- Department of Neurosurgery, NIMHANS, Bengaluru, Karnataka, India
- Department of Neuroradiology, NIMHANS, Bengaluru, Karnataka, India
Correspondence Address:
B. Indira Devi
Department of Neuroradiology, NIMHANS, Bengaluru, Karnataka, India
DOI:10.4103/sni.sni_414_17
Copyright: © 2018 Surgical Neurology International This is an open access journal, and articles are distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as appropriate credit is given and the new creations are licensed under the identical terms.How to cite this article: V. Gunal, Amey R. Savardekar, B. Indira Devi, Rose D. Bharath. Preoperative functional magnetic resonance imaging in patients undergoing surgery for tumors around left (dominant) inferior frontal gyrus region. 26-Jun-2018;9:126
How to cite this URL: V. Gunal, Amey R. Savardekar, B. Indira Devi, Rose D. Bharath. Preoperative functional magnetic resonance imaging in patients undergoing surgery for tumors around left (dominant) inferior frontal gyrus region. 26-Jun-2018;9:126. Available from: http://surgicalneurologyint.com/surgicalint-articles/preoperative-functional-magnetic-resonance-imaging-in-patients-undergoing-surgery-for-tumors-around-left-dominant-inferior-frontal-gyrus-region/
Abstract
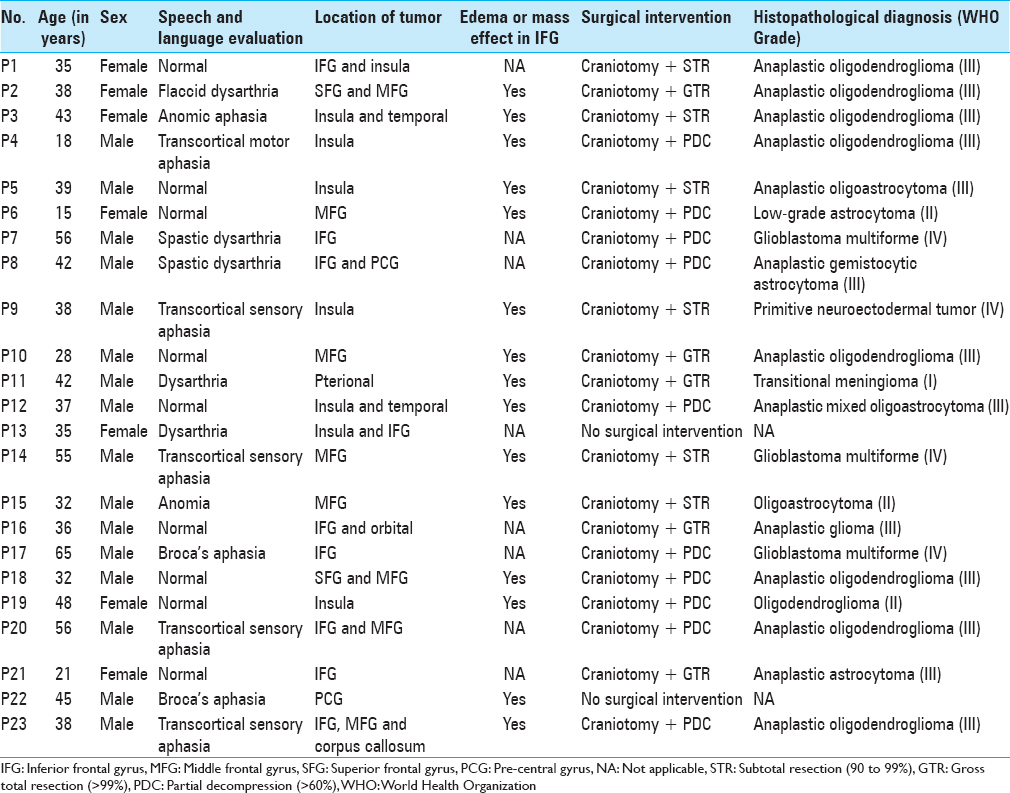
Background:Preoperative functional magnetic resonance imaging (fMRI) helps to preserve neurological function and ensure maximal tumor tissue excision. We studied the lateralization and localization of speech centers in select cases of tumors around the left (dominant) inferior frontal gyrus (IFG).
