- Department of Neurological Surgery, Oregon Health and Science University, Portland, Oregon, USA
- Department of Neurosurgery, West Virginia University, Morgantown, West Virginia, USA
- Department of Diagnostic Radiology, Oregon Health and Science University, Portland, Oregon, USA
- Department of ENT, Kaiser Permanente, Sacramento, California, USA
- Department of Neurosurgery, Kaiser Permanente, Sacramento, California, USA
Correspondence Address:
Jeremy Ciporen
Department of ENT, Kaiser Permanente, Sacramento, California, USA
DOI:10.4103/2152-7806.196773
Copyright: © 2016 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Jeremy Ciporen, Brandon P. Lucke-Wold, Gustavo Mendez, Anton Chen, Amit Banerjee, Paul T. Akins, Ben J. Balough. Single-staged resections and 3D reconstructions of the nasion, glabella, medial orbital wall, and frontal sinus and bone: Long-term outcome and review of the literature. 26-Dec-2016;7:
How to cite this URL: Jeremy Ciporen, Brandon P. Lucke-Wold, Gustavo Mendez, Anton Chen, Amit Banerjee, Paul T. Akins, Ben J. Balough. Single-staged resections and 3D reconstructions of the nasion, glabella, medial orbital wall, and frontal sinus and bone: Long-term outcome and review of the literature. 26-Dec-2016;7:. Available from: http://surgicalneurologyint.com/surgicalint_articles/single%e2%80%91staged-resections-and-3d-reconstructions-of-the-nasion-glabella-medial-orbital-wall-and-frontal-sinus-and-bone-long%e2%80%91term-outcome-and-review-of-the-literature/
Abstract
Background:Aesthetic facial appearance following neurosurgical ablation of frontal fossa tumors is a primary concern for patients and neurosurgeons alike. Craniofacial reconstruction procedures have drastically evolved since the development of three-dimensional computed tomography imaging and computer-assisted programming. Traditionally, two-stage approaches for resection and reconstruction were used; however, these two-stage approaches have many complications including cerebrospinal fluid leaks, necrosis, and pneumocephalus.
Case Description:We present two successful cases of single-stage osteoma resection and craniofacial reconstruction in a 26-year-old female and 65-year-old male. The biopolymer implants were preselected and contoured based on imaging prior to surgery. The ideal selection of appropriate flaps for reconstruction was imperative. The flaps were well vascularized and included a pedicle for easy translocation. Using a titanium mesh biopolymer implant for reconstruction in conjunction with a forehead flap proved advantageous, and the benefits of single-stage approaches were apparent. The patients recovered quickly after the surgery with complete resection of the osteoma and good aesthetic appearance. The flap adhered to the biopolymer implant, and the cosmetic appearance years after surgery remained decent. The gap between the bone and implant was less than 2 mm. The patients are highly satisfied with the symmetrical appearance of the reconstruction.
Conclusions:Advances in technology are allowing neurosurgeons unprecedented opportunities to design complex yet feasible single-stage craniofacial reconstructions that improve a patient's quality of life by enhancing facial contours, aesthetics, and symmetry.
