- Virtual Primary-Care Connection, LLC and Canopy Health, Houston,
- Department of Neurology, Houston Methodist Hospital, Houston,
- Department of Neurosurgery, Houston Methodist Hospital, Houston,
- Department of Hospitality, Houston Methodist Clear Lake Hospital, Nassau Bay, Texas, United States.
Correspondence Address:
Richard W. Walker, Virtual Primary-Care Connection, LLC and Canopy Health, Houston, Texas, United States.
DOI:10.25259/SNI_486_2022
Copyright: © 2022 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Richard W. Walker1, Gustavo C. Roman2, Yi Jonathan Zhang3, Omar Acres4. Pleural effusion and respiratory compromise from spontaneous migration of a ventriculoperitoneal shunt catheter in a patient with normal-pressure hydrocephalus. 02-Sep-2022;13:393
How to cite this URL: Richard W. Walker1, Gustavo C. Roman2, Yi Jonathan Zhang3, Omar Acres4. Pleural effusion and respiratory compromise from spontaneous migration of a ventriculoperitoneal shunt catheter in a patient with normal-pressure hydrocephalus. 02-Sep-2022;13:393. Available from: https://surgicalneurologyint.com/surgicalint-articles/11849/
Abstract
Background: Ventriculoperitoneal shunt (VPS) insertion is one of the most common neurosurgical procedures done around the world to treat hydrocephalus. The occurrence of spontaneous migration of the peritoneal shunt catheter into the thoracic cavity is a very rare complication; we report here case number 27 of respiratory complications of a VPS in a patient with normal-pressure hydrocephalus (NPH).
Case Description: A 76-year-old woman with Alzheimer’s disease and anosognosia was diagnosed idiopathic NPH treated surgically with a VPS. Pleural effusion and pulmonary complications occurred 4 weeks after the insertion of the shunt due to the spontaneous migration of the peritoneal catheter of the VPS into the thoracic cavity. The hydrothorax of cerebrospinal fluid was drained and the distal catheter was removed and replaced. The patient made an uneventful recovery.
Conclusion: Due to the rarity of this complication, there are no standard corrective procedures. Some of the methods used to diagnose and successfully treat this rare complication of the VPS are presented.
Keywords: Alzheimer’s disease, Anosognosia, Hydrothorax, Normal-pressure hydrocephalus, Pleural effusion, Ventriculoperitoneal shunt
INTRODUCTION
Pleural effusion is a rare complication of a ventriculoperitoneal shunt (VPS) surgery with only 26 cases reported between 1977 and 2020[
CASE REPORT
This 76-year-old Black woman developed Alzheimer’s disease (AD) progressively and slowly over a 21-year period followed in the past 2 years by alterations in ambulation with slightly irregular gait. Over the next 18 months, she began to demonstrate further declines in cognition, bladder and stool incontinence, and worsening gait accompanied by loss of balance. Six months before the VPS placement, the patient was ambulatory but required constant assistance due to severe loss of balance and near-complete incontinence. The diagnosis of AD had been confirmed with neuropsychiatric testing and brain imaging in 2015. She received regular assessments from primary care, neurology, and geriatric psychiatry; medications included sertraline (100 mg QHS), quetiapine (25 mg QHS), lisinopril (25 mg QD), Vitamin D3 (5000 IUs QD), and 5-methyltetrahydrofolate (1330 mcg QD) due to a single-nucleotide polymorphism of 5-MTHFR C677T.
In February of 2021, the patient fell for the 3rd time in 4 months. The previous two falls were minor resulting in abrasions. However, with the third fall, she hit the right posterior side of her head, which prompted a computerized tomography (CT) of the head and neck in the emergency department. On review of the two CT scans (2015 and 2021), her neurologist (GCR) noted that ventriculomegaly had developed. The Evans index[
Her immediate postoperative recovery was progressing well with resolution of the incontinence, imbalance, and with cognitive improvement. To regain strength, mobility, and function, she received physical and occupational therapy (PT/ OT) and cognitive nursing care resulting in improvement in all of the above fields. Within the 4th week, some regression of the gait problem was observed and a repeated CT of the head showed an Evans index still at 0.38. Therefore, the flow of the VPS valve was increased by neurosurgery.
