- Department of Neurosurgery, Post Graduate Institute of Medical Education and Research, Chandigarh, India
- Department of Neurosurgery, Neuro-nursing Division, National Institute of Nursing Education, Post Graduate Institute of Medical Education and Research, Chandigarh, India
Correspondence Address:
Sivashanmugam Dhandapani
Department of Neurosurgery, Post Graduate Institute of Medical Education and Research, Chandigarh, India
DOI:10.4103/2152-7806.168067
Copyright: © 2015 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Dhandapani S, Aggarwal A, Srinivasan A, Meena R, Gaudihalli S, Singh H, Dhandapani M, Mukherjee KK, Gupta SK. Serum lipid profile spectrum and delayed cerebral ischemia following subarachnoid hemorrhage: Is there a relation?. Surg Neurol Int 23-Oct-2015;6:
How to cite this URL: Dhandapani S, Aggarwal A, Srinivasan A, Meena R, Gaudihalli S, Singh H, Dhandapani M, Mukherjee KK, Gupta SK. Serum lipid profile spectrum and delayed cerebral ischemia following subarachnoid hemorrhage: Is there a relation?. Surg Neurol Int 23-Oct-2015;6:. Available from: http://surgicalneurologyint.com/surgicalint_articles/serum-lipid-profile-spectrum-and-delayed-cerebral-ischemia/
Abstract
Background:Serum lipid abnormalities are known to be important risk factors for vascular disorders. However, their role in delayed cerebral ischemia (DCI), the major cause of morbidity after subarachnoid hemorrhage (SAH) remains unclear. This study was an attempt to evaluate the spectrum of lipid profile changes in SAH compared to matched controls, and their relation with the occurrence of DCI.
Methods:Admission serum lipid profile levels were measured in patients of SAH and prospectively studied in relation to various factors and clinical development of DCI.
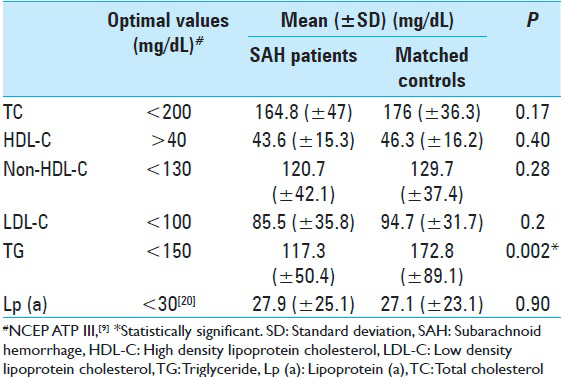
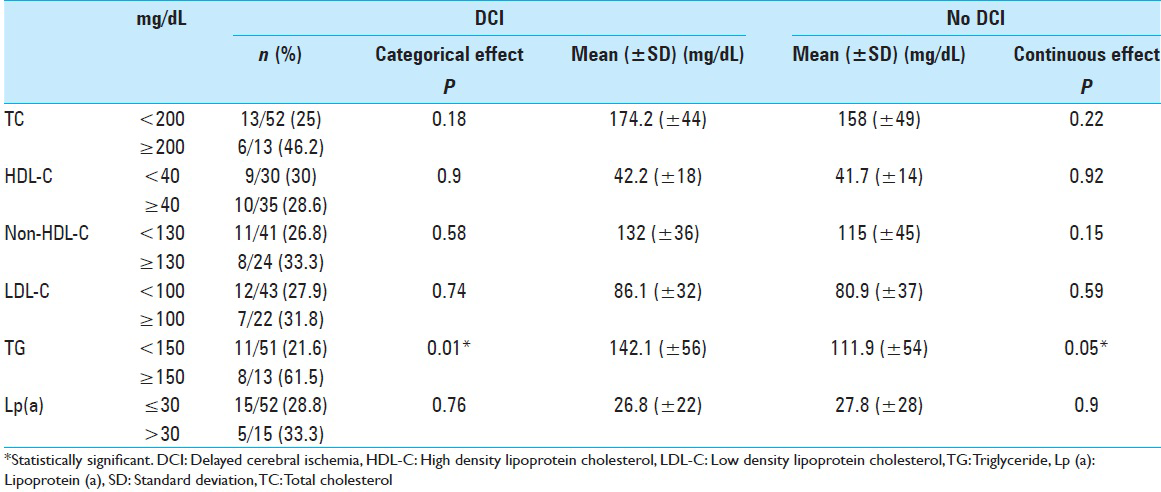
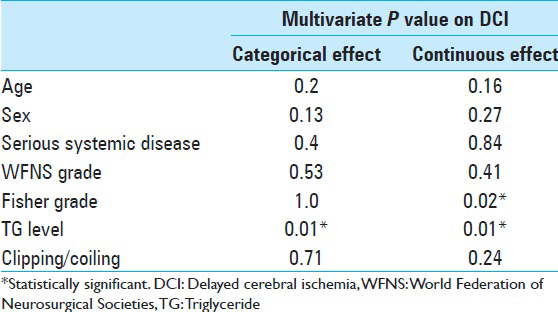