Keywords: Biopolymers, craniofacial reconstruction, implants, osteoma, single-stage approach
INTRODUCTION
Neurosurgical treatment for anterior cranial fossa tumors often requires extensive alteration to the surrounding bony anatomy.[
Utilizing the criteria established by Yano et al. for skull-based defects, it may be possible for the neurosurgeon to determine when it is safe and efficient to use a one-stage vs. two-stage approach.[
CASE SERIES
Case 1
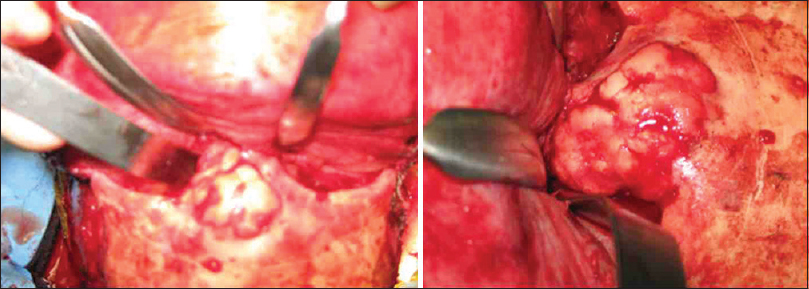
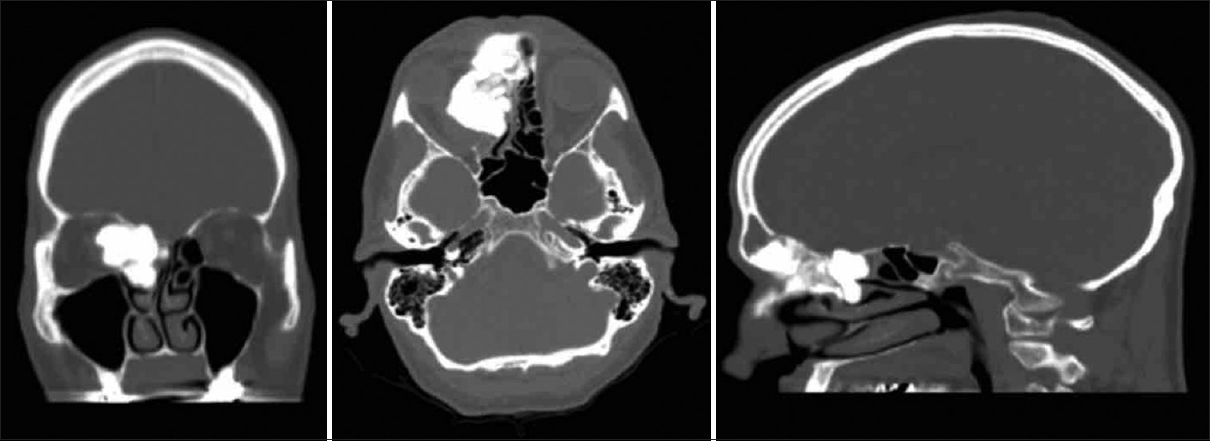
A 26-year-old female florist presented with a craniofacial defect in the area of the nasion and glabella. She was concerned about the cosmetic appearance of the bony growth. A computed tomography (CT) scan revealed an enlarged osteoma in the area of the nasion and glabella and medial orbital wall. A surgical plan was developed for osteoma resection and craniofacial reconstruction. Preoperative planning in real time web meetings was utilized to design the plan for resection as well as 3D implant for reconstruction. An intraoperative view of the osteoma can be appreciated in
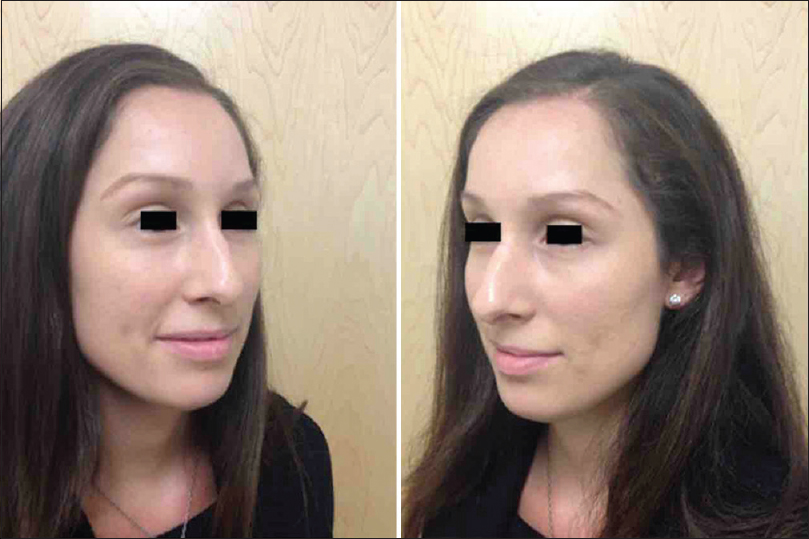
Good cosmetic outcomes and restoration of normal skull and skin contour was achieved. The patient had an intraoperative lumbar drain placed for brain relaxation that was done to avoid frontal lobe retraction during tumor resection. The patient required a blood patch for low-pressure headache after lumbar drain removal. No cerebrospinal fluid (CSF) rhinorrhea occurred. The patient and her family stated that they were highly satisfied with the outcome [
Case 2
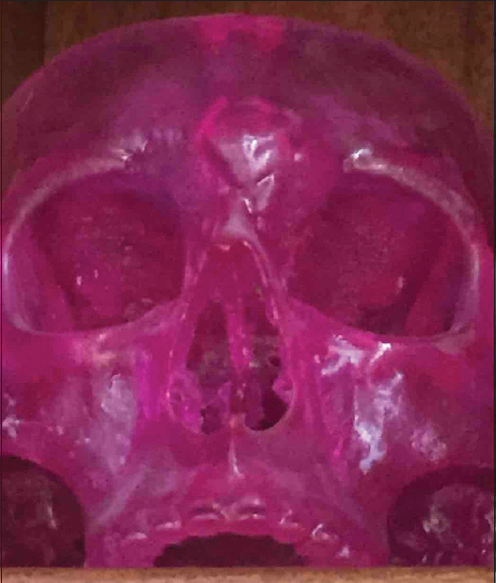
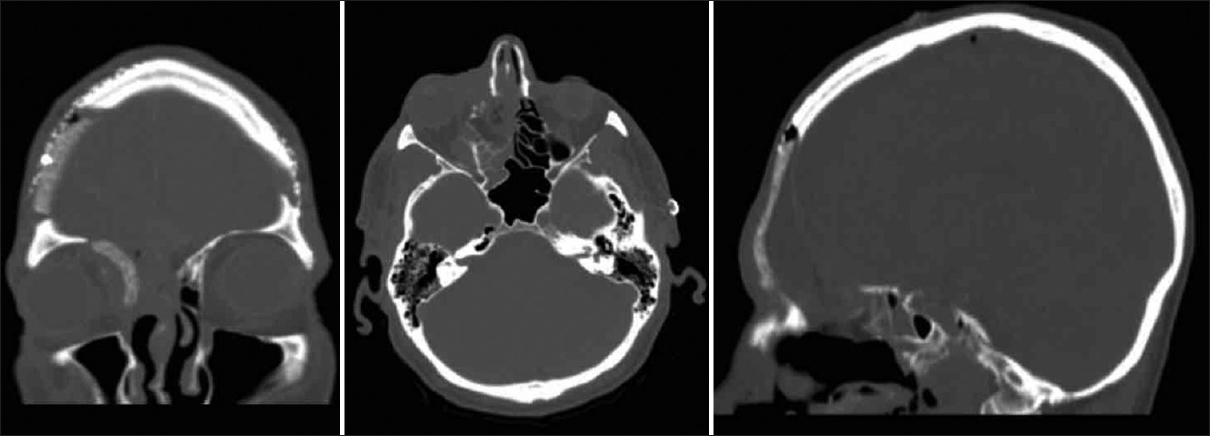
A 65-year-old male presented with diplopia secondary to medial rectus displacement by the large osteoma. A CT scan revealed a large osteoma invading the medial orbital wall, glabella, nasion, and frontal sinus [
