- Department of Neurosurgery, Hakodate Shintoshi Hospital, Hakodate, Japan
- Department of Neurology, Hakodate Municipal Hospital, Hakodate, Japan
- Department of Neurosurgery, Sapporo Medical University, Sapporo, Hokkaido, Japan
Correspondence Address:
Koichi Haraguchi
Department of Neurosurgery, Sapporo Medical University, Sapporo, Hokkaido, Japan
DOI:10.4103/sni.sni_152_18
Copyright: © 2018 Surgical Neurology International This is an open access journal, and articles are distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as appropriate credit is given and the new creations are licensed under the identical terms.How to cite this article: Tomoyoshi Kuribara, Koichi Haraguchi, Shunya Ohtaki, Tadakazu Shimizu, Nobuki Matsuura, Kazumi Ogane, Yasunori Maruo, Noriyuki Yokoyama, Takeshi Mikami, Takeo Itou, Nobuhiro Mikuni. Progressive edematous lesions in subacute phase after neuroendovascular therapy. 28-Aug-2018;9:173
How to cite this URL: Tomoyoshi Kuribara, Koichi Haraguchi, Shunya Ohtaki, Tadakazu Shimizu, Nobuki Matsuura, Kazumi Ogane, Yasunori Maruo, Noriyuki Yokoyama, Takeshi Mikami, Takeo Itou, Nobuhiro Mikuni. Progressive edematous lesions in subacute phase after neuroendovascular therapy. 28-Aug-2018;9:173. Available from: http://surgicalneurologyint.com/surgicalint-articles/8990/
Abstract
Background:The appearance of edematous lesions in the subacute phase is a rare complication following neuroendovascular therapy. Effective management of these lesions remains unclear. In this report, a case with progressive edematous lesions in the subacute phase after neuroendovascular therapy was described, and the clinical features and therapeutic strategies were discussed.
Case Description:A 54-year-old female with a large, right cavernous internal carotid artery aneurysm was treated with a flow diverter. Left hemiparesis developed 15 days after the procedure, and multiple edematous lesions in areas of prior catheter procedures were revealed on radiological findings. Steroid pulse therapy was employed, and the lesions were gradually reduced without any additional neurological deficits. No recurrence was recognized in the follow-up study.