Results:Serum triglyceride (TG) levels were significantly lower among SAH patients compared to matched controls (mean [±standard deviation (SD)] mg/dL: 117.3 [±50.4] vs. 172.8 [±89.1], P = 0.002), probably because of energy consumption due to hypermetabolic response. Patients who developed DCI had significantly higher TG levels compared to those who did not develop DCI (mean [±SD] mg/dL: 142.1 [±56] vs. 111.9 [±54], P = 0.05). DCI was noted in 62% of patients with TG >150 mg/dL, compared to 22% among the rest (P = 0.01). Total cholesterol, low-density lipoprotein cholesterol, high-density lipoprotein cholesterol and lipoprotein (a) neither showed a significant difference between SAH and controls and nor any significant association with DCI. Multivariate analysis using binary logistic regression adjusting for the effects of age, sex, systemic disease, World Federation of Neurosurgical Societies grade, Fisher grade, and clipping/coiling, revealed higher TG levels to have significant independent association with DCI (P = 0.01).
Conclusions:Higher serum TG levels appear to be significantly associated with DCI while other lipid parameters did not show any significant association. This may be due to their association with remnant cholesterol or free fatty acid-induced lipid peroxidation.
Keywords: Cholesterol, delayed cerebral ischemia, lipid, remnant cholesterol, subarachnoid hemorrhage, triglyceride
INTRODUCTION
Subarachnoid hemorrhage (SAH) remains a serious disease with high morbidity and mortality, despite improvements in diagnostic modalities, better Intensive Care Unit (ICU) facilities, microsurgical, and endovascular advancements.[
Serum lipid abnormalities have been noted as important risk factors in a variety of vascular disorders such as coronary artery disease, ischemic stroke, and peripheral arterial disease.[
The present study was to evaluate the spectrum of lipid profile changes in SAH compared to matched controls, and their relation with the occurrence of DCI.
METHODS
All adult patients with spontaneous SAH reporting within 48 h of ictus to the neurosurgical emergency of the Post Graduate Institute of Medical Education and Research, Chandigarh, India were included in the study. Patients with known history of hyperlipidemia, renal or liver dysfunction were excluded.
