- Clinical Professor of Neurosurgery, School of Medicine, State University of NY at Stony Brook, and Editor-in-Chief Surgical Neurology International NY and c/o Dr. Marc Agulnick 1122 Franklin Avenue Suite 106, Garden City, NY 11530, United States.
- Department of Orthopedics, Assistant Clinical Professor of Orthopedics, NYU Langone Hospital, Long Island, Garden City, New York, 1122 Franklin Avenue Suite 106 Garden City, NY 11530, United States.
Correspondence Address:
Nancy E Epstein, M.D., F.A.C.S., Clinical Professor of Neurosurgery, School of Medicine, State University of NY at Stony Brook, and Editor-in-Chief Surgical Neurology International NY and c/o Dr. Marc Agulnick 1122 Franklin Avenue Suite 106, Garden City, NY 11530, United States.
DOI:10.25259/SNI_648_2023
Copyright: © 2023 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Nancy E Epstein1, Marc A Abulnick2. Perspective: Transthoracic, posterolateral, or transpedicular approaches to thoracic disks, not laminectomy. 25-Aug-2023;14:303
How to cite this URL: Nancy E Epstein1, Marc A Abulnick2. Perspective: Transthoracic, posterolateral, or transpedicular approaches to thoracic disks, not laminectomy. 25-Aug-2023;14:303. Available from: https://surgicalneurologyint.com/surgicalint-articles/12510/
Abstract
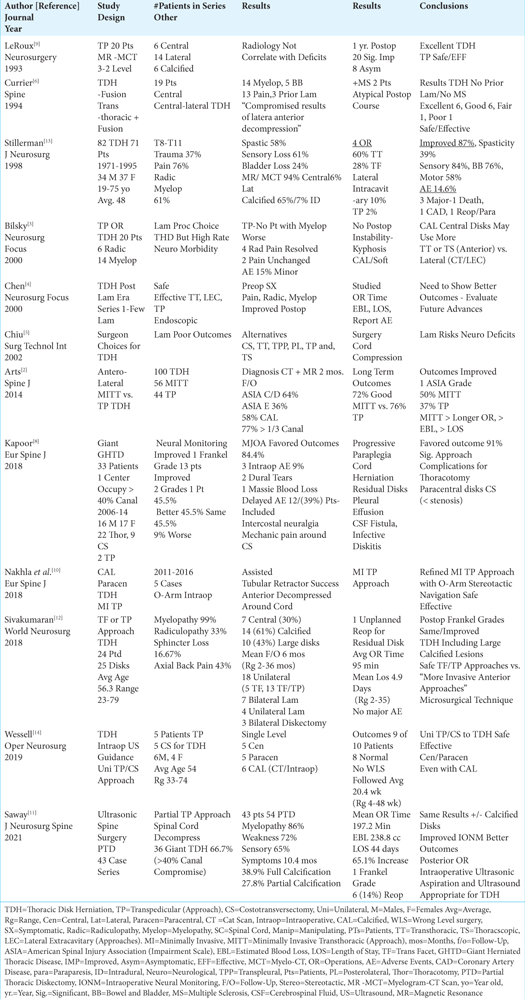
Background: Anterior transthoracic, posterolateral (i.e., costotransversectomy/lateral extracavitary), and transpedicular approaches are now utilized to address anterior, anterolateral, or lateral thoracic disk herniations (TDH). Notably, laminectomy has not been a viable option for treating TDH for decades due to the much lower rate of acceptable outcomes (i.e., 57% for decompressive laminectomy vs. over 80% for the posterolateral, lateral, and transthoracic procedures), and a higher risk of neurological morbidity/paralysis.
Methods: Patients with TDH averaged 48-56.3 years of age, and presented with pain (76%), myelopathy (61%-99%), radiculopathy (30%-33%), and/or sphincter loss (16.7%-24%). Those with anterior/anterolateral TDH (30-74%) were usually myelopathic while those with more lateral disease (50-70%) exhibited radiculopathy. Magnetic resonance (MR) studies best defined soft-tissue/disk/cord pathology, CAT scan (CT)/Myelo-CT studies identified attendant discal calcification (i.e. fully calcified 38.9% -65% vs. partial calcification 27.8%), while both exams documented giant TDH filling > 30 to 40% of the canal (i.e., in 43% to 77% of cases).
