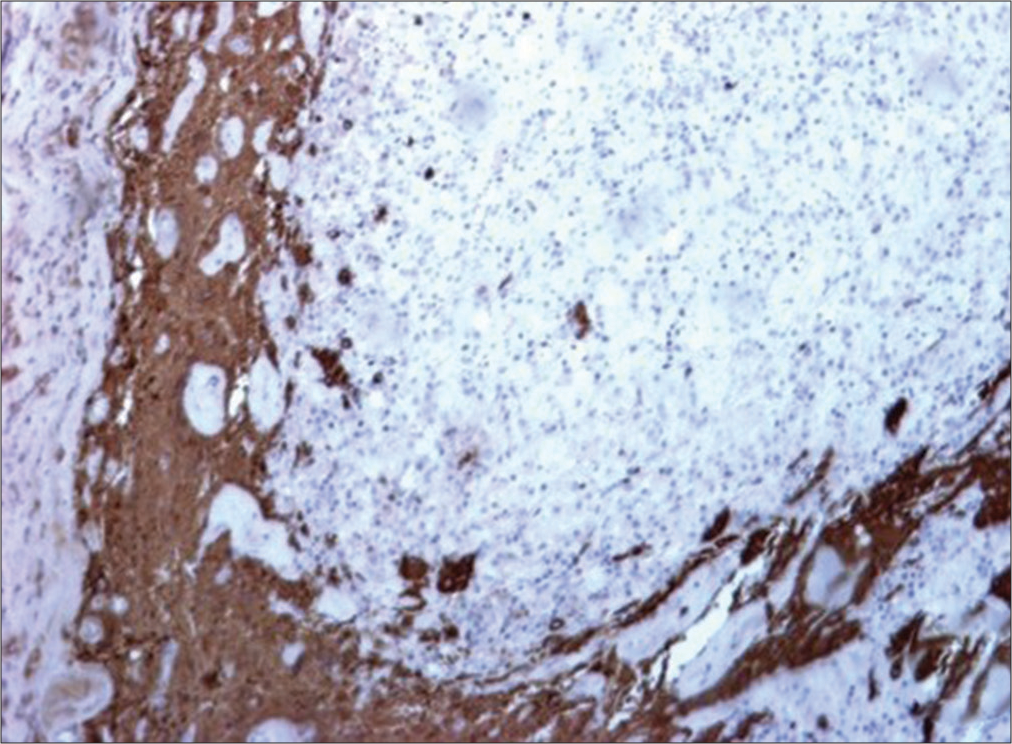
The conventional H&E (×100) showed a chronic granulomatous inflammatory process with the presence of body giant cells [
Postoperative
The postoperative period was uneventful; neurological examination did not reveal any additional deficits, and imaging control did not show any residual or recurrent tumor [
DISCUSSION
KFS is a congenital condition that was first described in 1912. The prevalence of this syndrome has been reported to be high (1 in 172 live births),[
Embryologically, during the 1st week of gestation, the formation of the neural tube occurs, which is a structure responsible for the formation of the central nervous system. Between the 3rd and 8th weeks of embryonic life, the formation of segmental cervical sclerotomes occurs; at this stage, defects may occur, and a hypothesis proposes that KFS may be a type of failure in the proper separation of the cervical sclerotomes during embryonic development. These sclerotomes must separate correctly to form the individual vertebrae and associated structures, such as the muscles and ligaments of the cervical spine.[
Intracranial teratoma is a rare type of tumor that makes up only 0.3–0.9% of all intracranial tumors and tends to present in midline sites such as the pineal gland and suprasellar regions. Teratomas usually occur in the first two decades of life; however, few cases have been reported in adults.[
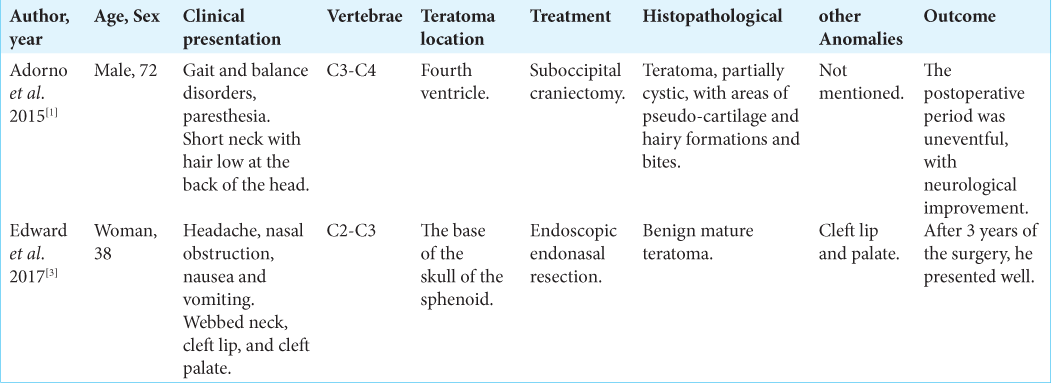
During the database search, only two reports involving KFS-associated teratoma were found, with different locations; one located in the fourth ventricle and the other located at the base of the skull [
Congenital DS arises from incomplete separation of surface ectoderm and neural ectoderm, resulting in focal segmental adhesion.[
CONCLUSION
The association between KFS, cerebellar teratoma, and DS has not yet been described in the literature, and the association of the first two is rare, with a reduced number of reported cases. Patients who present KFS or alterations in the posterior fossa need to be investigated for the early diagnosis of other possible associated congenital lesions, looking for alterations from the posterior fossa to the complete cervical spine. As well as patients with a history of recurrent meningitis, they should be investigated to assess whether they have a DS and other associated congenital pathologies. There is no established single treatment, requiring a careful and individualized assessment of each case.