Methods:Twenty-three right-handed patients, harboring tumors involving the left (dominant) IFG or causing mass effect or edema extending onto the left IFG, were recruited over 17 months. Preoperatively, all patients underwent language and speech assessment followed by MRI and fMRI with paradigm (picture naming). Normative data for language fMRI was taken from the institute's imaging data bank.
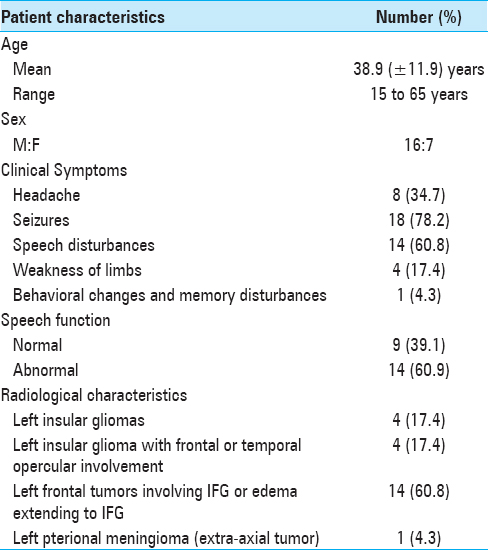
Results:The study included 23 patients [mean age: 38.9 (±11.9) years; M: F = 16:7; 9 – normal speech, 14 – abnormal speech]. Group analysis of controls showed significant activation in the region of interest (ROI) – left Brodmann's areas (BAs) 44,45. Group analysis of patients with normal speech showed no activation in the left BAs 44,45; however, activation was noted in the immediate adjacent areas, left BAs 13,47 and contralateral prefrontal cortex. Group analysis of patients with impaired speech showed no activation in BAs 44,45 or in the immediate adjacent areas.
Conclusions:Neuroplasticity in the brain may enable functional language areas to shift to adjoining or distant regions in the brain when the primary areas are involved by intrinsic tumors. This phenomenon is more likely in slow-growing compared to fast-growing tumors. Preoperative language fMRI may help us in identifying and protecting these areas during surgery.
Keywords: Cerebral re-organization, functional MRI, glioma surgery, left (dominant) inferior frontal gyrus, neuro-plasticity mechanisms, surgery for tumors involving the left inferior frontal gyrus
INTRODUCTION
Tumors located in eloquent cortical areas of the brain pose significant risks of neurological deficits during surgical extirpation. Current strategy of maximal safe resection is aimed at preserving neurological function to the fullest.[
In addition to the effects caused by the tumor itself, surgical intervention for tumor excision can cause additional deficits in these patients from either swelling or stroke or disruption of fiber tracts; these effects may be transient or permanent. Intraoperative cortical and subcortical stimulation is considered a high yield method for localizing language areas during neurosurgical procedures.[
Several studies have recognized the good spatial correlation between physiological intraoperative cortical mapping and anatomical localization of motor fMRI findings based on blood oxygenation level derived (BOLD) imaging.[
Our study aimed at studying the effects of tumors involving the left (dominant) IFG (commonly designated as the Broca's area) on the lateralization and localization of speech centers. It is an effort to understand the “functional reshaping” that occurs in tumors around the left (dominant) IFG and its effect on the language function in affected patients.
PATIENTS AND METHODS
A prospective study was conducted and patients were recruited over a period of 17 months from August 2012 to December 2013. Clearance from the institutional ethics committee was taken before commencement of the study. A written informed consent was obtained from the patients or relatives before recruitment for the study.
Inclusion criteria
Lesions involving the left IFG (gliomas, extension of insular glioma, mass effect, or edema involving the left IFG) Both genders Right-handed individuals Age 15–65 years Ability and willingness to participate in a follow-up fMRI study With normal/corrected hearing and vision.
Exclusion criteria
Neuropsychiatric conditions – major depressive disorder, schizophrenia, etc. Alcohol or drug dependence Chronic neurological illnesses Any contraindication to MRI Left-handed individuals Unwillingness to participate in the study or refusal of consent.