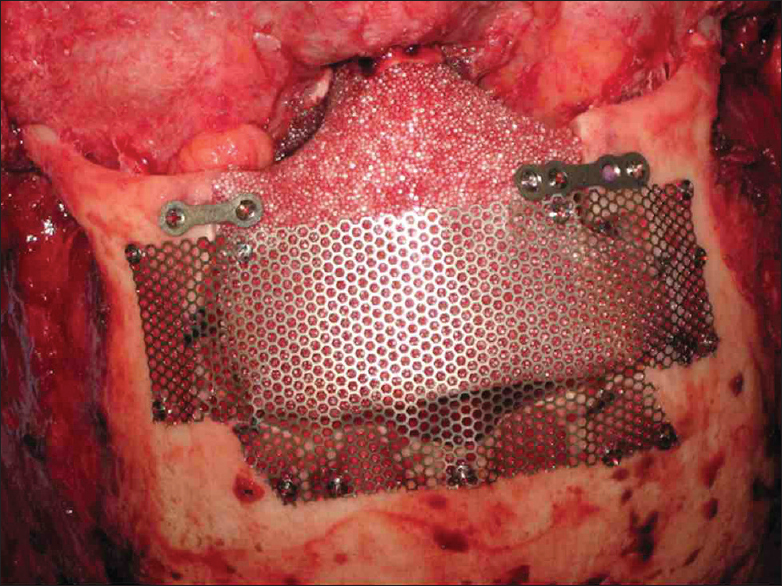
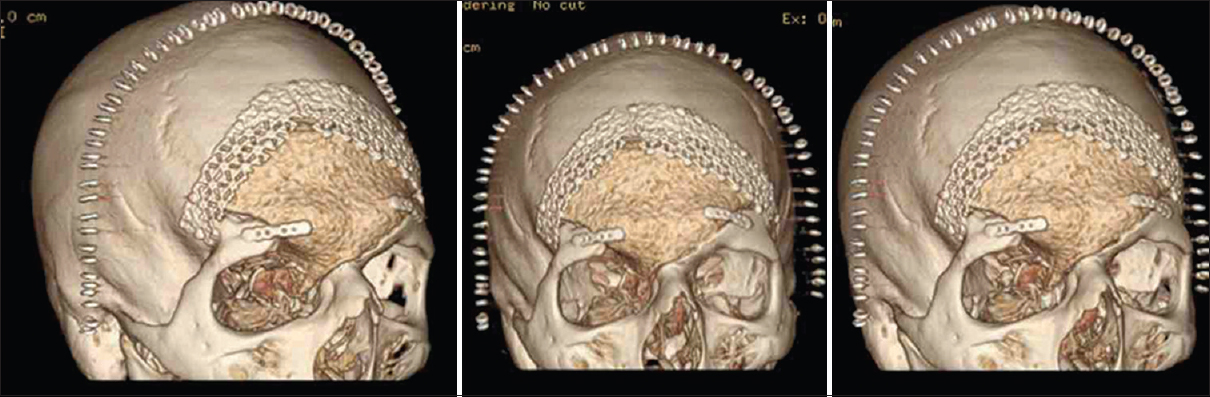
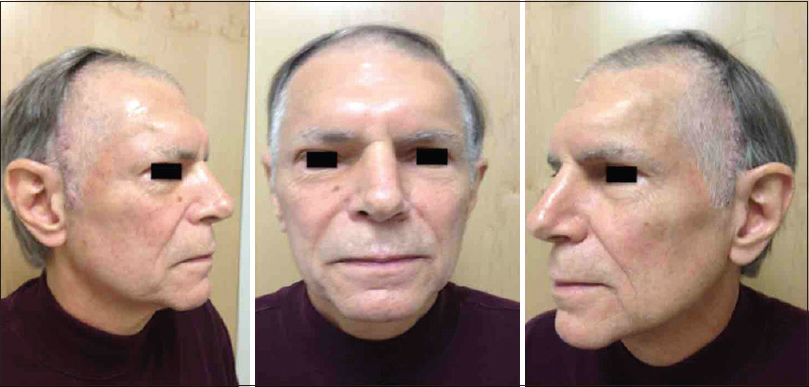
Post-surgical recovery was uneventful. Objective outcomes were considered good with the implant resting passively on the surrounding bone. The “masquerade mask” 3D reconstruction was performed to ensure a great fit with contoured symmetry. The patient reports high satisfaction with good aesthetic appearance [
DISCUSSION
Complex bony deformities in the craniofacial region are common following surgical tumor resection.[
Depending on case complexity, the neurosurgeon must decide if a single-stage or two-stage approach is best for resection and reconstruction. A two-stage approach has clear limitations. By re-exposing the patient to a second surgery, the likelihood of flap-associated infection drastically increases.[
Autologous bone grafts are the gold-standard for reconstruction, but due to a lack of sufficient donor sites, implants are more commonly used.[
The nasion-glabellar region is a very challenging region to reconstruct. The goal is to return a patient to an appearance that is consistent with their normal anatomy. Both of these patients had significant and noticeable preoperative deformities involving the regions of the nasion, glabella, and medial orbital wall. Preoperative real time online web-based 3D planning of the resection was essential in understanding the extent of the defect. This approach also allowed the surgeon to actively plan a tailored reconstruction. During the planning of the osteoma resections, it was determined that a larger area of frontal bone was to be removed laterally to create a more uniform appearance of the frontal bone. As such, we have coined the phrase “masquerade mask” reconstruction to describe the technique.
CONCLUSION