Conclusion:In some reports, pathological findings indicate that these lesions result from the presence of foreign bodies, and emboli could be caused by cotton fibers or hydrophilic polymers used as surface coatings on endovascular catheters. In this case, the edematous lesions were most likely caused by hydrophilic polymer emboli. Steroid pulse therapy had a beneficial effect on the lesions. It is important to effectively manage prescribed periods after the procedure to avoid such a rare complication.
Keywords: Flow diverter, foreign body emboli, hydrophilic polymer, neuroendovascular therapy
INTRODUCTION
Neuroendovascular therapy is commonly used for intracranial aneurysms. Recently, the complication rate of this procedure is relatively low and mostly includes thromboembolic events and intraprocedure aneurysm rupture. Foreign body emboli in the distal cerebral arteries during neuroendovascular therapy are occasionally encountered, and are a potential cause of ischemic stroke and parenchymal hemorrhage. Of these cases, there have been only a few reports concerning focal edematous lesions.[
CASE REPORT
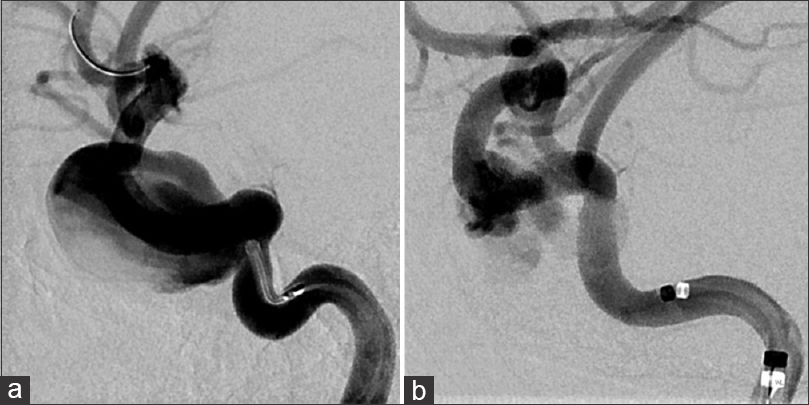
A 54-year-old female with a history of nephrotic syndrome and hypertension presented with a persistent headache at her local clinic. A right thrombosed cavernous internal carotid artery aneurysm measuring 22.5 × 19.7 × 19.2 mm was detected on computed tomography angiography (CTA). She was introduced to our hospital and underwent flow diverter deployment for the aneurysm with a pipeline embolization device (PED) (Covidien, Irvine, California, USA). Under general anesthesia, a 5 Fr Navien 058 Intracranial Support Catheter (Covidien, Irvine, California, USA) and a Marksman Catheter (Covidien, Irvine, California, USA) were advanced over the neck of the aneurysm using a 0.014 inch Asahi Chikai microguidewire (Asahi Intecc, Aichi, Japan) through the shuttle sheath. A 4.75/20 mm PED was deployed to cover the neck of the aneurysm through the triaxial system, and the stent was re-sheathed twice. The procedure was performed in 61 min without any complications [Figure
