- Department of Neuroscience, Neurosurgery Unit, Città Della Scienza e Della Salute, Italy.
- Department of Pathologic, Città Della Scienza e Della Salute, Turin, Italy.
Correspondence Address:
Giuseppe Di Perna
Department of Neuroscience, Neurosurgery Unit, Città Della Scienza e Della Salute, Italy.
DOI:10.25259/SNI_903_2020
Copyright: © 2021 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Bianca Maria Baldassarre1, Federica Penner1, Luca Bertero2, Giuseppe Di Perna1, Marco Ajello1, Nicola Marengo1, Francesco Zenga1, Diego Garbossa1. Solitary late spinal metastasis from apocrine salivary duct carcinoma: Case report. 30-Mar-2021;12:122
How to cite this URL: Bianca Maria Baldassarre1, Federica Penner1, Luca Bertero2, Giuseppe Di Perna1, Marco Ajello1, Nicola Marengo1, Francesco Zenga1, Diego Garbossa1. Solitary late spinal metastasis from apocrine salivary duct carcinoma: Case report. 30-Mar-2021;12:122. Available from: https://surgicalneurologyint.com/?post_type=surgicalint_articles&p=10689
Abstract
Background: The salivary duct carcinomas (SDCs) are rare, high-grade neoplasms involving major salivary glands. Parotid is the most frequently involved gland (85%). Apocrine phenotype (histological presence of decapitation secretions) and androgen reception expression define SDC. The clinical course of these tumors is characterized by aggressive local behavior with extraglandular extension, high recurrence rates, early metastases, and poor prognoses. Despite aggressive surgical/radiation therapy management, the rates of locoregional and metastatic relapses are high, and the mortality rates over 48 months approach 65%. Notably, there is no treatment algorithm available for managing vertebral metastases from apocrine SDC.
Case Description: An elderly male presented with MR/CT findings of an isolated T11 vertebral metastasis attributed to a previously treated parotid SDC. On both CT/MR, it was an osteolytic lesion and demonstrated spinal canal infiltration. The patient underwent surgical biopsy/decompression/resection, following which the lesion histopathologically proved to be a SDC. The patient was subsequently treated with 30 Gy in 10 fractions within 2 weeks of discharge. One-month later, the MRI confirmed adequate epidural decompression without recurrence, and 9 months post-operatively, patient remained disease free.
