- Clinical Professor of Neurosurgery, School of Medicine, State University of NY at Stony Brook, NY and ℅ Dr. Marc Agulnick 1122 Frankllin Avenue Suite 106, Garden City, NY 11530, USA.
Correspondence Address:
Nancy E. Epstein, MD, Clinical Professor of Neurosurgery, School of Medicine, State University of NY at Stony Brook, NY and ℅ Dr. Marc Agulnick 1122 Frankllin Avenue Suite 106, Garden City, NY 11530, USA.
DOI:10.25259/SNI_170_2022
Copyright: © 2022 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Nancy E. Epstein. Review/Perspective: Operations for Cauda Equina Syndromes - “The Sooner the Better”. 25-Mar-2022;13:100
How to cite this URL: Nancy E. Epstein. Review/Perspective: Operations for Cauda Equina Syndromes - “The Sooner the Better”. 25-Mar-2022;13:100. Available from: https://surgicalneurologyint.com/surgicalint-articles/11490/
Abstract
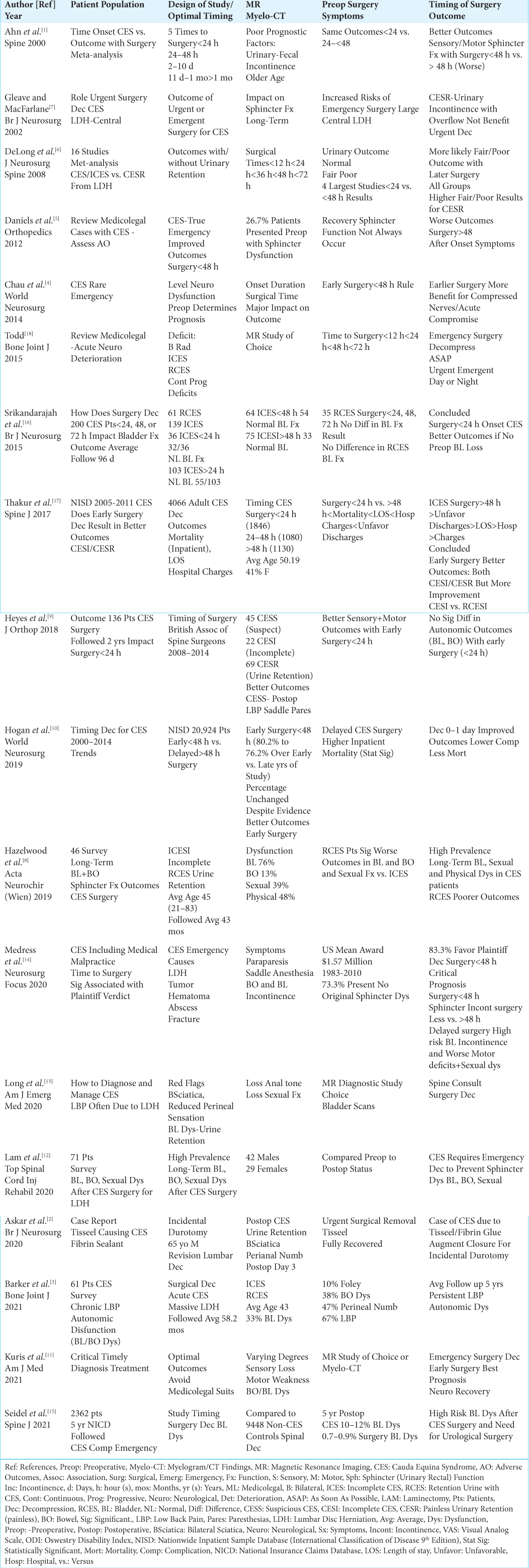
Background: Although most studies recommended that early surgery for cauda equina syndromes (CES) be performed within
Methods: The 2 major variants of CES include; incomplete/partial ICES, and those with urinary retention/bowel incontinence (RCES). Those with ICES often exhibit varying combinations of motor weakness, sensory loss (i.e. including perineal numbness), and urinary dysfunction, while RCES patients typically exhibit more severe paraparesis, sensory loss including saddle anesthesia, and urinary/bowel incontinence. The pathology responsible for ICES/RCES syndromes may include; acute disc herniations/stenosis, trauma (i.e. including iatrogenic/ surgical hematomas etc.), infections, abscesses, and other pathology. Surgery for either ICES/RCES may include decompressions to multilevel laminectomies/fusions.
Results: Following early surgery, most studies showed that ICES and RCES patients exhibited improvement in motor weakness and sensory loss. However, recovery of sphincter function was more variable, being poorer for RCES patients with preoperative urinary retention/bowel incontinence.