The serum samples for lipid measurements were taken at the time of admission. TG, TC, and HDL-C were analyzed by enzymatic colorimetric assay using Hitachi 704 Analyzer (Roche Diagnostics, Indianapolis, USA). LDL-C was calculated from measured values of TG, TC, and HDL-C in mg/dL according to the Friedewald equation: “LDL-C = TC − HDL-C − TG/5”. Non-HDL-C was calculated by subtracting HDL-C from TC.[
SAH patients were assessed since admission using World Federation of Neurosurgical Societies (WFNS) grading.[
Outcome
DCI or symptomatic vasospasm was implicated if there was occurrence of focal neurological impairment (such as hemiparesis, aphasia, apraxia, hemianopia, or neglect), or a decrease of at least two points on the Glasgow Coma Scale (either on the total score or on one of its individual components [eye, motor on either side, verbal]),[
Statistical analysis
SPSS 21 software (IBM Corp., New York, USA) was used for the statistical analyses. Univariate analyses of continuous variables across binary categories were compared using the independent samples t-test. The bivariate relationship between two continuous variables was assessed using the Pearson correlation coefficient. Proportions were compared using Chi-square or Fisher's exact test wherever appropriate, and subgroup analyses were done using the Breslow–Day test of homogeneity of odds ratios. Two-sided significance tests were used throughout, and the significance level was kept at P < 0.05. Multivariate analyses were conducted using binary logistic regression with mandatory significance of the model coefficient being <0.05 for validity of outcome prediction after adjusting for known prognostic factors such as age, sex, serious systemic disease, WFNS grade, Fisher grade, and definitive treatment in relation to lipid parameters found significant in univariate analysis.
RESULTS
There was a total of 86 patients enrolled initially in our study. Out of these, samples from only 77, 73, and 75 patients could be properly analyzed for Lp(a), TG, and other cholesterol levels, respectively, due to technical issues of blood samples. Of the total 86 patients, 75 who were available under treatment at 7 days following ictus were included in the outcome analysis. Their ages were normally distributed ranging from 20 to 76 years. The mean age was 49 years, and there were 17 patients aged 60 years or more. There were 39 males and 36 females. Among the 75 patients analyzed, 67, 64, and 65 patients had Lp(a), TG, and other cholesterol levels, respectively.
