- Spine Deformity Unit, Royal National Orthopaedic Hospital, London, Stanmore, United Kingdom.
Correspondence Address:
Nitin Adsul, Spine Deformity Unit Royal National Orthopaedic Hospital, London, Stanmore, United Kingdom.
DOI:10.25259/SNI_1042_2021
Copyright: © 2021 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Nitin Adsul, Idris Hamim, Mohan Banglore, Robert Lee. Reactivation of herpes simplex labialis following adult spine deformity correction surgery. 16-Nov-2021;12:562
How to cite this URL: Nitin Adsul, Idris Hamim, Mohan Banglore, Robert Lee. Reactivation of herpes simplex labialis following adult spine deformity correction surgery. 16-Nov-2021;12:562. Available from: https://surgicalneurologyint.com/surgicalint-articles/11227/
Abstract
Background: A depressed host defense is a major contributor to the oral shedding of herpes simplex virus (HSV) type 1. Here, we present an instance in which herpes simplex labialis was reactivated following major spinal deformity surgery.
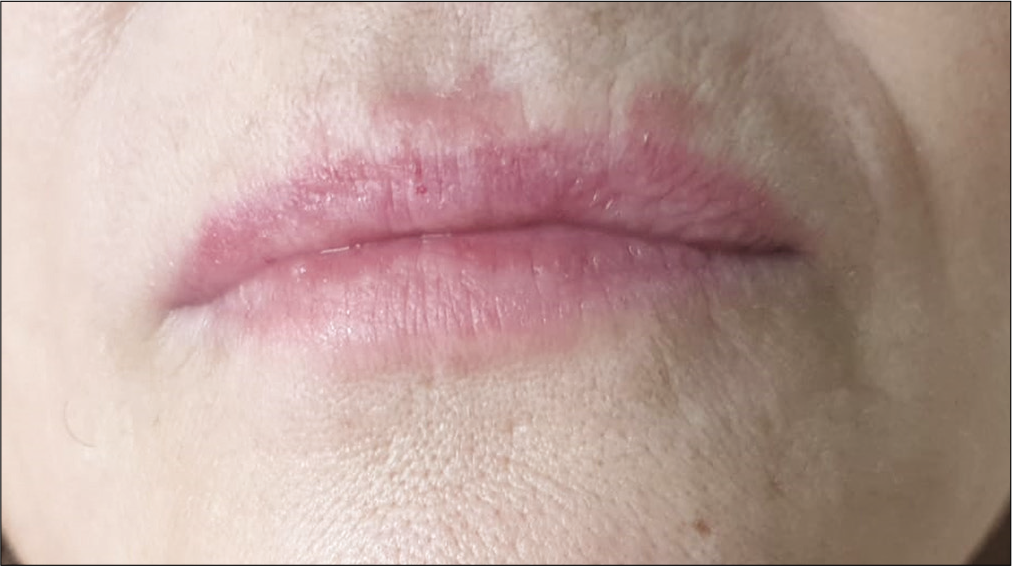
Case Description: A 59-year-old female underwent spinal deformity correction for lumbar degenerative scoliosis. On postoperative days 2–3, she presented with pyrexia (38°C) and tachycardia (94/min); by day 5 she had multiple ulcers around her lips and was HSV IgG positive. She had a remote history of herpes simplex I infection 7 years previously. Once started on oral acyclovir, the lesions improved, and by day 15 postoperative, her pyrexia and all lesions completely resolved.
Conclusion: HSV-1 should be suspected in patients with a previous history of HSV and postoperative pyrexia. Adequate prophylactic administration of acyclovir should result in resolution of these outbreaks, in this case, attributed to overly extensive spinal deformity surgery.
Keywords: Herpes labialis, Herpes simplex virus, Spine deformity surgery
INTRODUCTION
Herpes simplex virus (HSV) type 1 is a latent infection of the dorsal root ganglia.[
CASE REPORT
A 59-year-old female underwent lumbar surgery for degenerative scoliosis. Stage I involved L1-L2, L2-L3, L3-L4, and L4-L5 oblique lateral interbody fusions and a L5-S1 anterior lumbar interbody fusion. Stage II included posterior fixation from L1 to the pelvis [
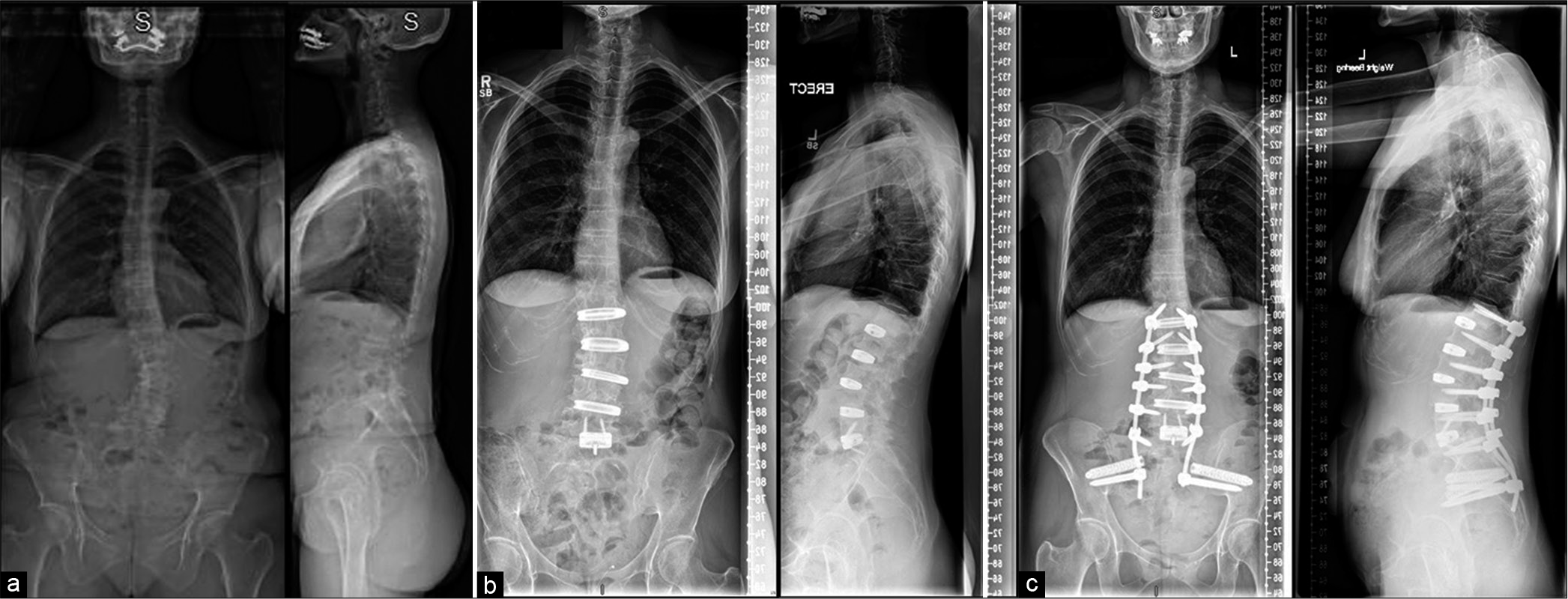
Figure 1:
X-rays of patient: (a) preoperative image of the patient showing adult degenerative scoliosis deformity. (b) Postoperative images after Stage I showing deformity correction with multiple oblique lumbar interbody fusions and anterior lumbar interbody fusion at L5-S1 level. (c) Postoperative images after Stage II showing posterior L1 to pelvis fixation.
Two to three days following these overly extensive procedures (i.e., Stages I and II), she presented with pyrexia (38°C) and tachycardia (94/min); on the 5th postoperative day, she developed multiple ulcers around the lips. As she had a history of herpes simplex I infection 7 years ago, and the antigen tests were positive for HSV IgG, she received a 15-day course of oral acyclovir. During this period, her pyrexia gradually resolved, and her oral lesions improved [