Conclusions: Although early CES surgery was defined in most studies as
Keywords: Cauda Equina Syndrome, Early Diagnosis, Optimal Timing, Surgery, “The Sooner the Better” Surgery
INTRODUCTION
Surgery performed “the sooner the better” for patients with cauda equina syndromes (CES) was best [
DIFFERENT COMBINATIONS OF PREOPERATIVE SYMPTOMS AND SIGNS OF ICES AND RCES
Multiple studies confirmed that the red flags for CES (i.e. including ICES and RCES syndromes) included; varying severities of sciatica, motor weakness, sensory loss with reduced perineal sensation, bladder/bowel dysfunction/urinary retention, and loss of sexual function [
SURGERY FOR ICES AND RCES SYNDROMES: “THE SOONER THE BETTER” IS BEST
Most smaller studies recommended early surgery for CES (ICES/RCES) within <48 h. [
LONG-TERM POSTOPERATIVE SPHINCTER DYSFUNCTION FOR BOTH ICES AND RCES PATIENTS
Several studies observed greater residual long-term bladder or bowel dysfunction after surgery in both ICES and RCES patients [
MEDICOLEGAL VIEW OF SURGICAL TIMING FOR CES
The medicolegal literature acknowledges that CES surgery performed “the sooner the better” is best [
CONCLUSION
Although many studies stated that CES surgery should be performed within <48 h. after symptom onset, two of the largest NISC series (i.e. involving 20, 924,[
Declaration of patient consent
Patient’s consent not required as there are no patients in this study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Ahn UM, Ahn NU, Buchowski JM, Garrett ES, Sieber AN, Kostuik JP. Cauda equina syndrome secondary to lumbar disc herniation: A meta-analysis of surgical outcomes. Spine. 2000. 25: 1515-22
2. Askar M, Gakhar H. Cauda equina syndrome after use of dural sealants in revision lumbar decompressive surgery. Br J Neurosurg. p. 1-3
3. Barker TP, Steele N, Swamy G, Cook A, Rai A, Crawford R. Long-term core outcomes in cauda equina syndrome. Bone Joint J. 2021. 103: 1464-71
4. Chau AM, Xu LL, Pelzer RN, Gragnaniello C. Timing of surgical intervention in cauda equina syndrome: A systematic critical review. World Neurosurg. 2014. 81: 640-50
5. Daniels EW, Gordon Z, French K, Ahn UM, Ahn NU. Review of medicolegal cases for cauda equina syndrome: What factors lead to an adverse outcome for the provider?. Orthopedics. 2012. 35: e414-9
6. DeLong WB, Polissar N, Neradilek B. Timing of surgery in cauda equina syndrome with urinary retention: Meta-analysis of observational studies. J Neurosurg Spine. 2008. 8: 305-20
7. Gleave JR, MacFarlane R. Cauda equina syndrome: What is the relationship between timing of surgery and outcome?. Br J Neurosurg. 2002. 16: 325-8
8. Hazelwood JE, Hoeritzauer I, Pronin S, Demetriades AK. An assessment of patient-reported long-term outcomes following surgery for cauda equina syndrome. Acta Neurochir (Wien). 2019. 161: 1887-94
9. Heyes G, Jones M, Verzin E, McLorinan G, Darwish N, Eames N. Influence of timing of surgery on Cauda equina syndrome: Outcomes at a national spinal centre. J Orthop. 2018. 15: 210-5
10. Hogan WB, Kuris EO, Durand WM, Eltorai AE, Daniels AH. Timing of surgical decompression for Cauda equina syndrome. World Neurosurg. 2019. 132: e732-8
11. Kuris EO, McDonald CL, Palumbo MA, Daniels AH. Evaluation and management of Cauda equina syndrome. Am J Med. 2021. 134: 1483-9
12. Lam J, DeSouza RM, Laycock J, Perera D, Burford C, Khan B. Patient-reported bladder, bowel, and sexual function after Cauda equina syndrome secondary to a herniated lumbar intervertebral disc. Top Spinal Cord Inj Rehabil. 2020. 26: 290-303
13. Long B, Koyfman A, Gottlieb M. Evaluation and management of cauda equina syndrome in the emergency department. Am J Emerg Med. 2020. 38: 143-8
14. Medress ZA, Jin MC, Feng A, Varshneya K, Veeravagu A. Medicolegal issues in neurosurgery. Neurosurg Focus. 2020. 49: E10
15. Seidel H, Bhattacharjee S, Pirkle S, Shi L, Strelzow J, Lee M. Long-term rates of bladder dysfunction after decompression in patients with cauda equina syndrome. Spine J. 2021. 21: 803-9
16. Srikandarajah N, Boissaud-Cooke MA, Clark S, Wilby MJ. Does early surgical decompression in cauda equina syndrome improve bladder outcome?. Br J Neurosurg. 2015. 40: 580-3
17. Thakur JD, Storey C, Kalakoti P, Ahmed O, Dossani RH, Menger RP. Early intervention in cauda equina syndrome associated with better outcomes: A myth or reality? Insights from the Nationwide Inpatient Sample database (2005-2011). Spine J. 2017. 17: 1435-48
18. Todd NV. Cauda equina syndrome: Is the current management of patients presenting to district general hospitals fit for purpose? A personal view based on a review of the literature and a medicolegal experience. Bone Joint J. 2015. 97: 1390-4