Craniofacial reconstruction following ablative neurosurgery is necessary to improve patient's quality of life. Historically, two-stage approaches have been used for resection and reconstruction. With the advent of 3D imaging, computer-assisted programming, and preoperative planning, it is now feasible to conduct single-stage approaches. Single-stage 3D preoperative planning provides the patient and healthcare system with multiple potential benefits. These benefits would be cost savings, avoiding multiple hospitalizations, decreased complications, good long-term cosmetic results, and high patient satisfaction.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form the patient(s) has/have given his/her/their consent for his/her/their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Acknowledgements
The authors would like to thank the Oregon Health and Science University for use of facilities. Brandon Lucke-Wold received a Pre-doctoral Grant from the American Foundation of Pharmaceutical Education and American Association of Pharmaceutical Scientists, an American Medical Association Seed Grant, Sigma Xi Grants in Aid of Research, and a Neurosurgery Research and Education Foundation Medical Student Summer Research Fellowship.
References
1. Akadiri OA. Evolution and trends in reconstructive facial surgery: An update. J Maxillofac Oral Surg. 2012. 11: 466-72
2. Bluebond-Langner R, Rodriguez ED. Application of skeletal buttress analogy in composite facial reconstruction. Craniomaxillofac Trauma Reconstr. 2009. 2: 19-25
3. Chae MP, Rozen WM, McMenamin PG, Findlay MW, Spychal RT, Hunter-Smith DJ. Emerging Applications of Bedside 3D Printing in Plastic Surgery. Front Surg. 2015. 2: 25-
4. Chenard KE, Teven CM, He TC, Reid RR. Bone morphogenetic proteins in craniofacial surgery: Current techniques, clinical experiences, and the future of personalized stem cell therapy. J Biomed Biotech 2012. 2012. p. 601549-