DISCUSSION
Reactivation of herpes simplex labialis type 1 (HSV) after major spine deformity surgery is rare. In this case, it was likely attributed to transient immunosuppression secondary to overly extensive spinal surgery. Stage I was four-level oblique lumbar interbody fusions (L1-L5) and L5-S1 anterior lumbar interbody fusion. Stage II was posterior pedicle screw instrumentation (L1-S2) and sacroiliac joint fusion using bedrock technique.
Etiology of HSV-1 postoperative spine infection
The massive stress associated with such overly extensive spinal deformity procedures contribute to her HSV-1 outbreak. This was associated with oral ulceration and symptoms/signs of neural hyperactivity characterized by anxiety, tachycardia, shallow breathing, and hypoxia. It is critical to identify and treat HSV-1 infections in postoperative patients, as in this case, as HSV-1 may cause necrotizing encephalitis[
Early antiviral therapy with acyclovir for HSV-1
Early antiviral therapy with acyclovir significantly reduces morbidity and mortality due to HSV encephalitis. To prevent relapse, a typical course of treatment lasts 21 days. In our patient, the treatment was stopped after 15 days when the symptoms resolved.[
Consideration of prophylaxis with acyclovir in patients with histories of oral HSV-1
Given the strong correlation between surgical procedures and HSV infections, prophylactic use of acyclovir in patients with a history of HSV orolabial ulcers should be considered routinely.[
CONCLUSION
Patients undergoing major spinal surgery (i.e., some overly extensive) may become transiently immunosuppressed, resulting reactivation of HSV-1. For patients, with documented postoperative HSV-1, a course of acyclovir should be administered; only select patients considered at increased risk for postoperative HSV-1 should prophylactically receive this therapy.
Declaration of patient consent
Patient’s consent not required as patients identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Gershon AA, Gershon MD, Breuer J, Levin MJ, Oaklander AL, Griffiths PD. Advances in the understanding of the pathogenesis and epidemiology of herpes zoster. J Clin Virol. 2010. 48: S2-7
2. Kennedy P, Chaudhuri A.editors. Herpes simplex encephalitis. United Kingdom: BMJ Publishing Group Ltd.; 2002. p.
3. Lungu O, Annunziato PW, Gershon A, Staugaitis SM, Josefson D, LaRussa P. Reactivated and latent varicellazoster virus in human dorsal root ganglia. Proc Natl Acad Sci USA. 1995. 92: 10980-4
4. Pazin GJ, Ho M, Jannetta PJ. Reactivation of herpes simplex virus after decompression of the trigeminal nerve root. J Infect Dis. 1978. 138: 405-9
5. Porteous C, Bradley JA, Hamilton D, Ledingham I, Clements GB, Robinson CG. Herpes simplex virus reactivation in surgical patients. Crit Care Med. 1984. 12: 626-8
6. Xu F, Schillinger JA, Sternberg MR, Johnson RE, Lee FK, Nahmias AJ. Seroprevalence and coinfection with herpes simplex virus Type 1 and Type 2 in the United States, 1988-1994. J Infect Dis. 2002. 185: 1019-24