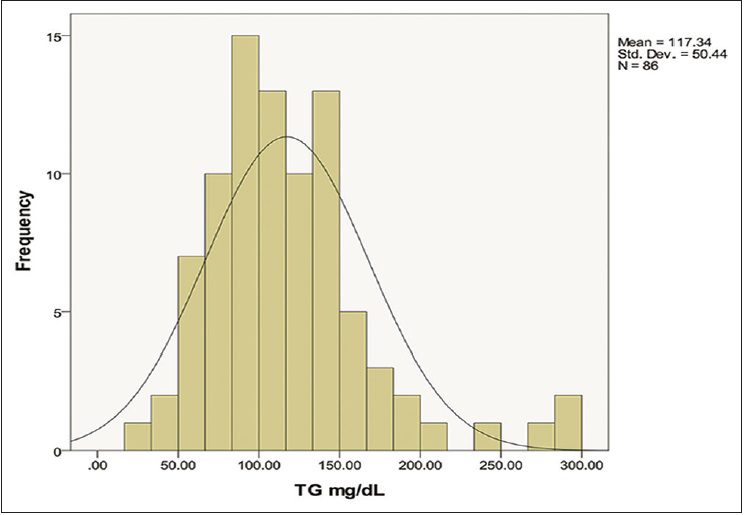
Serum TG levels were found to be lower among SAH patients when compared to matched controls (mean [±standard deviation (SD)] mg/dL: 117.3 [±50.4] vs. 172.8 [±89.1]) and the same was statistically significant (P = 0.002). These levels were normally distributed as shown in
DCI developed in 22 out of 75 patients. Patients who developed DCI had significantly higher TG levels compared to those who did not develop DCI (mean [±SD] mg/dL: 142.1 [±56] vs. 111.9 [±54], P = 0.05). DCI was noted in 8 out of 13 (62%) patients with TG >150 mg/dL, compared to 11 out of 51 (22%) among the rest (P = 0.01) [
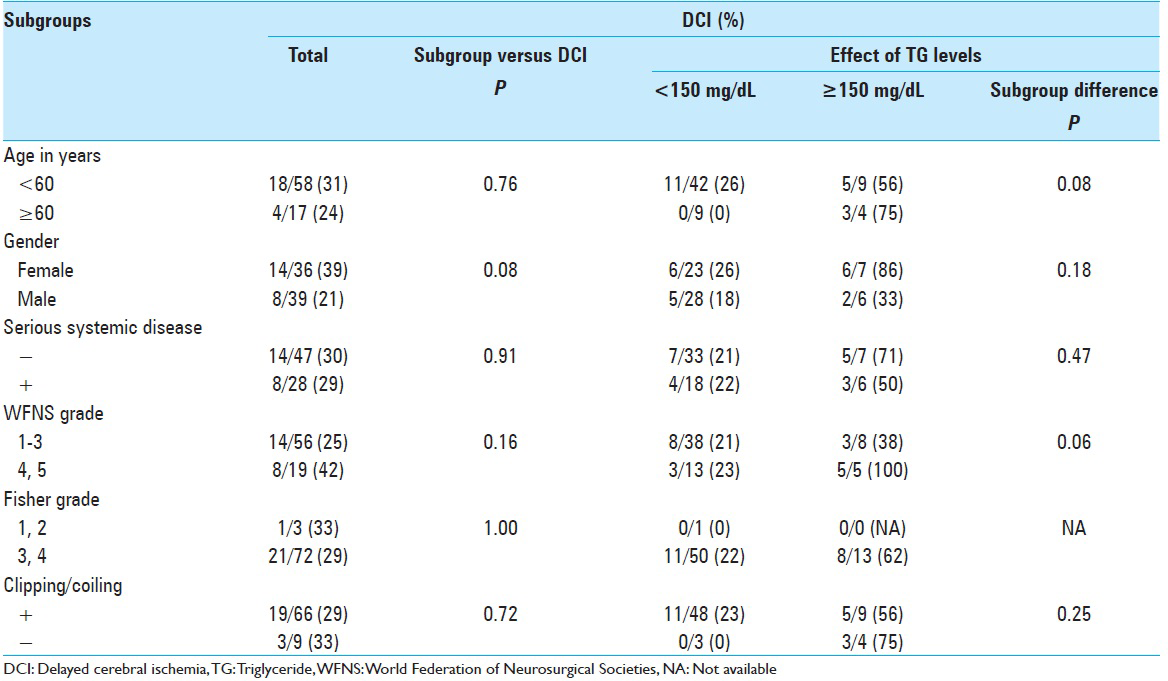
The difference in the occurrence of DCI in relation to TG levels in various subgroups is shown in
The multivariate analysis using binary logistic regression adjusting for the effects of various factors on DCI is shown in
DISCUSSION
The variety of metabolic responses following the stress of SAH similar to traumatic brain injury is often strained further by pathophysiological challenges of compromised perfusion due to DCI.[
Though hyperlipidemia has been significantly implicated in several ischemic disorders, their association with SAH has been controversial.[
Though the impact of serum lipids on DCI may appear intuitive, and studies of cholesterol reducing agents (statins) in SAH are increasing,[
Among the lipid profile components, LDL, the main carrier of cholesterol to tissues became the prime focus for vascular disease prevention, which on entry into intima, undergoes oxidation and taken up by macrophages to kick start atherothrombosis.[
Serum TG levels heretofore ignored has recently generated much evidence to be suggestive of remnant cholesterol in TG rich lipoproteins and more significantly associated with vascular diseases than others.[
The impact of serum TG levels indicating remnant cholesterol on the occurrence of DCI after SAH, as noted in this study has never been reported. The sample size of this study may appear small, but the potential for therapeutic modulation make it relevant. We need larger studies with complete categorization of all lipid parameters in relation to better outcome measures, so that we can fully validate the role of remnant cholesterol of TG rich lipoproteins in patients of SAH.