Assessment of speech and language
Patients underwent language and speech assessment at the department of speech pathology and audiology of our institution by a certified speech pathologist using Western Aphasia Battery (Indian version by Karanth et al.).[
Functional magnetic resonance imaging protocol
All patients underwent preoperative MRI and fMRI with paradigms designed at our institute.[
Functional magnetic resonance imaging paradigm
Picture naming: A block design was used. There were four blocks of “active condition” alternating with four blocks of “rest.” During the “active” condition, individual pictures were presented once every 4 seconds and the patient was asked to silently generate the name of the picture in his/her native language. During the rest condition, the patient would view a cross hair in the centre of the screen. The imaging protocol was as follows: TR = 4000 ms, TE = 30 ms, matrix = 64 × 64, number of dynamics = 85, slice thickness = 4 mm.
Clinical data collection
The nature and duration of symptoms including speech disturbances, behavioral symptoms, motor, or sensory symptoms were documented. Clinical examination included neurological assessment of higher mental functions (MMSE), bedside speech assessment, and assessment of cranial nerves, motor, sensory, and cerebellar system.
Preoperative radiological investigations
MRI of the brain and/or CT scan of head Functional BOLD MRI of the brain with the abovementioned paradigms.
Functional magnetic resonance imaging data analysis and visualization
Anatomical and functional images were processed using Statistical Parametric Mapping 8 (SPM8) software (Wellcome Trust Centre for Neuroimaging, London, UK). Preprocessing involved re-orientation of functional and structural images to anterior commissure-posterior commissure plane followed by realignment of images. When a dataset exhibited more than a 2-mm translation in the x, y, or z plane during the imaging procedure, it was not used for analysis. Normalization with the Montreal Neurological Institute (MNI) template was done for both structural and functional data such that processed data was superimposed on the structural images and inter-subject comparison could be done.[
Laterality index
Laterality index (LI) as a measure of hemispheric dominance was measured for all controls and all patients preoperatively using picture naming task. LI was measured for the whole hemisphere (excluding cerebellum) as well as for the specific ROI. ROI was determined as the frontal lobes bilaterally with other areas masked using a pre-formed mask.
LI was measured using the formula LI = tl−tr/tl+ tr,
where tl and tr are a sum of all t values in left and right hemisphere above a predefined individual threshold “T” which is mean of the 5% most activated voxels within each hemisphere or ROI. Sum of all the t values of all voxels above 50% of this mean was considered to be tl and tr for left and right, respectively.
RESULTS
The study was conducted from August 2012 to December 2013 over a duration of 17 months accrual time. Twenty-three patients, who fulfilled the inclusion criteria and were willing to participate in the study, were recruited. These included 16 males (~69%) and 7 females (~31%). The patients’ age ranged from 15 to 65 years (mean = 38.9 ± 11.9 years). Normative data for language fMRI was taken from the institute's data bank for imaging in healthy controls. The control group consisted of 9 males and 1 female. Age ranged from 26 to 51 years (mean = 33 years). Their minimum qualification was high school graduation, with 8 being college graduates.
Both the patient group as well as the control group gave no history of prior neurological illness and did not have any contraindications for MRI. The clinical and radiological profile of the study group is presented in
Clinical presentation
Patients presented with symptoms including headache, seizures, speech disturbances, weakness of limbs, or memory disturbances [
Speech and language assessment
All 23 patients underwent formal clinical speech-language assessment by a trained speech pathologist. The test included Western Aphasia Battery.[
Of the 23 patients, 9 patients had normal results on speech assessment. The rest had some form of speech/language abnormality, ranging from dysarthria to aphasia.
Radiological evaluation
All patients underwent routine CT scan and MRI of the brain as part of routine preoperative assessment. All, but one patient, had an intra-axial tumor. This single patient (P11) had an extra-axial tumor in the left pterional region. Among intra-axial tumors, four tumors were located exclusively in the insula, four tumors were predominantly insular with surrounding extension to either frontal or temporal lobes, and the remaining 14 were situated around the IFG, with edema extending to the IFG or with mass effect on the IFG.