Ethical approval
Institutional Review Board approval is not required.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript, and no images were manipulated using AI.
Disclaimer
The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Journal or its management. The information contained in this article should not be considered to be medical advice; patients should consult their own physicians for advice as to their specific medical needs.
References
1. Adorno A, Alafaci C, Sanfilippo F, Cafarella D, Scordino M, Granata F. Malignant teratoma in klippel-feil syndrome: A case report and review of the literature. J Med Case Rep. 2015. 9: 229
2. Bohara M, Yonezawa H, Karki P, Bakhtiar Y, Hirano H, Kitazono I. Mature posterior fossa teratoma mimicking dermoid cyst. Brain Tumor Pathol. 2012. 30: 262-5
3. Edward JA, Psaltis AJ, Williams RA, Charville GW, Dodd RL, Nayak JV. Endoscopic resection of skull base teratoma in klippel-feil syndrome through use of combined ultrasonic and bipolar diathermy platforms. Case Rep Otolaryngol. 2017. 2017: 6384586
4. Foster MT, Moxon CA, Weir E, Sinha A. Dermal sinus tracts. BMJ. 2019. 366: l5202
5. Gobel U, Calaminus G, Engert J, Kaatsch P, Gadner H, Bökkerink JP. Teratomas in infancy and childhood. Med Pediatr Oncol. 1998. 31: 8-15
6. Gruber J, Saleh A, Bakhsh W, Rubery PT, Mesfin A. The prevalence of klippel-feil syndrome: A computed tomography-based analysis of 2,917 patients. Spine Deform. 2018. 6: 448-53
7. Gumerlock MK, Spollen LE, Nelson MJ, Bishop RC, Cooperstock MS. Cervical neurenteric fistula causing recurrent meningitis in klippel-feil sequence: Case report and literature review. Pediatr Infect Dis J. 1991. 10: 532-5
8. Harms D, Zahn S, Gobel U, Schneider DT. Pathology and molecular biology of teratomas in childhood and adolescence. Clin Pediatr. 2006. 218: 296-302
9. Hinojosa M, Tatagiba M, Harada K, Samii M. Dermoid cyst in the posterior fossa accompanied by klippel-feil syndrome. Childs Nerv Syst. 2001. 17: 97-100
10. Madhugiri VS, Bhagavatula ID. Congenital anomalies at the craniovertebral junction-posterior fossa region: Report of two cases. J Neurol Surg A Cent Eur Neurosurg. 2013. 74: e279-83
11. Ramzan A, Khursheed N, Rumana M, Abrar W, Ashish J. Posterior fossa dermoid with klippel-feil syndrome in a child. Pediatr Neurol. 2011. 45: 197-9
12. Samartzis DD, Herman J, Lubicky JP, Shen FH. Classification of congenitally fused cervical patterns in klippel-feil patients: Epidemiology and role in the development of cervical spine-related symptoms. Spine (Phila Pa 1976). 2006. 31: E798-804
13. Song Y, Xia Z, Qiu S, Gao P, Yang B, Bao N. Surgical treatment of congenital dermal sinus: An experience of 56 cases. Pediatr Neurosurg. 2021. 56: 416-23
14. Thomsen MN, Schneider U, Weber M, Johannisson R, Niethard FU. Scoliosis and congenital anomalies associated with klippel-feil syndrome types I-III. Spine (Phila Pa 1976). 1997. 22: 396-401
15. Turgut M. Klippel-feil syndrome in association with posterior fossa dermoid tumour. Acta Neurochir (Wien). 2009. 151: 269-76