Figure 2
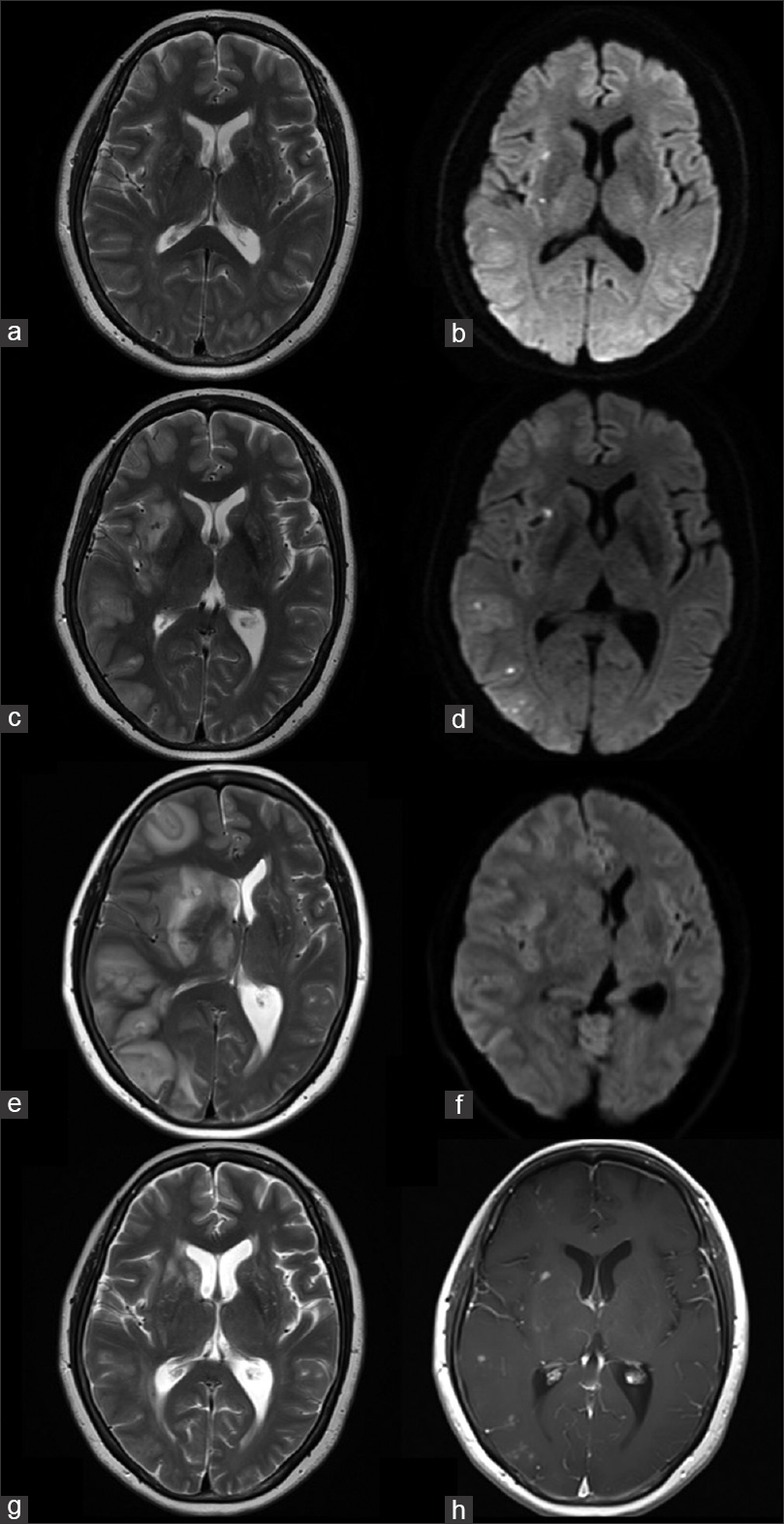
T2-weighted magnetic resonance imaging after the procedure showed no edema (a) and diffusion-weighted imaging showed a few hyperintensities in the right hemisphere (b). Magnetic resonance imaging 28 days after the procedure showed multiple edematous lesions in the right hemisphere (c), although there were no remarkable changes in diffusion-weighted imaging (d). Magnetic resonance imaging 42 days after the procedure showed a progression of edemas (e), though there were no remarkable changes on diffusion-weighted imaging (f). Magnetic resonance imaging after three courses of steroid pulse therapy (81 days after the procedure) showed a regression of edemas (g), and Gd-enhanced T1-weighted magnetic resonance imaging showed residual multiple enhancing lesions (h)
DISCUSSION
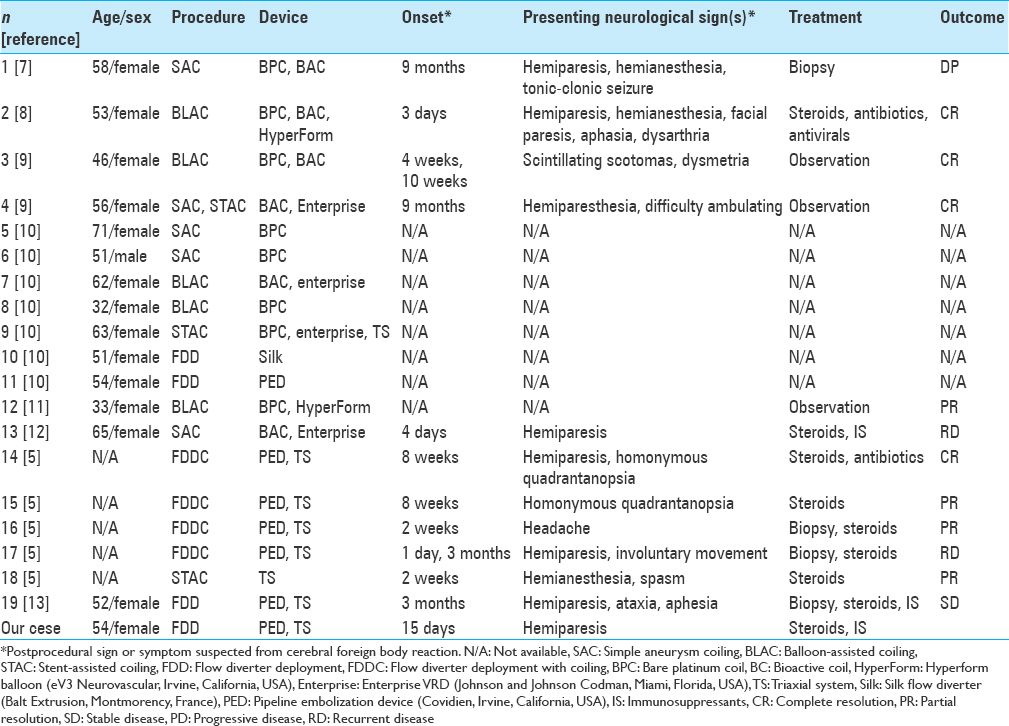
Clinical features of edematous lesions following neuroendovascular therapy are summarized in
Cerebral foreign body emboli could be related to intercatheter friction, made increasingly common by the adoption of the triaxial catheter technique for deployment of a flow diverter. For that reason, the incidence of this complication is expected to increase in the near future. Treatment with a flow diverter is currently on the rise for both large and giant unruptured aneurysms. Advanced catheter stability is necessary to obtain a precise and secure delivery of the device. To achieve this stability, it is customary to use the triaxial catheter technique, which can lead to increased friction between catheters and may create a predisposition for small emboli from the coating of the catheters. Our case might also be associated with the use of tight-fitting catheter combinations to deploy the device. The accumulation of similar cases is necessary to more fully understand the technical factors involved.