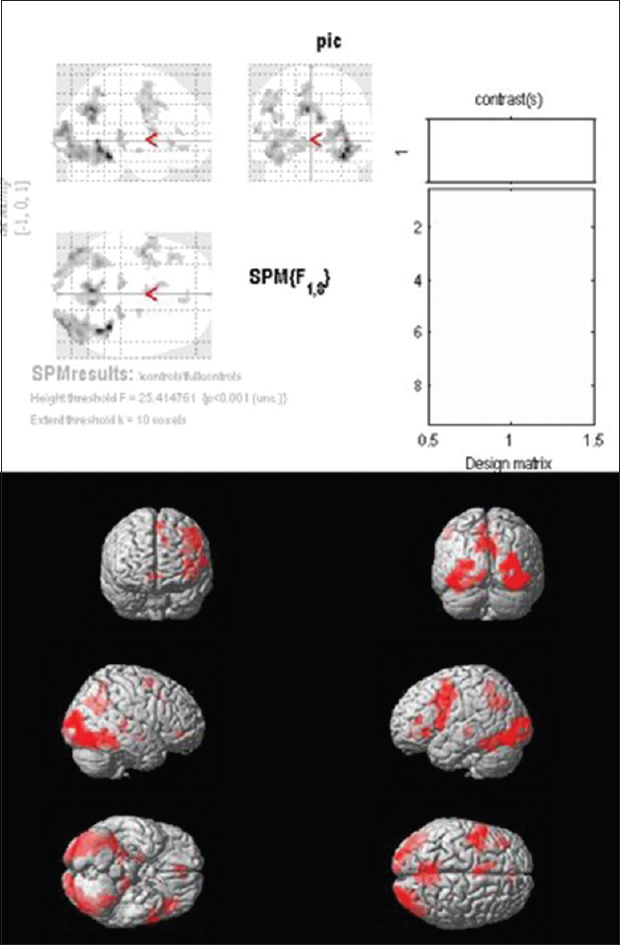
Functional magnetic resonance imaging analysis of controls
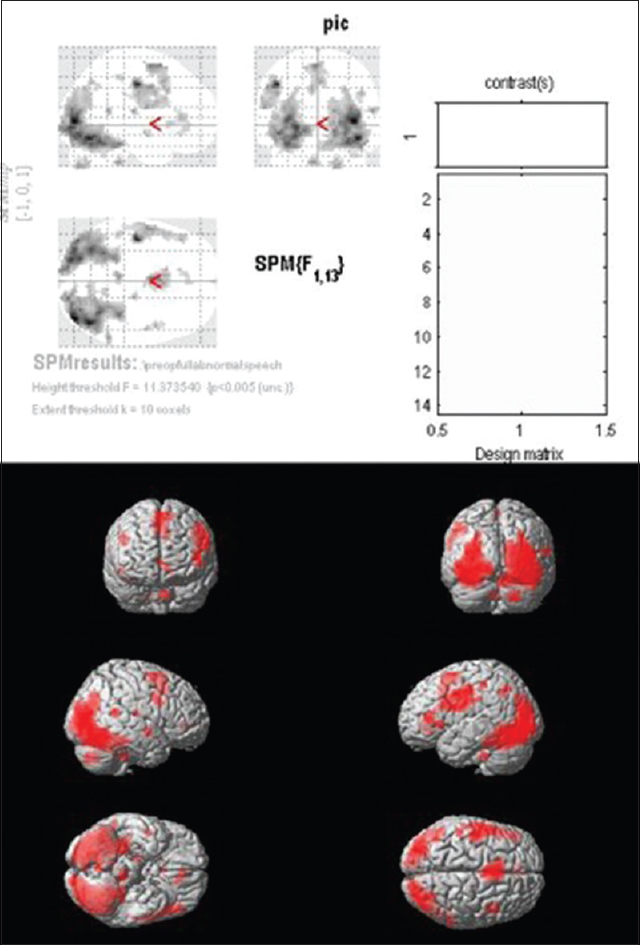
Group analysis of controls shows significant activation in the ROI (Brodmann areas 44,45). Significant activation was also noted in Brodmann area 6 in the precentral gyrus and Brodmann area 46 in the middle frontal gyrus of the left hemisphere [
Functional magnetic resonance imaging analysis of patients
The patients were divided into two groups. The first group did not display any speech deficits on testing. The second group had an identifiable speech/language deficit. fMRI results were analyzed for each group separately.