CONCLUSION
Higher serum TG levels at admission appear to be significantly associated with DCI following SAH, probably due to their association with remnant cholesterol or free fatty acid-induced lipid peroxidation, while other lipid parameters did not show significant association with DCI.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Amarenco P, Labreuche J. Lipid management in the prevention of stroke: Review and updated meta-analysis of statins for stroke prevention. Lancet Neurol. 2009. 8: 453-63
2. Badjatia N, Seres D, Carpenter A, Schmidt JM, Lee K, Mayer SA. Free fatty acids and delayed cerebral ischemia after subarachnoid hemorrhage. Stroke. 2012. 43: 691-6
3. Dhandapani S, Goudihalli S, Mukherjee KK, Singh H, Srinivasan A, Danish M. Prospective study of the correlation between admission plasma homocysteine levels and neurological outcome following subarachnoid hemorrhage: A case for the reverse epidemiology paradox?. Acta Neurochir (Wien). 2015. 157: 399-407
4. Dhandapani S, Kapoor A, Gaudihalli S, Dhandapani M, Mukherjee KK, Gupta SK. Study of trends in anthropometric nutritional indices and the impact of adiposity among patients of subarachnoid hemorrhage. Neurol India. 2015. 63: 531-6
5. Dhandapani S, Pal SS, Gupta SK, Mohindra S, Chhabra R, Malhotra SK. Does the impact of elective temporary clipping on intraoperative rupture really influence neurological outcome after surgery for ruptured anterior circulation aneurysms? – A prospective multivariate study. Acta Neurochir (Wien). 2013. 155: 237-46
6. Dhandapani S, Sharma A, Sharma K, Das L. Comparative evaluation of MRS and SPECT in prognostication of patients with mild to moderate head injury. J Clin Neurosci. 2014. 21: 745-50
7. Dhandapani SS, Manju D, Mahapatra AK. The economic divide in outcome following severe head injury. Asian J Neurosurg. 2012. 7: 17-20
8. Dhandapani SS, Manju D, Vivekanandhan S, Agarwal M, Mahapatra AK. Prospective longitudinal study of biochemical changes in critically ill patients with severe traumatic brain injury: Factors associated and outcome at 6 months. Indian J Neurotrauma. 2010. 7: 23-7
9. . Expert Panel on Detection, Evaluation and Treatment of High Blood Cholesterol in Adults. Executive summary of the third report of the national cholesterol education program (NCEP) expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel III). JAMA. 2001. 285: 2486-97
10. Fisher CM, Kistler JP, Davis JM. Relation of cerebral vasospasm to subarachnoid hemorrhage visualized by computerized tomographic scanning. Neurosurgery. 1980. 6: 1-9
11. Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972. 18: 499-502
12. Lin Y, Mousa SS, Elshourbagy N, Mousa SA. Current status and future directions in lipid management: Emphasizing low-density lipoproteins, high-density lipoproteins, and triglycerides as targets for therapy. Vasc Health Risk Manag. 2010. 6: 73-85
13. Liu Z, Liu L, Zhang Z, Chen Z, Zhao B. Cholesterol-reducing agents for aneurysmal subarachnoid haemorrhage. Cochrane Database Syst Rev. 2013. 4: CD008184-
14. Lynch JR, Wang H, McGirt MJ, Floyd J, Friedman AH, Coon AL. Simvastatin reduces vasospasm after aneurysmal subarachnoid hemorrhage: Results of a pilot randomized clinical trial. Stroke. 2005. 36: 2024-6
15. Maranhão RC, Carvalho PO, Strunz CC, Pileggi F. Lipoprotein (a): Structure, pathophysiology and clinical implications. Arq Bras Cardiol. 2014. 103: 76-84
16. Mora S, Otvos JD, Rifai N, Rosenson RS, Buring JE, Ridker PM. Lipoprotein particle profiles by nuclear magnetic resonance compared with standard lipids and apolipoproteins in predicting incident cardiovascular disease in women. Circulation. 2009. 119: 931-9
