- Department of Neurosurgery, Loma Linda University, Loma Linda, California, United States.
- Department of Otolaryngology, Loma Linda University, Loma Linda, California, United States.
Correspondence Address:
Brian Hanak, Department of Neurosurgery, Loma Linda University, Loma Linda, California, United States.
DOI:10.25259/SNI_95_2023
Copyright: © 2023 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Emmanuel Omosor1, Lindsay Hunt1, Brian Hanak1, Vikrum Ashok Thimmappa2. Single dose intralesional doxycycline foam infusion promotes bone growth and resolution of an odontoid aneurysmal bone cyst. 26-May-2023;14:179
How to cite this URL: Emmanuel Omosor1, Lindsay Hunt1, Brian Hanak1, Vikrum Ashok Thimmappa2. Single dose intralesional doxycycline foam infusion promotes bone growth and resolution of an odontoid aneurysmal bone cyst. 26-May-2023;14:179. Available from: https://surgicalneurologyint.com/surgicalint-articles/12340/
Abstract
Background: Aneurysmal bone cysts (ABCs) are locally invasive bone tumors that most commonly arise in long bone metaphyses, the vertebral column, and pelvis, often presenting in the second decade of life. ABCs can be treated with resection, radiation, arterial embolization, and intralesional curettage. More recently intralesional doxycycline foam injections, which appear to act through the inhibition of matrix metalloproteinases and angiogenesis, have been used successfully, although multiple treatments are often required with this approach.
Case Description: A 13-year-old male with an incidentally discovered ABC filling much of the odontoid process but not violating the native odontoid cortex was treated with a single intralesional doxycycline foam injection delivered through a transoral approach with an excellent radiographic result. After placing a Crowe-Davis retractor, a transoral exposure of the odontoid process was performed with neuronavigation guidance. A fluoroscopy-guided Jamshidi needle biopsy was performed and via the needle doxycycline foam (2 mL 50 mg/mL doxycycline, 2 mL 25% albumin, and 1 mL Isovue 370 mixed with 5 mL of air) was infused, filling the cystic cavities of the odontoid process. The patient tolerated the operation well. Two months postoperatively, a computed tomography (CT) scan demonstrated a decrease in the size of the lesion with substantial new bone formation. Repeat CT at 6 months showed no residual cystic lucency, formation of dense new bone, and only minimal irregularity of the cortex at the prior needle biopsy site.
Conclusion: This case illustrates that the use of doxycycline foam can be an excellent option when managing ABCs that cannot be resected without incurring significant morbidity.
Keywords: Aneurysmal bone cyst, Doxycycline, Minimally invasive neurosurgery (MIN)
INTRODUCTION