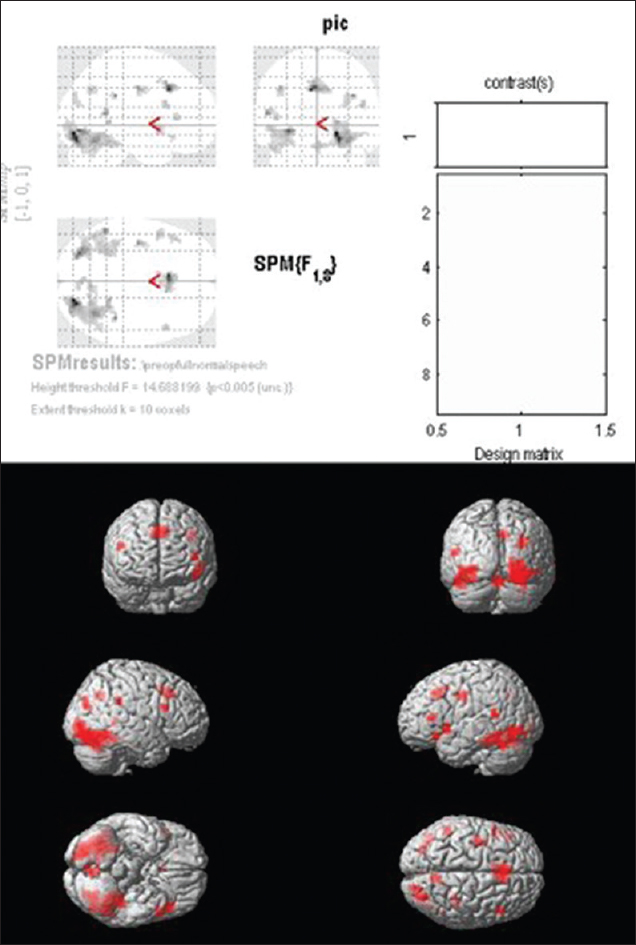
Group I (patients with normal speech and language): Group analysis of patients in this group showed no activation in Brodmann areas 44 and 45 on the left frontal lobe. Significant activations were found in left BA 13 and 47 which are in the areas immediately adjacent to Brodmann areas 44 and 45. Activations were also noted in the contralateral IFG (BA 9) [
Group II (patients with impaired speech/language): Group analysis of patients in this group showed no activation in left BA 44 and 45, i.e., the Broca's area. There was no significant activation even in the adjacent areas of the left IFG. Significant activation was noted in BA 6, i.e. the supplementary cortex [
The areas activated in the two groups, i.e. groups I and II, and the corresponding voxels of activation showed that Broca's area (BA 44 and 45) was not activated in either group [Figures
Laterality index
LI was calculated in both patients as well as controls for picture naming task with a P value of 0.001 for the whole hemisphere (excluding cerebellum) as well as for the ROI, as described above. Normality of data was tested before further analysis.
The mean hemispheric LI value for the controls was 0.22 ± 0.23, and the mean ROI LI was 0.36 ± 0.33. The mean hemispheric LI across the patient group was 0.19 ± 0.18, and mean ROI LI across patient group was 0.29 ± 0.38.
The preoperative LI values of controls were compared to the patient group using independent-sample t-test. There was no statistical significance between LI values of the two groups in either the hemispheric or ROI LIs. The LIs were compared across controls and patients using analysis of variance (ANOVA) test, with the patient group divided into those who had normal speech and those who had speech/language deficits. This analysis showed that there was no significant difference in LI values (both hemispheric and ROI) among patients who did not have speech deficits, those who had speech deficits, and controls.