5. Demonte F, Moore BA, Chang DW. Skull base reconstruction in the pediatric patient. Skull Base. 2007. 17: 39-51
6. Dlouhy BJ, Chae MP, Teo C. The supraorbital eyebrow approach in children: Clinical outcomes, cosmetic results, and complications. J Neurosurg Pediatr. 2015. 15: 12-9
7. Gil Z, Abergel A, Leider-Trejo L, Khafif A, Margalit N, Amir A. A comprehensive algorithm for anterior skull base reconstruction after oncological resections. Skull Base. 2007. 17: 25-37
8. Gordon CR, Murphy RJ, Coon D, Basafa E, Otake Y, Al Rakan M. Preliminary development of a workstation for craniomaxillofacial surgical procedures: Introducing a computer-assisted planning and execution system. J Craniofac Surg. 2014. 25: 273-83
9. Hemmy DC, David DJ, Herman GT. Three-dimensional reconstruction of craniofacial deformity using computed tomography. Neurosurgery. 1983. 13: 534-41
10. Kim HJ, Lee KH, Park SY, Kim HK. One-stage reconstruction for midfacial defect after radical tumor resection. Clin Exp Otorhinolaryngol. 2012. 5: 53-6
11. Kung WM, Chen ST, Lin CH, Lu YM, Chen TH, Lin MS. Verifying three-dimensional skull model reconstruction using cranial index of symmetry. PloS One. 2013. 8: e74267-
12. Kung WM, Lin FH, Hsiao SH, Chiu WT, Chyau CC, Lu SH. New reconstructive technologies after decompressive craniectomy in traumatic brain injury: The role of three-dimensional titanium mesh. J Neurotrauma. 2012. 29: 2030-7
13. Laedrach K, Lukes A, Raveh J. Reconstruction of Skull Base and Fronto-orbital Defects following Tumor Resection. Skull Base. 2007. 17: 59-72
14. Lau D, McDermott MW. A Method for Combining Thin and Thick Malleable Titanium Mesh in the Repair of Cranial Defects. Cureus. 2015. 7: e267-
15. Lee SJ, Lee HP, Tse KM, Cheong EC, Lim SP. Computer-aided design and rapid prototyping-assisted contouring of costal cartilage graft for facial reconstructive surgery. Craniomaxillofac Trauma Reconstr. 2012. 5: 75-82