Aneurysmal bone cysts (ABCs) are benign but locally invasive tumors that have been observed in most bones but most often arise in the long bone metaphyses, spinal column, and pelvis during the first two decades of life.[
The current treatment modalities include combinations of en bloc resection, intralesional curettage with or without bone grafting, endovascular arterial embolization, and radiation.[
CASE REPORT/CASE PRESENTATION
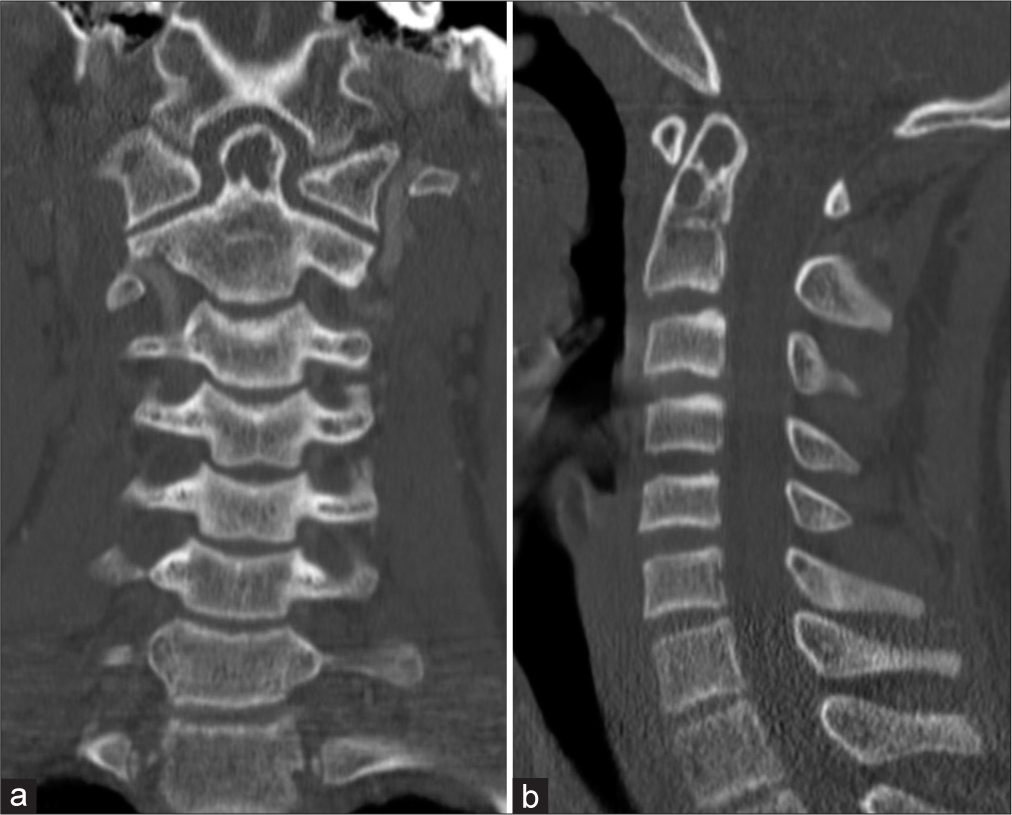
A 13-year-old male with a history of prematurity, meningococcal meningitis with post-meningitic neuropathic pain, tics, and staring spells concerning for seizures presented after being struck by a car while riding his bicycle. Polytrauma imaging workup revealed an incidentally discovered odontoid process cystic lesion [
The patient was allowed to make full recovery from his traumatic injuries prior to embarking on the management of the ABC. The indication for surgical intervention was primarily the location of the lesion. Any progression of the lesion could potentially affect cervical stability given the role of the odontoid process in neck range of motion biomechanics. Invasive intervention was decided upon to limit the chance that this ABC will expand and erode through the outer cortex of the odontoid process. The alternative option proposed would be conservative management with serial imaging or non-invasive therapies such as radiation.
A preoperative angiogram was performed 10 weeks after first discharge from the hospital for two reasons. The first reason was to evaluate the vasculature of the ABC and potentially embolize the main arterial feeders. The second reason was to confirm resolution of the previously noted traumatic right transverse/sigmoid sinus thrombosis. The diagnostic query failed to show any identifiable target for ABC embolization. However, there was confirmation of good flow with no residual clot in the right transverse/sigmoid sinus.
The surgery was performed 2 days after the diagnostic angiogram. There was the use of intraoperative neuronavigation (Stealth S8 AxiEM system) and intraoperative fluoroscopy. The patient was supine on a standard operating room table and his head was aligned in a neutral position on a horseshoe head holder. The operative exposure was performed by our otolaryngology colleagues. A Crowe-Davis mouth gag was placed. Two red rubber catheters were placed through the mouth and nose to elevate the palate. The posterior oropharynx was exposed, C1 arch was palpable and confirmed with navigation. A midline vertical incision was made in the posterior oropharynx. Dissection was then taken through the mucosa, muscles, and vertically through the anterior longitudinal ligament to expose the arch of C1 and then carried slightly inferiorly to expose the base of the ventral odontoid and cephalad aspect of the ventral C2 body.