Surgical intervention and histopathological diagnoses
Of the 23 patients included in the patient group, 21 underwent open surgical intervention. Two patients refused surgical intervention. Out of 21 surgical interventions, only six patients had a gross total resection of the tumor, five patients had subtotal resection, and ten patients underwent decompression of the tumor. The histopathological diagnosis was grade III astrocytoma in 13 patients, grade II astrocytoma in 3 patients, grade IV astrocytoma (glioblastoma multiforme) in 3 patients, primitive neuroectodermal tumor (PNET) in 1 patient, and transitional meningioma (grade I) in 1 patient. Patient-specific data is presented in
DISCUSSION
The canonical view that the brain has a fixed functional organization that is similar in all individuals has been challenged by recent advances in functional brain imaging that have demonstrated large interindividual variability in the functional organization of the brain.[
When an intracranial lesion infiltrates or impinges on an eloquent region/cortex of the human brain, plasticity is postulated as the mechanism by which the brain re-organizes connectivity for functional areas and possibly shifts the function from one initially eloquent (functional) area to another noneloquent (nonfunctional) area of the brain. With the help of preoperative fMRI studies, we wanted to study cases of intracranial tumors involving or compressing the predominant motor language area, i.e. the left (dominant) IFG region and we also wanted to assess the areas of functional language cortex in this setting.
Our normative data showed significant activation in the region of interest (ROI), i.e., the BAs 44 and 45 on the left side. In addition to this, significant activation was also noted in BA 6 in the precentral gyrus and BA 46 in the middle frontal gyrus of the left hemisphere. This substantiates the previously established concept, that in normal individuals, the motor speech areas are located in the BAs 44 and 45 on the left side.
In patients harboring a tumor in this locale but with clinically preserved speech and language function (n = 9), group analysis showed that there was no residual activation in BAs 44 and 45 of the left frontal lobe. However, significant activations were found in the immediate adjacent areas – left BAs 13 and 47. Activations were also noted in the right IFG (BA 9). In tumor patients with impaired/absent speech and language function (n = 14), group analysis showed that in addition to the absence of activation in the left BA 44 and 45, there was no such activation in the adjacent cortex of BA 13 and 47. However, there was significant activation noted in the supplementary cortex (BA 6).
In the present study, the LI measured for the whole hemisphere ranged from −0.04 to 0.33 in patients who had normal speech with a mean of 0.23. This implies that the majority of patients with intact speech had left-sided lateralization. Two patients with intact speech had LI in the range of −0.1 to 0.1, indicating bilateral representation of speech areas.
It has been stated that slow-growing lesions permit the induction progressive functional reshaping of brain networks.[
Our study shows that in the group of patients harboring tumors in and around the left IFG (BA 44 and 45), with preserved speech and language function, the activation had shifted from BAs 44 and 45 to the adjacent BAs 13 and 47, and even to the contralateral IFG (BA 9). Significant activity was noted in the adjacent perilesional areas including BA 47, 9, and 13 in patients who had intact speech/language compared to patients who had speech/language deficits. These findings are in line with the theories of functional redistribution mentioned above.
Furthermore, bilateral dominance, as observed in the LI studies in two patients, indicates recruitment of homologous structures in the right hemisphere, which may account for preserved speech/language in a subgroup of patients.
Yousry et al. demonstrated shifting of hand-motor cortex to perilesional areas in a case of a tumor affecting the hand area of the motor cortex.[
“Remote plasticity” has also been described in patients harboring intracerebral gliomas within the language system. fMRI revealed speech-related activations were (i) in the left superior temporal gyrus following tumor invasions of Broca's area[
Kośla et al. showed that in patients with LGGs located in the left frontal lobe near the frontal operculum of insula, 75% of the patients showed activation of Broca's area in the right hemisphere with half of the LGG tumors showing a strong right-sided lateralization (LI = −1), whereas in the other half the LI was left-sided (strong: LI = 1 and weak: LI = 0.28).[
The fMRI findings in the set of 9 patients with intact speech/language function demonstrate that a multitude of plasticity mechanisms work synchronously to reorganize functional brain networks and shift foci to adjacent as well as remote cortical areas within the brain to maintain language function.