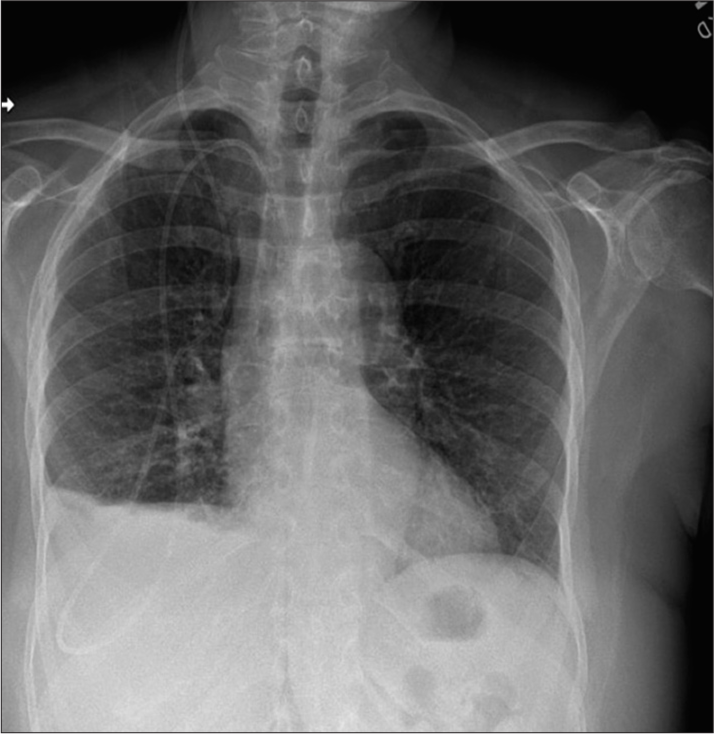
Within a day or two of the increased VPS flow, the patient began having dyspnea on exertion, shortness of breath, fatigue, and worsening cognition. Blood pressures remained normal, with repeated pulse oximetry (SaO2) levels of 97–98%. Her pulse rate during the postoperative period and during PT/OT ranged from the mid-90s to mid-80s, but then she began having tachycardia (pulse 100–110 bpm). She was now also experiencing mouth breathing and grunting with exertion. She developed 2+ bilateral pitting edema that did not resolve after resting supine for 8 h. No venous neck distention nor orthopnea was observed. The increasing respiratory challenges necessitated the discontinuation of all PT/OT. Physical examination by her PCP was unremarkable, including no clinical evidence of respiratory disease and normal laboratory workup. EKG showed left ventricular deviation. A 2-view chest X-ray showed, “new, small right costophrenic angle effusion. VP shunt tubing on the right. Tubing was coiled over the thoracic region with tip projected near the thoracolumbar junction; position of the distal shunt tubing is unclear” [
The interventional radiologist performed ultrasound-guided thoracentesis and removed from the right pleural space 400 cc of clear fluid consistent with CSF that was sent for extensive infectious workup and cytology review. Immediately postprocedure, the patient’s respiratory challenges began to resolve.
Neurosurgical treatment
The patient was transferred from the local hospital to a tertiary care hospital at the Texas Medical Center, where the original neurosurgeon (YJZ) replaced the VPS catheter. Surgical removal of the distal portion of the shunt is, generally, the main method of treatment for this complication. In this case, the distal portion of the shunt that extended from the level of the right clavicle into the thoracic cavity was removed and replaced with another catheter going cephalad from the peritoneum to reconnect with the proximal portion of the VPS at that point. After recent VPS placement, noncontrast CT head is routinely performed. Furthermore, radiographs of the skull, neck, chest, and abdomen (“shunt series”) are essential in evaluating the structural integrity and proper anatomical position of the system. Although very rare in occurrence, distal catheter migration can be readily detected, and appropriate corrective measures can be applied.
In addition to AD and NPH, this patient concomitantly suffered also from anosognosia or “impaired awareness of the illness.”[
The possible presence of anosognosia should be suspected in adult patients with cognitive or neurological deficits and when present it requires the clinical team to work closer with the family members and caregivers to prevent potential complications caused by the patients’ lack of complaints.[
DISCUSSION
Annually, approximately 30,000 VPS procedures are performed in the US alone.[
The mechanism of VPS pleural effusions is unclear and several hypotheses have been proposed including leaking of CSF into the thoracic cavity causing a pleural effusion through congenital diaphragmatic openings such as the Morgagni foramen and/or Bochdalek foramen,[
In general, the major differential diagnoses for pleural effusions include cardiovascular, infectious, malignancy, or iatrogenic causes. Other etiologies such as vasculitis, connective tissue diseases, and lymphatic abnormalities as well as other rarer causes would be less likely to present initially as an isolated pleural effusion without any other signs or symptoms of the coexisting disease condition.
The differential diagnosis of a unilateral pleural effusion may be further narrowed to primarily iatrogenic, infectious, or due to a malignancy. Pleural effusions due to heart failure are typically bilateral but when unilateral, they are usually right sided as seen in this case.
CONCLUSION
In this case review, we described the development of a CSF pleural effusion in a patient with a VPS malfunction due to migration of the distal catheter from the peritoneal space into the thoracic cavity through the diaphragm. Drainage of the CSF pleural effusion by an interventional radiologist followed by neurosurgical removal and replacement of the distal catheter from the level of the sternum to the pleural cavity with adjustment of the VPS valve allowed optimal drainage of the NPH.
Declaration of patient consent
Patient’s consent not required as patient’s identity is not disclosed or compromised.