With the anterior aspect of C2 exposed, a Jamshidi bone biopsy needle was used to enter the cortical bone at the anterior aspect of C2, near the base of the odontoid process under direct fluoroscopic guidance. A core sample of tissue from within the odontoid lesion was removed for permanent pathology. When the Jamshidi needle was withdrawn from C2, egress of dark (chronic) blood products was noted from the hole left in C2 by the needle pass.
Next, a doxycycline-albumin foam was created using the protocol first described by Shiels and Mayerson,[
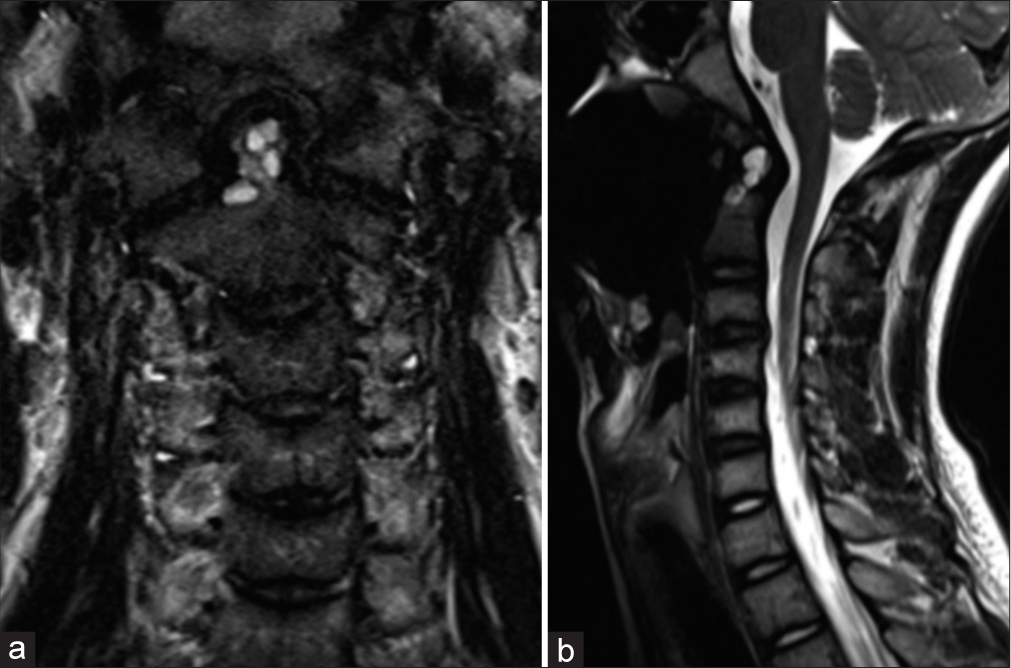