The findings in our cohort subset of 14 study participants with impaired speech/language function demonstrate that plasticity mechanisms have not been able to compensate for the loss of function. This may be attributable to rapid progression of the lesions (outpacing the plasticity mechanisms) or the eventual involvement of the adjacent areas (to which the language function had shifted) by the tumor.[
Roux et al. analyzed the utility of preoperative fMRI for identifying functional language areas in patients with brain tumors.[
Giussani et al. reviewed the pertinent literature (nine language brain mapping studies) studying the reliability of preoperative language fMRI in patients operated on for brain tumors by comparing language fMRI with direct cortical stimulation (DCS).[
Limitations of our study
The paradigms designed and administered in the present study included only picture naming as majority of the patients could not perform other tasks to test other domains of language. This partly depends on the educational status of the participant as well as the intellectual level and partly on the complexity of tasks. Hence, short and effective paradigms with high sensitivity and specificity which can be easily performed are better suited in such patients.
Because the performance of tasks was limited in the patient group, uncorrected group results (P < 0.005) with a cluster size of 10 was used for group analysis as this was the least threshold at which we could observe any changes. Thus, our results might contain a higher percentage of false positives. As similar thresholds were applied for both groups (control and patient) and we did not get any significant difference between the groups, this might not affect the results between the two patient groups. Other techniques such as seed to voxel connectivity or resting state fMRI could be used to improve this analysis.[
Further, as demonstrated in
Our study, conducted with a 3.0-Tesla MR scanner is a purely preoperative study. The fact that these preoperative language fMRI results were not validated by further intraoperative DCS and were not compared with postoperative language fMRI results in the same patient group is another limitation of our study.
CONCLUSION
Preoperative fMRI should be considered in the work-up of patients with intra-axial or extra-axial lesions in the vicinity of functional language areas in the human brain for surgical planning and counseling. Due to plasticity mechanisms, select functional areas may have migrated to adjacent or distant regions in the brain, as shown in our study. fMRI with evaluation of language function may help us to identify and protect these areas during surgery. In this aspect, fMRI may act as an adjunct to other “function-preserving” modalities such as intraoperative cortical and subcortical stimulation and mapping.[
Funding: No funding was received for this research.
Conflict of Interest: All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical approval: All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent: Informed consent was obtained from all individual participants included in the study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Bharath RD. Functional MRI: Genesis, state of the art and the sequel. Indian J Radiol Imaging. 2014. 24: 6-12
2. Dalal PK, Sivakumar T. Cognitive psychiatry in India. Indian J Psychiatry. 2010. 52: S128-35
3. DeAngelis LM. Brain tumors. N Engl J Med. 2001. 344: 114-23
4. Duffau H. Brain plasticity: From pathophysiological mechanisms to therapeutic applications. J Clin Neurosci. 2006. 13: 885-97
5. Duffau H. Lessons from brain mapping in surgery for low-grade glioma: Insights into associations between tumour and brain plasticity. Lancet Neurol. 2005. 4: 476-86
6. Dym RJ, Burns J, Freeman K, Lipton ML. Is functional MR imaging assessment of hemispheric language dominance as good as the Wada test?.: A meta-analysis. Radiology. 2011. 261: 446-55
7. Fandino J, Kollias SS, Wieser HG, Valavanis A, Yonekawa Y. Intraoperative validation of functional magnetic resonance imaging and cortical reorganization patterns in patients with brain tumors involving the primary motor cortex. J Neurosurg. 1999. 91: 238-50