Results: Surgical options for anterior/anterolateral TDH largely included transthoracic or posterolateral approaches (i.e. costotransversectomy, lateral extracavitary procedures) with the occasional use of transfacet/transpedicular procedures mostly applied to lateral disks. Notably, patients undergoing transthoracic, lateral extracavitary/costotransversectomy/ transpedicular approaches may additionally warrant fusions. Good/excellent outcomes were quoted in from 45.5% to 87% of different series, with early postoperative adverse events reported in from 14 to 14.6% of patients.
Conclusion: Anterior/anterolateral TDH are largely addressed with transthoracic or posterolateral procedures (i.e. costotransversectomy/extracavitary), with a subset also utilizing transfacet/transpedicular approaches typically adopted for lateral TDH. Laminectomy is essentially no longer considered a viable option for treating TDH.
Keywords: Thoracic disk herniation, Anterior surgery, Transthoracic: posterolateral surgery, Costotransversectomy/ lateral extracavitary, Posterior surgery, Transfacet/transpedicular
INTRODUCTION
For many decades, laminectomy has no longer been considered a viable option for treating TDH due to its’ high risk of paralysis/increased morbidity [
Series Citing Abandonment of Laminectomy for TDH Due to Risk of Paralysis
Several studies emphasized that performing laminectomy for TDH has been abandoned for decades [
Analysis of Subset of 345 Patients with TDH in 10 of 14 Studies
This review yielded 345 patients treated for TDH from 10 studies [
Average Age for Patients with TDH
Patients with TDH usually averaged between 48-56.3 years of age [
THD: Clinical Presentations Included Pain, Myelopathy, Radiculopathy, and Sphincter Dysfunction
Patients in these 10 series of TDH[
MR Studies Best Documented TDH Location and Spinal Cord Pathology
For TDH, most typically found between the T8-T11 levels, MR studies best documented the soft tissues (i.e., disks, ligaments, stenosis etc.), spinal cord pathology (i.e., intrinsic high cord signals, edema, extent of cord compression), and location of TDH (i.e. central/anterior (30%), central/ anterolateral (50%-100%), or lateral (6%-70%)) [
CT/Myelo-CT Studies Best Documented Calcification of TDH
CT and Myelo-CT exams best showed whether TDH were fully calcified (i.e., range 38.9% to 65%) vs. partially calcified (up to 27.8*); the extent of calcification of these TDH was critical in determining optimal surgical approaches [
Incidence of Giant TDH Defined as Occupying > 30 to > 40% of the Spinal Canal
Several studies documented varying frequencies (i.e., 43%-100%) of large to giant TDH occupying > 30 to > 40% of the spinal canal [
Outcomes for the Different Operative Approaches to TDH
Thoracic disk surgery is usually more complex with often poorer outcomes when compared to lumbar or cervical disk herniations. In the thoracic spine, exposure of thoracic disks, typically requiring working anterior and/or anterolateral to the cord, limits midline access which is compounded by the high incidence of calcified disks. Further, limited corridors of access make addressing cerebrospinal fluid (CSF) leaks/dural repairs more difficult.