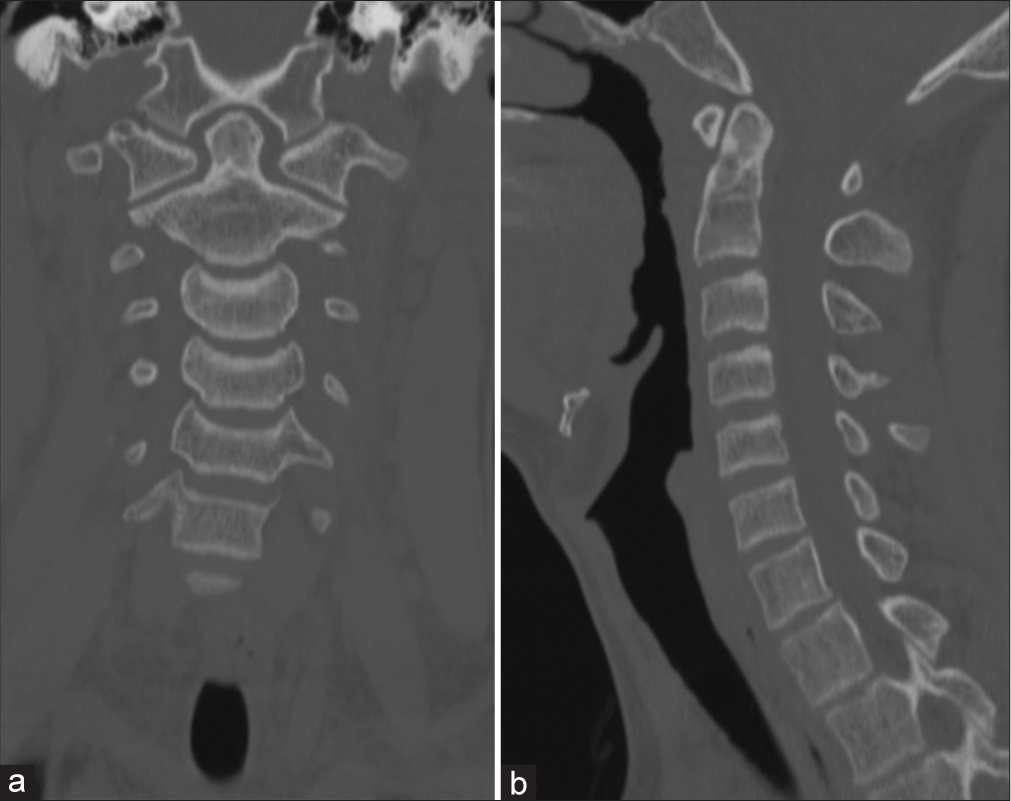
The otolaryngology team commenced with a multilayer closure after adequate hemostasis was obtained. The patient tolerated the procedure well. There were no postoperative complications, and he was discharged the next day. 2-month postoperatively, a CT scan demonstrated a decrease in the size of the lesion with substantial new bone formation [
DISCUSSION
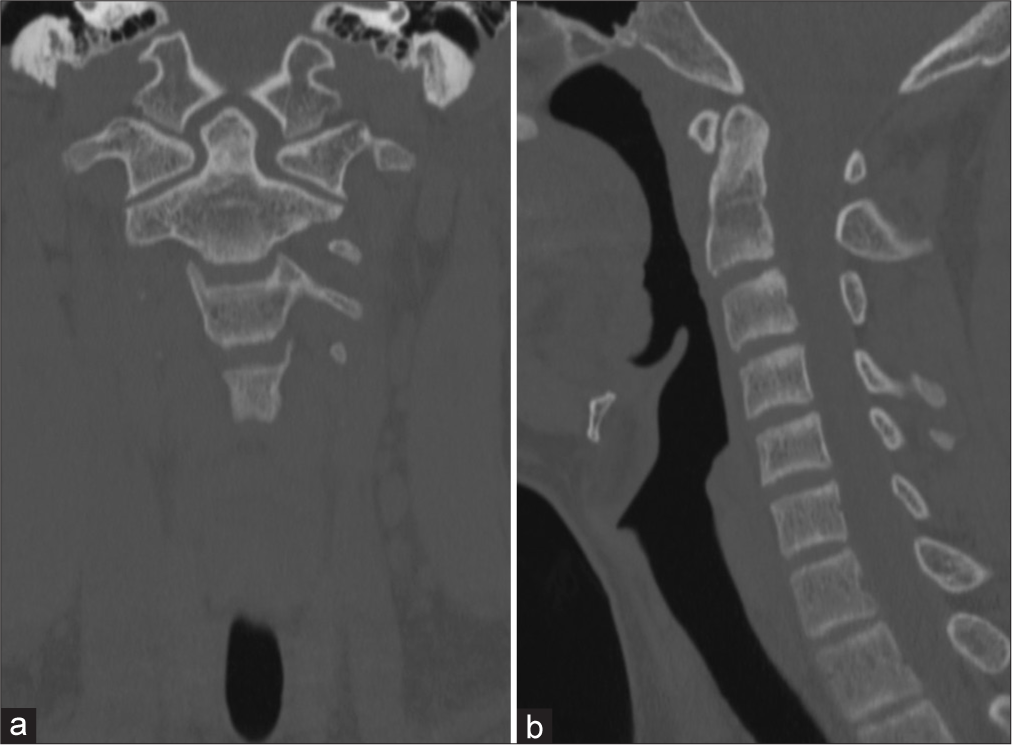
This case demonstrates a unique case of a single dose of doxycycline foam resulting in complete radiographic resolution of an ABC cystic cavity by 6 months following the initial procedure.