17. Nonaka N, Matsukado Y, Hirate Y, Mihara Y, Mimata C, Miura G. Lipid metabolism of the patients with subarachnoid hemorrhage due to ruptured intracranial aneurysm – With special reference to the occurrence of cerebral angiospasm. No To Shinkei. 1989. 41: 67-72
18. Nordestgaard BG, Varbo A. Triglycerides and cardiovascular disease. Lancet. 2014. 384: 626-35
19. Raaymakers TW. Aneurysms in relatives of patients with subarachnoid hemorrhage: Frequency and risk factors. MARS Study Group. Magnetic resonance angiography in relatives of patients with subarachnoid hemorrhage. Neurology. 1999. 53: 982-8
20. Rader DJ, Hovingh GK. HDL and cardiovascular disease. Lancet. 2014. 384: 618-25
21. Ridker PM. LDL cholesterol: Controversies and future therapeutic directions. Lancet. 2014. 384: 607-17
22. Sandvei MS, Lindekleiv H, Romundstad PR, Müller TB, Vatten LJ, Ingebrigtsen T. Risk factors for aneurysmal subarachnoid hemorrhage – BMI and serum lipids: 11-year follow-up of the HUNT and the Tromsø Study in Norway. Acta Neurol Scand. 2012. 125: 382-8
23. Sasani M, Yazgan B, Celebi I, Aytan N, Catalgol B, Oktenoglu T. Hypercholesterolemia increases vasospasm resulting from basilar artery subarachnoid hemorrhage in rabbits which is attenuated by Vitamin E. Surg Neurol Int. 2011. 2: 29-
24. Schwartz EA, Reaven PD. Lipolysis of triglyceride-rich lipoproteins, vascular inflammation, and atherosclerosis. Biochim Biophys Acta. 2012. 1821: 858-66
25. Stein DT, Devaraj S, Balis D, Adams-Huet B, Jialal I. Effect of statin therapy on remnant lipoprotein cholesterol levels in patients with combined hyperlipidemia. Arterioscler Thromb Vasc Biol. 2001. 21: 2026-31
26. Teasdale GM, Drake CG, Hunt W, Kassell N, Sano K, Pertuiset B. A universal subarachnoid hemorrhage scale: Report of a committee of the World Federation of Neurosurgical Societies. J Neurol Neurosurg Psychiatry. 1988. 51: 1457-
27. Terao S, Yilmaz G, Stokes KY, Ishikawa M, Kawase T, Granger DN. Inflammatory and injury responses to ischemic stroke in obese mice. Stroke. 2008. 39: 943-50
28. Tewari M, Aggarwal A, Mathuriya S, Gupta V. The outcome after aneurysmal sub arachnoid hemorrhage: A study of various factors. Ann Neurosci. 2015. 22: 78-80
29. Tokuda Y, Stein GH. Serum lipids as protective factors for subarachnoid hemorrhage. J Clin Neurosci. 2005. 12: 538-41
30. Tseng MY, Hutchinson PJ, Turner CL, Czosnyka M, Richards H, Pickard JD. Biological effects of acute pravastatin treatment in patients after aneurysmal subarachnoid hemorrhage: A double-blind, placebo-controlled trial. J Neurosurg. 2007. 107: 1092-100
31. Varbo A, Benn M, Tybjærg-Hansen A, Nordestgaard BG. Elevated remnant cholesterol causes both low-grade inflammation and ischemic heart disease, whereas elevated low-density lipoprotein cholesterol causes ischemic heart disease without inflammation. Circulation. 2013. 128: 1298-309
32. Varbo A, Freiberg JJ, Nordestgaard BG. Extreme nonfasting remnant cholesterol vs extreme LDL cholesterol as contributors to cardiovascular disease and all-cause mortality in 90000 individuals from the general population. Clin Chem. 2015. 61: 533-43
33. Vergouwen MD, Vermeulen M, van Gijn J, Rinkel GJ, Wijdicks EF, Muizelaar JP. Definition of delayed cerebral ischemia after aneurysmal subarachnoid hemorrhage as an outcome event in clinical trials and observational studies: Proposal of a multidisciplinary research group. Stroke. 2010. 41: 2391-5