8. Giussani C, Roux FE, Ojemann J, Sganzerla EP, Pirillo D, Papagno C. Is preoperative functional magnetic resonance imaging reliable for language areas mapping in brain tumor surgery?. Review of language functional magnetic resonance imaging and direct cortical stimulation correlation studies. Neurosurgery. 2010. 66: 113-20
9. Heiss WD, Thiel A, Kessler J, Herholz K. Disturbance and recovery of language function: Correlates in PET activation studies. Neuroimage. 2003. 20: S42-9
10. Holodny AI, Schulder M, Ybasco A, Liu WC. Translocation of Broca's area to the contralateral hemisphere as the result of the growth of a left inferior frontal glioma. J Comput Assist Tomogr. 2002. 26: 941-3
11. Huang CM, Lee SH, Hsiao IT, Kuan WC, Wai YY, Ko HJ. Study-specific EPI template improves group analysis in functional MRI of young and older adults. J Neurosci Methods. 2010. 189: 257-66
12. Karanth P ND, Pandit R, Shivashankar N, Chopra J. Cross cultural studies of aphasia. Modern Trends in Neurology: Churchill Livingstone. 1995. p. 95-105
13. Karbe H, Thiel A, Weber-Luxenburger G, Herholz K, Kessler J, Heiss WD. Brain plasticity in poststroke aphasia: What is the contribution of the right hemisphere?. Brain Lang. 1998. 64: 215-30
14. Kosla K, Pfajfer L, Bryszewski B, Jaskolski D, Stefanczyk L, Majos A. Functional rearrangement of language areas in patients with tumors of the central nervous system using functional magnetic resonance imaging. Pol J Radiol. 2012. 77: 39-45
15. Kundu B, Penwarden A, Wood JM, Gallagher TA, Andreoli MJ, Voss J. Association of functional magnetic resonance imaging indices with postoperative language outcomes in patients with primary brain tumors. Neurosurg Focus. 2013. 34: E6-
16. Levesque MF, Wilson CL, Behnke EJ, Zhang J ×. Accuracy of MR-guided stereotactic electrode implantation. 1. Preimplantation correlation with the Talairach system. Stereotact Funct Neurosurg. 1990. 54-55: 51-5
17. Manglore S, Bharath RD, Panda R, George L, Thamodharan A, Gupta AK. Utility of resting fMRI and connectivity in patients with brain tumor. Neurol India. 2013. 61: 144-51
18. Meyer PT, Sturz L, Schreckenberger M, Spetzger U, Meyer GF, Setani KS. Preoperative mapping of cortical language areas in adult brain tumour patients using PET and individual non-normalised SPM analyses. Eur J Nucl Med Mol Imaging. 2003. 30: 951-60
19. Petrovich NM, Holodny AI, Brennan CW, Gutin PH. Isolated translocation of Wernicke's area to the right hemisphere in a 62-year-man with a temporo-parietal glioma. AJNR Am J Neuroradiol. 2004. 25: 130-3
20. Roux FE, Boulanouar K, Lotterie JA, Mejdoubi M, LeSage JP, Berry I. Language functional magnetic resonance imaging in preoperative assessment of language areas: correlation with direct cortical stimulation. Neurosurgery. 2003. 52: 1335-
21. Taniguchi M, Kato A, Ninomiya H, Hirata M, Cheyne D, Robinson SE. Cerebral motor control in patients with gliomas around the central sulcus studied with spatially filtered magnetoencephalography. J Neurol Neurosurg Psychiatry. 2004. 75: 466-71
22. Thiel A, Habedank B, Herholz K, Kessler J, Winhuisen L, Haupt WF. From the left to the right: How the brain compensates progressive loss of language function. Brain Lang. 2006. 98: 57-65
23. Thiel A, Herholz K, Koyuncu A, Ghaemi M, Kracht LW, Habedank B. Plasticity of language networks in patients with brain tumors: A positron emission tomography activation study. Ann Neurol. 2001. 50: 620-9
24. Thiel A, Herholz K, von Stockhausen HM, van Leyen-Pilgram K, Pietrzyk U, Kessler J. Localization of language-related cortex with 15O-labeled water PET in patients with gliomas. Neuroimage. 1998. 7: 284-95
25. Tzourio-Mazoyer N, Josse G, Crivello F, Mazoyer B. Interindividual variability in the hemispheric organization for speech. Neuroimage. 2004. 21: 422-35
26. Weng HH, Noll KR, Johnson JM, Prabhu SS, Tsai YH, Chang SW. Accuracy of presurgical functional MR imaging for language mapping of brain tumors: A systematic review and meta-analysis. Radiology. 2018. 286: 512-23
27. Wunderlich G, Knorr U, Herzog H, Kiwit JC, Freund HJ, Seitz RJ. Precentral glioma location determines the displacement of cortical hand representation. Neurosurgery. 1998. 42: 18-26
28. Yousry TA, Schmid UD, Jassoy AG, Schmidt D, Eisner WE, Reulen HJ. Topography of the cortical motor hand area: Prospective study with functional MR imaging and direct motor mapping at surgery. Radiology. 1995. 195: 23-9