This patient’s case was ideal for a minimally invasive treatment option given the location of the cystic cavity. Gross total resection of the odontoid would have unequivocally resulted in spinal instability and necessitated occipitocervical fusion, thus increasing morbidity and permanently reducing this young patient’s neck range of motion. Also, at initial diagnosis and subsequent treatment, the lesion had not yet expanded to the point of violating the cortex of the odontoid. This allowed for the lesion to be completely filled with doxycycline foam within the confines of native cortical bone, which may explain why our patient’s ABC responded so well to a single doxycycline treatment. A large exophytic component extending well away from the confines of the bone of origin may have precluded the excellent result we obtained.
ABCs often occur in long bones such as the femur, tibia, and humerus followed by the spine, pelvis, and small bones of the hand and feet.[
Recurrence of ABCs is common after treatment, and some patient-related factors may contribute to the risk of recurrence. In a retrospective cohort of 53 patients with a median age of 14 years with primary ABCs, the authors found proximity to an open physis and age <12 years associated with a higher risk of recurrence following curettage of the lesion over a 24-month follow-up.[
Reported recurrence rates of ABC following various treatment modalities are diverse. In a single-center, retrospective review of ABC management, recurrence rates were found to be 38.2% for intralesional curettage and 25% for en bloc resection. 90.6% of ABCs treated with percutaneous instillation of sclerotherapy (polidocanol) had no recurrence but demonstrated persistent disease.[
The literature on the use of doxycycline foam in the treatment of ABCs is far more limited and, although existing reports suggest promising efficacy, outstanding questions remain about the appropriate timeframe to obtain follow up imaging and perform repeat doxycycline treatments in the setting of residual disease. The first report of doxycycline foam (10 mg/ mL of doxycycline mixed with 25% albumin and air) for the treatment of ABCs was published in 2013.[
In another retrospective and cohort study, it is suggested that a single injection of doxycycline foam followed by a waiting period of 12 weeks may be an appropriate timeline to assess for radiographic changes.[
Certainly, the case presented in this manuscript provides evidence that ongoing bone healing is possible between months 2 and 6 after doxycycline foam without repeat injections. Similar ongoing “late” healing after a single doxycycline treatment was also reported by Doyle et al who used doxycycline foam to successfully treat a recurrent ABC with a single treatment. They reported the case of a 12-year-old female who presented with neck pain found to have a large second and third cervical vertebral ABC for which she underwent resection and iliac crest bone grafting from C2 to C4. Six-month follow-up demonstrated cystic recurrence proximal to the iliac graft site and she underwent posterior-fusion occiput to C5 with iliac bone graft removal. Five months following the second operation, she was found to have a second recurrence, and at this time underwent intralesional doxycycline foam injection. A follow-up CT scan at 6 weeks demonstrated early bone formation and at 7 months, there was an extensive filling of the cavities with new bone.[
Small case series have thus far suggested that recurrence rates following doxycycline foam injection are comparable to surgical resection. A case series of seven children with ABCs located in either the spine, pelvis, or femur, found six of the seven patients to have clinical and imaging signs of improvement after a single doxycycline-albumin foam injection.[