16. Logan H, Wolfaardt J, Boulanger P, Hodgetts B, Seikaly H. Evaluation of the accuracy of cone beam computerized tomography (CBCT): Medical imaging technology in head and neck reconstruction. J Otolaryngol Head Neck Surg. 2013. 42: 25-
17. Mao JJ, Giannobile WV, Helms JA, Hollister SJ, Krebsbach PH, Longaker MT. Craniofacial tissue engineering by stem cells. J Dent Res. 2006. 85: 966-79
18. Mao JJ, Stosich MS, Moioli EK, Lee CH, Fu SY, Bastian B. Facial reconstruction by biosurgery: Cell transplantation versus cell homing. Tissue Eng Part B Rev. 2010. 16: 257-62
19. Parthasarathy J. 3D modeling, custom implants and its future perspectives in craniofacial surgery. Ann Maxillofac Surg. 2014. 4: 9-18
20. Rosen CL, Sedney C, Sivak J. Single-stage complex reconstruction for fibrous dysplasia: A case report. Skull Base. 2008. 18: 275-9
21. Sindou M, Nebbal M, Guclu B. Cavernous sinus meningiomas: Imaging and surgical strategy. Adv Tech Stand Neurosurg. 2015. 42: 103-21
22. Wan DC, Wong VW, Longaker MT. Craniofacial reconstruction with induced pluripotent stem cells. J Craniofac Surg. 2012. 23: 623-6
23. Xia JJ, Gateno J, Teichgraeber JF. New clinical protocol to evaluate craniomaxillofacial deformity and plan surgical correction. J Oral Maxillofac Surg. 2009. 67: 2093-106
24. Yadav P. Recent advances in head and neck cancer reconstruction. Indian J Plast Surg. 2014. 47: 185-90
25. Yano T, Okazaki M, Tanaka K, Iida H, Aoyagi M, Tsunoda A. A new concept for classifying skull base defects for reconstructive surgery. J Neurol Surg B Skull Base. 2012. 73: 125-31
26. Zhao J, Liu C, Wu Z, Duan F, Wang K, Jia T. Craniofacial reconstruction evaluation by geodesic network. Comput Math Methods Med 2014. 2014. p. 943647-
27. Zhao L, Patel PK, Cohen M. Application of virtual surgical planning with computer assisted design and manufacturing technology to cranio-maxillofacial surgery. Arch Plast Surg. 2012. 39: 309-16