Transpedicular (TP), and Partial Transpedicular (PTP) Approaches For Addressing Central, Paracentral, and Lateral TDH
Three studies effectively utilized transpedicular (TP), minimally invasive TP (MI TP), and partial TP (PTP) approaches to TDH [
Transpedicular and Costotransversectomy Approaches to Central/Paracentral TDH
Wessell et al. (2019) 10 patients with single level disease underwent 5 transpedicular vs. 5 costotransversectomy approaches to 5 central and 5 paracentral TDH [
Transfacet (TF)/Transfacet-Transpedicular (TF-TP) Approaches
Sivakumaran et al. (2018) 25 TDH were managed with 18 unilateral approaches (i.e. 5 transfacet (TF) and 13 transfacet/ transpedicular procedures) vs. 7 laminectomies; all patients’ preoperative Frankel grades were the same or better [
Outcomes and Adverse Events Following Transthoracic, Transfacet, Costotransversectomy, Intracavitary, and Transpedicular Surgery for TDH
Multiple studies cited the safety/efficacy of transthoracic, transfacet, costotransversectomy, intracavitary and transpedicular surgical approaches for TDH [
CONCLUSION
Anterior/anterolateral TDH are typically addressed with transthoracic or posterolateral procedures (i.e. costotransversectomy/extracavitary), with occasional patients also undergoing transfacet/transpedicular approaches; with any of these approaches, subsequent fusion may be warranted.[
Declaration of patient consent
Patients’ consent not required as patients’ identities were not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflict of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The author(s) confirms that there was no use of Artificial Intelligence (AI)-Assisted Technology for assisting in the writing or editing of the manuscript and no images were manipulated using the AI.
Disclaimer
The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Journal or its management. The information contained in this article should not be considered to be medical advice; patients should consult their own physicians for advice as to their specific medical needs.
References
1. Arce CA, Dohrmann GJ. Thoracic disk herniation. Improved diagnosis with computed tomographic scanning and a review of the literature. Surg Neurol. 1985. 23: 356-61
2. Arts MP, Bartels RH. Anterior or posterior approach of thoracic disk herniation? A comparative cohort of mini-transthoracic versus transpedicular discectomies. Spine J. 2014. 14: 1654-62
3. Bilsky MH. Transpedicular approach for thoracic disk herniations. Neurosurg Focus. 2000. 9: e3
4. Chen TC. Surgical outcome for thoracic disk surgery in the postlaminectomy era. Neurosurg Focusm. 2000. 9: e12
5. Chiu JC, Clifford TJ, Sison R. Percutaneous microdecompressive endoscopic thoracic discectomy for herniated thoracic disks. Surg Technol Int. 2002. 10: 266-9
6. Currier BL, Eismont FJ, Green BA. Transthoracic disk excision and fusion for herniated thoracic disks. Spine (Phila Pa 1976). 1994. 19: 323-8
7. Fessler RG, Sturgill M. Review: Complications of surgery for thoracic disk disease. Surg Neurol. 1998. 49: 609-18
8. Kapoor S, Amarouche M, Al-Obeidi F, U-King-Im JM, Thomas N, Bell D. Giant thoracic disks: Treatment, outcome, and follow-up of 33 patients in a single centre. Eur Spine J. 2018. 27: 1555-66
9. Le Roux PD, Haglund MM, Harris AB. Thoracic disk disease: Experience with the transpedicular approach in twenty consecutive patients. Neurosurgery. 1993. 33: 58-66
10. Nakhla J, Bhashyam N, De la Garza Ramos R, Nasser R, Kinon MD, Yassari R. Minimally invasive transpedicular approach for the treatment of central calcified thoracic disk disease: A technical note. Eur Spine J. 2018. 27: 1575-85
11. Saway BF, Alshareef M, Lajthia O, Cunningham C, Shope C, Martinez JL. Ultrasonic spine surgery for every thoracic disk herniation: A 43-patient case series and technical note demonstrating safety and efficacy using a partial transpedicular thoracic discectomy with ultrasonic aspiration and ultrasound guidance. J Neurosurg Spine. 2021. 36: 800-8
12. Sivakumaran R, Uschold TD, Brown MT, Patel NR. Transfacet and transpedicular posterior approaches to thoracic disk herniations: Consecutive case series of 24 patients. World Neurosurg. 2018. 120: e921-31
13. Stillerman CB, Chen TC, Couldwell WT, Zhang W, Weiss MH. Experience in the surgical management of 82 symptomatic herniated thoracic disks and review of the literature. J Neurosurg. 1998. 88: 623-33
14. Wessell A, Mushlin H, Fleming C, Lewis E, Sansur C. Thoracic discectomy through a unilateral transpedicular or costotransversectomy approach with intraoperative ultrasound guidance. Oper Neurosurg (Hagerstown). 2019. 17: 332-7