One study has retrospectively compared doxycycline foam to open surgical resection. Liu et al. compared 14 patients treated with multiple injections of doxycycline foam with 11 patients treated with open surgery for spinal ABCs. The doxycycline foam injection group had significantly decreased lesion size after the injections for the 24-month follow-up period, improvement in neurologic symptoms when present, and improvement in pain ratings on the visual analog scale (VAS).[
A recent systematic meta-analysis on the management of juvenile bone cysts and ABCs reached the conclusion that surgical intervention was the most effective treatment; although, there were variations in success rates depending on the specific surgical approach employed. However, given that doxycycline injection and surgical excision both had high rates of success with 98% healing in both groups and 11% and 6% recurrence rates, respectively, the authors acknowledged that doxycycline injection remains a reasonable alternative. Importantly, the primary reason that the authors did not favor doxycycline injection as a first line treatment in cases without neurovascular compromise or structure instability was related to the frequent need for repeat doxycycline treatments to achieve complete bone healing in the published literature to date.[
Thus, given an excellent safety profile from existing literature, suggestion of efficacy that is at least on par with open surgical approaches, and the reduced morbidity and hospital length of stay that can be realized by avoiding upfront open resective surgical procedures the authors assert that doxycycline foam injections should be strongly considered in the management of newly diagnosed ABCs that are not associated with frank biomechanical instability or compression of neurovascular elements, particularly when it is feasible to deliver doxycycline foam treatment via a minimally invasive image guided percutaneous approach and in instances when a resective procedure is likely to be associated with morbidity.
CONCLUSION
We describe a case of odontoid ABC treatment with single intralesional doxycycline foam injection with excellent recovery and new bone formation. For patients where ABC resection is not feasible or would be associated with morbidity, doxycycline foam injections may be an effective and less morbid treatment option, particularly when it is feasible to deliver doxycycline foam treatment through a minimally invasive image guided percutaneous approach. While randomized controlled trials comparing doxycycline foam injections to open surgery are lacking, this case report adds to the growing body of literature around the excellent safety profile of this approach and highlights the fact that bone remodeling/healing can continue for months after a single doxycycline treatment.
Declaration of patient consent
Patient’s consent not required as patient’s identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Disclaimer
The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Journal or its management. The information contained in this article should not be considered to be medical advice; patients should consult their own physicians for advice as to their specific medical needs.
References
1. Adamsbaum C, Mascard E, Guinebretière JM, Kalifa G, Dubousset J. Intralesional Ethibloc injections in primary aneurysmal bone cysts: An efficient and safe treatment. Skeletal Radiol. 2003. 32: 559-66
2. Bavan L, Wijendra A, Kothari A. Efficacy of treatment interventions for primary aneurysmal bone cysts: A systematic review. Bone Jt Open. 2021. 2: 125-33
3. Cottalorda J, Bourelle S. Modern concepts of primary aneurysmal bone cyst. Arch Orthop Trauma Surg. 2007. 127: 105-14
4. Deventer N, Schulze M, Gosheger G, de Vaal M, Deventer N. Primary aneurysmal bone cyst and its recent treatment options: A comparative review of 74 cases. Cancers (Basel). 2021. 13: 2362