CONCLUSION
A case with progressive edematous lesions in the subacute phase after neuroendovascular therapy was described. In this case, the edematous lesions were most likely caused by hydrophilic polymer emboli. Steroid pulse therapy had beneficial effect on the lesions. It is important to effectively manage prescribed periods after the procedure to avoid such a rare complication.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form the patient(s) has/have given his/her/their consent for his/her/their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Barnwell SL, D’Agostino AN, Shapiro SL, Nesbit GM, Kellogg JX. Foreign bodies in small arteries after use of an infusion microcatheter. AJNR Am J Neuroradiol. 1997. 18: 1886-9
2. Cruz JP, Marotta T, O’Kelly C, Holtmannspötter M, Saliou G, Willinsky R. Enhancing brain lesions after endovascular treatment of aneurysms. AJNR Am J Neuroradiol. 2014. 35: 1954-8
3. Deguchi K, Kawahara Y, Deguchi S, Morimoto N, Kurata T, Ikeda Y. A patient develops transient unique cerebral and cerebellar lesions after unruptured aneurysm coiling. BMC Neurol. 2015. 15: 49-
4. Deus-Silva L, Lum C, De Meulemeester C, dos Santos MP. Severe aggressive acute disseminated encephalomyelitis-like reaction after aneurysm coiling. Neurosurgery. 2010. 66: E222-3
5. Fealey ME, Edwards WD, Giannini C, Piepgras DG, Cloft H, Rihal CS. Complications of endovascular polymers associated with vascular introducer sheaths and metallic coils in 3 patients, with literature review. Am J Surg Pathol. 2008. 32: 1310-6
6. Grewal SS, López Del Valle EM, Gupta V, Ramon N, Freeman WD, Tawk RG. Neurological changes with abnormal brain reactivity following coiling of cerebral aneurysm. Possible reactivity to endovascular devices and material?. J Vasc Interv Neurol. 2015. 8: 28-36
7. Lorentzen AO, Nome T, Bakke SJ, Scheie D, Stenset V, Aamodt AH. Cerebral foreign body reaction after carotid aneurysm stenting. Interv Neuroradiol. 2016. 22: 53-7
8. Mehta RI, Mehta RI. Polymer-induced central nervous system complications following vascular procedures: Spectrum of iatrogenic injuries and review of outcomes. Hum Pathol. 2016. 53: 178-90
9. Mehta RI, Mehta RI, Solis OE, Jahan R, Salamon N, Tobis JM. Hydrophilic polymer emboli: An under-recognized iatrogenic cause of ischemia and infarct. Mod Pathol. 2010. 23: 921-30
10. Shannon P, Billbao JM, Marotta T, Terbrugge K. Inadvertent foreign body embolization in diagnostic and therapeutic cerebral angiography. AJNR Am J Neuroradiol. 2006. 27: 278-82
11. Shapiro M, Ollenschleger MD, Baccin C, Becske T, Spiegel GR, Wang Y. Foreign body emboli following cerebrovascular interventions: Clinical, radiographic, and histopathologic features. AJNR Am J Neuroradiol. 2015. 36: 2121-6
12. Silberman J, Cravioto H, Feigin I. Foreign body emboli following cerebral angiography. Trans Am Neurol Assoc. 1960. 85: 123-5
13. Skolarus LE, Gemmete JJ, Braley T, Morgenstern LB, Pandey A. Abnormal white matter changes after cerebral aneurysm treatment with polyglycolic-polylactic acid coils. World Neurosurg. 2010. 74: 640-4