- Clinical Professor of Neurosurgery, School of Medicine, State University of NY at Stony Brook, NY and c/o Dr. Marc Agulnick 1122 Frankllin Avenue Suite 106, Garden City, NY 11530, USA.
Correspondence Address:
Nancy E. Epstein MD, Clinical Professor of Neurosurgery, School of Medicine, State University of NY at Stony Brook, NY and c/o Dr. Marc Agulnick 1122 Frankllin Avenue Suite 106, Garden City, NY 11530, USA.
DOI:10.25259/SNI_278_2022
Copyright: © 2022 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Nancy E. Epstein. Review/perspective on hysterical paralysis: A diagnosis of exclusion for spinal surgeons. 29-Apr-2022;13:172
How to cite this URL: Nancy E. Epstein. Review/perspective on hysterical paralysis: A diagnosis of exclusion for spinal surgeons. 29-Apr-2022;13:172. Available from: https://surgicalneurologyint.com/surgicalint-articles/11572/
Abstract
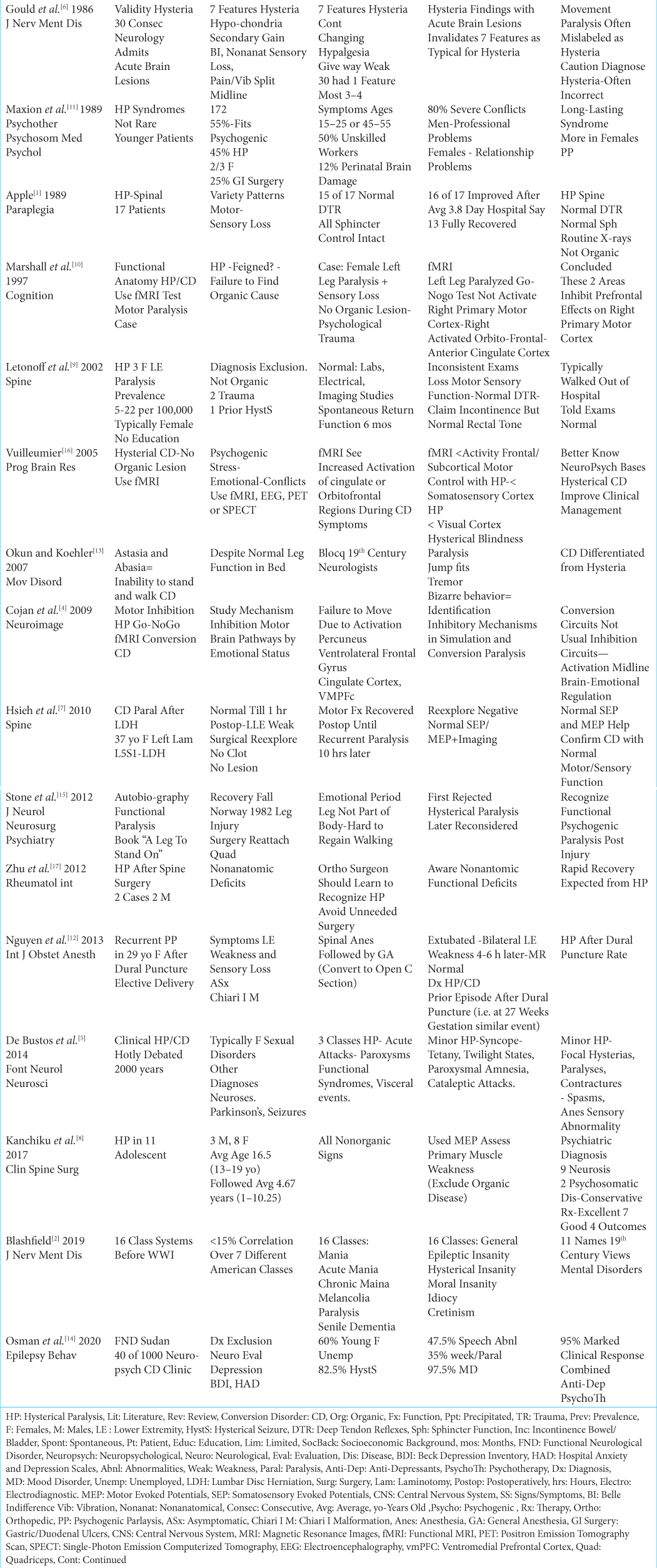
Background: Hysterical paralysis (HP) and/or conversion disorders (CD) are diagnoses of exclusion for spine surgeons. Before assigning this diagnosis to a patient, they must first undergo a full neurodiagnostic evaluation (i.e., X-rays, MR, CT/Myelo-CT) to rule out organic spinal pathology. Here, we reviewed select articles highlighting how to differentiate HP/CD patients from those with spinal disease.
Methods: Several case studies and small series of patients with HP/CD were included in our analysis. Notably, prior to being assigned the diagnoses of HP/CD, patients had to first undergo X-ray, MR, CT, and/or Myelo-CT evaluations to rule out spinal disorders; typically, their neurodiagnostic studies were normal.
Results: Patients with HP/CD often presented with varying clinical complaints of motor paralysis despite intact reflexes, normal sensory examinations, and lack of sphincter disturbance (i.e. intact rectal tone). Further, go and nogo functional MRI (fMRI) examinations demonstrated inconsistencies in areas of brain activation for patients with HP/CD complaints.
Conclusions: HP/CD are diagnoses of exclusion, and patients should first undergo a full panel of neurodiagnostic studies to rule out organic spinal disease. While those with HP/CD should not have unnecessary operations, those with real “surgical pathology” should have appropriate spine surgery performed in a timely fashion.