Financial support and sponsorship
Doctor Román’s research is supported by the Jack S. Blanton Presidential Distinguished Chair for the Study of Neurological Disease, the Fondren Fund, and the Wareing Family Fund.
Conflicts of interest
There are no conflicts of interest.
References
1. Akyüz M, Uçar T, Göksu E. A thoracic complication of ventriculoperitoneal shunt: Symptomatic hydrothorax from intrathoracic migration of a ventriculoperitoneal shunt catheter. Br J Neurosurg. 2004. 18: 171-3
2. Aparicio-López C, Rodríguez-Rajo P, Sánchez-Carrión R, Ensenat A, García-Molina A. Rehabilitation of anosognosia in patients with unilateral visuospatial neglect. Rev Neurol. 2019. 69: 190-8
3. Bober J, Rochlin J, Marneni S. Ventriculoperitoneal shunt complications in children: An evidence-based approach to emergency department management. Pediatr Emerg Med Pract. 2016. 13: 1-22
4. Born M, Reichling S, Schirrmeister J. Pleural effusion: Beta-Trace protein in diagnosing ventriculoperitoneal shunt complications. J Child Neurol. 2008. 23: 810-2
5. Doh JW, Bae HG, Lee KS, Yun IG, Byun BJ. Hydrothorax from intrathoracic migration of a ventriculoperitoneal shunt catheter. Surg Neurol. 1995. 43: 340-3
6. Drake JM, Kestle JR, Milner R, Cinalli G, Boop F, Piatt J. Randomized trial of cerebrospinal fluid shunt valve design in pediatric hydrocephalus. Neurosurgery. 1998. 43: 294-303
7. Evans WA. An encephalographic ratio for estimating ventricular enlargement and cerebral atrophy. Arch Neurol Psychiatry. 1942. 47: 931-7
8. Fuller Torrey EF. Impaired Awareness of Illness: Anosognosia. Available from: https://mentalillnesspolicy.org/medical/anosognosia-studies.html [Last accessed on 2022 May 23].
9. Glöbl H, Kaufmann H. Shunts and complications. Prog Pediatr Radiol. 1978. 6: 231-71
10. Harischandra LS, Sharma A, Chattargee S. Shunt migration in ventriculoperitoneal shunting: A comprehensive review of literature. Neurol India. 2019. 67: 85-99
11. Hilmani S, Mesbahi T, Bouaggad A, Lakhdar A. A rare complication of ventriculoperitoneal shunt: Pleural effusion without intrathoracic ventriculoperitoneal shunt catheter. Surg Neurol Int. 2020. 11: 291
12. Kahle KT, Kulkarni AV, Limbrick DD, Warf BC. Hydrocephalus in children. Lancet. 2016. 387: 788-99
13. Karapolat S, Onen A, Sanli A. Intrathoracic migration of ventriculoperitoneal shunt: A case report. Cases J. 2008. 1: 42
14. Khan F, Rehman A, Shamim MS, Bari ME. Factors affecting ventriculoperitoneal shunt survival in adult patients. Surg Neurol Int. 2015. 6: 25
15. Mograbi DC, Morris R. Anosognosia. Cortex. 2018. 103: 385-6
16. Oliveira LM, Nitrini R, Román GC. Normal-pressure hydrocephalus: A critical review. Dement Neuropsychol. 2019. 13: 133-43
17. Orfei MD, Caltagirone C, Spalletta G. The evaluation of anosognosia in stroke patients. Cerebrovasc Dis. 2009. 27: 280-9
18. Park HY, Kim M, Suh CH, Lee DH, Shim WH, Kim SJ. Diagnostic performance and interobserver agreement of the callosal angle and Evans’ index in idiopathic normal pressure hydrocephalus: A systematic review and meta-analysis. Eur Radiol. 2021. 31: 5300-11
19. Patel AP, Dorantes-Argandar A, Raja AI. Cerebrospinal fluid hydrothorax without ventriculoperitoneal shunt migration in an infant. Pediatr Neurosurg. 2011. 47: 74-7
20. Porcaro F, Procaccini E, Paglietti MG, Schiavino A, Petreschi F, Cutrera R. Pleural effusion from intrathoracic migration of a ventriculo-peritoneal shunt catheter: Pediatric case report and review of the literature. Ital J Pediatr. 2018. 44: 42
21. Savolaine ER, Khimji T. Ventriculoperitoneal shunt failure resulting from complications of the thoracic segment of the shunt catheter. Case report. Clin Imaging. 1991. 15: 35-40
22. Taub E, Lavyne MH. Thoracic complications of ventriculoperitoneal shunts: Case report and review of the literature. Neurosurgery. 1994. 34: 181-3
23. Vacca VM. Ventriculoperitoneal shunts: What nurses need to know. Nursing. 2018. 48: 20-6
24. Yéboles RM, Vázquez L, Seoane M, Castro S, Ruiz B. Hydrothorax as a complication of a ventricle peritoneal shunt. A case report. Neurocirugía (Astur). 2017. 28: 202-6