5. Doyle A, Field A, Graydon A. Recurrent aneurysmal bone cyst of the cervical spine in childhood treated with doxycycline injection. Skeletal Radiol. 2015. 44: 609-12
6. Lin PP, Brown C, Raymond AK, Deavers MT, Yasko AW. Aneurysmal bone cysts recur at juxtaphyseal locations in skeletally immature patients. Clin Orthop Relat Res. 2008. 466: 722-8
7. Liu X, Han SB, Si G, Yang SM, Wang CM, Jiang L. Percutaneous albumin/doxycycline injection versus open surgery for aneurysmal bone cysts in the mobile spine. Eur Spine J. 2019. 28: 1529-36
8. Mankin HJ, Hornicek FJ, Ortiz-Cruz E, Villafuerte J, Gebhardt MC. Aneurysmal bone cyst: A review of 150 patients. J Clin Oncol. 2005. 23: 6756-62
9. Mann M, Munavalli GS, Amatangelo L, Morrison N. Improper potency and impurities in compounded polidocanol. J Drugs Dermatol. 2019. 18: 1124-7
10. Mascard E, Gomez-Brouchet A, Lambot K. Bone cysts: Unicameral and aneurysmal bone cyst. Orthop Traumatol Surg Res. 2015. 101: S119-27
11. Mendenhall WM, Zlotecki RA, Gibbs CP, Reith JD, Scarborough MT, Mendenhall NP. Aneurysmal bone cyst. Am J Clin Oncol. 2006. 29: 311-5
12. Nakayama S, Nishio J, Aoki M, Koga K, Nabeshima K, Yamamoto T. Ubiquitin-specific peptidase 6 (USP6)-associated fibroblastic/myofibroblastic tumors: Evolving concepts. Cancer Genomics Proteomics. 2021. 18: 93-101
13. Novais EN, Rose PS, Yaszemski MJ, Sim FH. Aneurysmal bone cyst of the cervical spine in children. J Bone Joint Surg Am. 2011. 93: 1534-43
14. Perlmutter DH, Campbell S, Rubery PT, Vates EG, Silberstein HJ. Aneurysmal bone cyst: Surgical management in the pediatric cervical spine. Spine (Phila Pa 1976). 2009. 34: E50-3
15. Rapp TB, Ward JP, Alaia MJ. Aneurysmal bone cyst. JAAOS J Am Acad Orthop Surg. 2012. 20: 233-41
16. Rastogi S, Varshney MK, Trikha V, Khan SA, Choudhury B, Safaya R. Treatment of aneurysmal bone cysts with percutaneous sclerotherapy using polidocanol. J Bone Joint Surg Br. 2006. 88: 1212-6
17. Sayago LR, Remondino RG, Tello CA, Piantoni L, Wilson IA, Galaretto E. Aneurysmal bone cysts of the spine in children: A review of 18 cases. Global Spine J. 2020. 10: 875-80
18. Sebaaly A, Ghostine B, Kreichati G, Mallet JF, Glorion C, Moussa R. Aneurysmal bone cyst of the cervical spine in children: A review and a focus on available treatment options. J Pediatr Orthop. 2015. 35: 693-702
19. Shiels WE 2nd, Beebe AC, Mayerson JL. Percutaneous doxycycline treatment of juxtaphyseal aneurysmal bone cysts. J Pediatr Orthop. 2016. 36: 205-12
20. Shiels WE 2nd, Mayerson JL. Percutaneous doxycycline treatment of aneurysmal bone cysts with low recurrence rate: A preliminary report. Clin Orthop Relat Res. 2013. 471: 2675-83
21. Strohm JA, Strohm PC, Kühle J, Schmal H, Zwingmann J. Management of juvenile and aneurysmal bone cysts: A systematic literature review with meta-analysis. Eur J Trauma Emerg Surg. 2022. 49: 361-72
22. Topouchian V, Mazda K, Hamze B, Laredo JD, Penneçot GF. Aneurysmal bone cysts in children: Complications of fibrosing agent injection. Radiology. 2004. 232: 522-6
23. Varshney MK, Rastogi S, Khan SA, Trikha V. Is sclerotherapy better than intralesional excision for treating aneurysmal bone cysts?. Clin Orthop Relat Res. 2010. 468: 1649-59
24. Wong MN, Nicol K, Murakami JW. Image-guided percutaneous management of skull and spine giant cell tumors: Case report of 2 challenging cases successfully treated with doxycycline sclerotherapy. World Neurosurg X. 2020. 5: 100061
25. Woon JT, Hoon D, Graydon A, Flint M, Doyle AJ. Aneurysmal bone cyst treated with percutaneous doxycycline: Is a single treatment sufficient?. Skeletal Radiol. 2019. 48: 765-71
26. Ye Y, Pringle LM, Lau AW, Riquelme DN, Wang H, Jiang T. TRE17/USP6 oncogene translocated in aneurysmal bone cyst induces matrix metalloproteinase production via activation of NF-kB. Oncogene. 2010. 29: 3619-29