Keywords: Computed Tomography (CT), Conversion Disorder (CD), Diagnosis of Exclusion, Functional MRI (fMRI), Hysterical Paralysis, Magentic Resonance Imaging (MR/MRI)
INTRODUCTION
Hysterical paralysis (HP) or conversion disorders (CF) are diagnoses of exclusion [
MULTIPLE FACTORS ASSOCIATED WITH HYSTERICAL PARALYSIS/ CONVERSION DISORDERS
Maxion et al. (1989) identified multiple factors in patients with “classical conversion syndromes” and/or psychogenic seizures/paralysis [
CLASSIFICATION SYSTEMS FOR PSYCHOLOGICAL/MENTAL DISORDERS
In 2019, Blashfield analyzed 16 different classification systems using 22 common terms to describe psychological/ mental disorders that had been used since the 19th century [
HISTORY OF HYSTERICAL PARALYSIS (HP)/CONVERSION DISORDERS (CD)
Several studies highlighted how inconsistencies in clinical evaluations and ultimately normal neurodiagnostic studies helped differentiate between HP/CD vs. organic spinal disorders [
DIFFERENTIATING HYSTERICAL PARALYSIS/ CONVERSION DISORDERS FROM ORGANIC SPINAL PATHOLOGY
The incidence of HP/CD is 5-22/100,000 in the overall population. Here we have summarized how the diagnoses of HP/CD can be differentiated from organic spinal pathology [
CONVERSION PARALYSIS AFTER SPINE SURGERY
Few papers focused on episodes of HP/CD “paralysis” immediately following spine surgery [
CONVERSION PARALYSIS AFTER LEG INJURY
In 2012, Stone reported his own experience with transient psychogenic paralysis of a leg following a fall resulting in a torn quadriceps muscle [
FUNCTIONAL MRI OF PARALYSIS
fMRI, performed in patients with unexplained/inconsistent neurological deficits and normal neurodiagnostic studies, helped identify/differentiate patients with HP/CD vs. organic spinal disease [
MEDICOLEGAL IMPACT OF MISDIAGNOSING HYSTERICAL PARALYSIS
The diagnoses of HP/CD are diagnoses of exclusion, and should not be established without first obtaining appropriate neurodiagnostic tests and other studies [
CONCLUSION
Patients should not be labeled with HP/CD paralysis until neurodiagnostic/other studies (i.e., variously including MR, CT-Myelo-CT, fMRI, SEP/EMG/ MEP) have ruled out the presence of organic disease.
Declaration of patient consent
Patient’s consent not required as there are no patients in this study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Apple DF. Hysterical spinal paralysis. Paraplegia. 1989. 27: 428-31
2. Blashfield RK. Pre-kraepelin names for mental disorders. J Nerv Ment Dis. 2019. 207: 726-30
3. Boudissa M, Castelain JE, Boissiere L, Mariey R, Pointillart V, Vital JM. Conversion paralysis after cervical spine arthroplasty: A case report and literature review. Orthop Traumatol Surg Res. 2015. 101: 637-41
4. Cojan Y Waber L, Carruzzo A, Vuilleumier P. Motor inhibition in hysterical conversion paralysis. Neuroimage. 2009. 47: 1026-37
5. De Bustos EM, Galli S, Haffen E, Moulin T. Clinical manifestations of hysteria: An epistemological perspective or how historical dynamics illuminate current practice. Front Neurol Neurosci. 2014. 35: 28-42
6. Gould R, Miller BL, Goldberg MA, Benson DF. The validity of hysterical signs and symptoms. J Nerv Ment Dis. 1986. 174: 593-7
7. Hsieh MK, Chang CN, Hsiao MC, Chen WJ, Chen LH. Conversion paralysis after surgery for lumbar disc herniation. Spine. 2010. 35: E308-10
8. Kanchiku T, Suzuki H, Imajo Y, Yoshida Y, Nishida N, Taguchi T. psychogenic low-back pain and hysterical paralysis in adolescence. Clin Spine Surg. 2017. 30: E1122-5
9. Letonoff EJ, Williams TR, Sidhu KS. Hysterical paralysis: a report of three cases and a review of the literature. Spine. 2002. 27: E441-5
10. Marshall JC, Halligan Fink GR, Wade DT, Frackowiak RS. The functional anatomy of a hysterical paralysis. Cognition. 1997. 64: B1-8
11. Maxion H, Fegers S, Pfluger R, Wiegand J. Risk factors of classical conversion syndrome--psychogenic seizures and paralyses--observations at a neurologic clinic with 172 patients]. Psychother Psychosom Med Psychol. 1989. 39: 121-6
12. Nguyen J, Abola R, Schabel J. Recurrent psychogenic paresis after dural puncture in a parturient. Int J Obstet Anesth. 2013. 22: 160-3
13. Okun MS, Koehler PJ. Paul Blocq and (psychogenic) astasia abasia. Mov Disord. 2007. 22: 1373-8
14. Osman AH, Alsharief SM, Siddig HE. Functional neurological disorder: Characteristics and outcome in a limited-resources country (Sudan). Epilepsy Behav. 2020. 111: 107151
15. Stone J, Perthen J, Carson AJ. “A leg to stand on” by Oliver Sacks: A unique autobiographical account of functional paralysis. J Neurol Neurosurg Psychiatry. 2012. 83: 864-7
16. Vuilleumier P. Hysterical conversion and brain function. Prog Brain Res. 2005. 150: 309-29
17. Zhu L, Ni B, Guo Q. Hysterical paralysis after spinal surgery. Rheumatol Int. 2012. 32: 4077-8