Conclusion: Isolated metastasis attributed to parotid SDC followed by radiation therapy may result in tumor control.
Keywords: Apocrine salivary duct carcinoma, Carbon fiber, Salivary gland tumors, Separation surgery, Spinal metastases
INTRODUCTION
Salivary duct carcinomas (SDCs) are rare and account for approximately 6% of all malignant salivary gland tumors. SDC are malignant, rapidly-growing aggressive tumors with a high local recurrence rate, and high frequency of early metastases.[
CASE REPORT
Initial presentation of SDC
A 77-year-old male with a medical history of the left-sided parotid carcinoma presented with the MR/CT finding of an isolated metastatic T11 vertebral lesion diagnosed following a radical parotidectomy. The histological diagnosis was consistent with a SDC with apocrine differentiation. At that time, 3 lymph nodes were positive, and the patient, therefore, underwent subsequently adjuvant local radiotherapy.
Presentation with T11 metastatic SDC disease
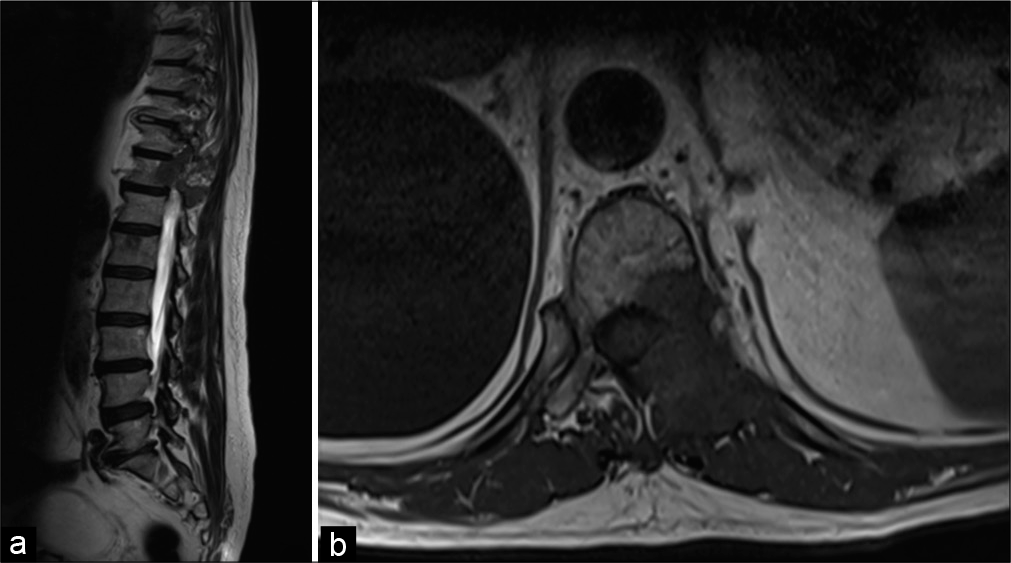
A chest-abdomen CT scan, performed 3 months later, showed an increase in size of the vertebral lesion and highlighted spinal instability. Further, the spinal MRI demonstrated infiltration of the spinal canal at the T11 level due to the SDC (i.e., Bilsky Grade 2 epidural compression) [
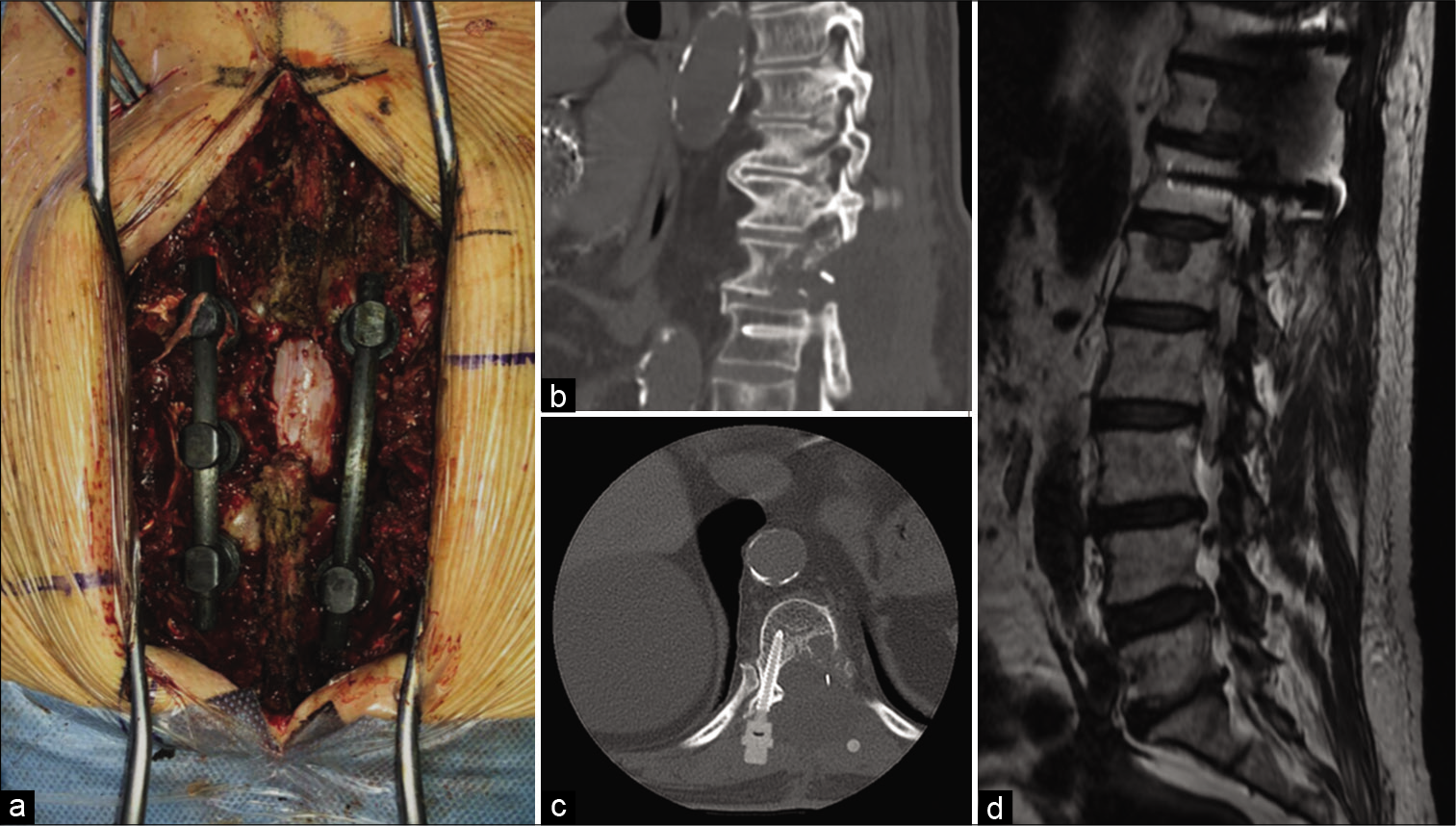
Figure 2:
Intraoperative image (a) displaying the circumferential decompression and vertebral fixation with carbon fiber system. Postoperative sagittal (b) and axial (c) CT scan showing the accuracy of fixation system placement. Postoperative MRI image (d) demonstrating the epidural decompression and the absence of local disease recurrence.
Histology
The initial histological diagnosis was consistent with a SDC with apocrine differentiation and 3 lymph nodes were positive. The T11 histopathology was the same. Immunohistochemical stainings for gross cystic disease fluid protein-15 (GCDFP15) and androgen receptor (e.g., for the T11 biopsy material) were diffusely positive; while cytokeratins 8/18 were focally positive [
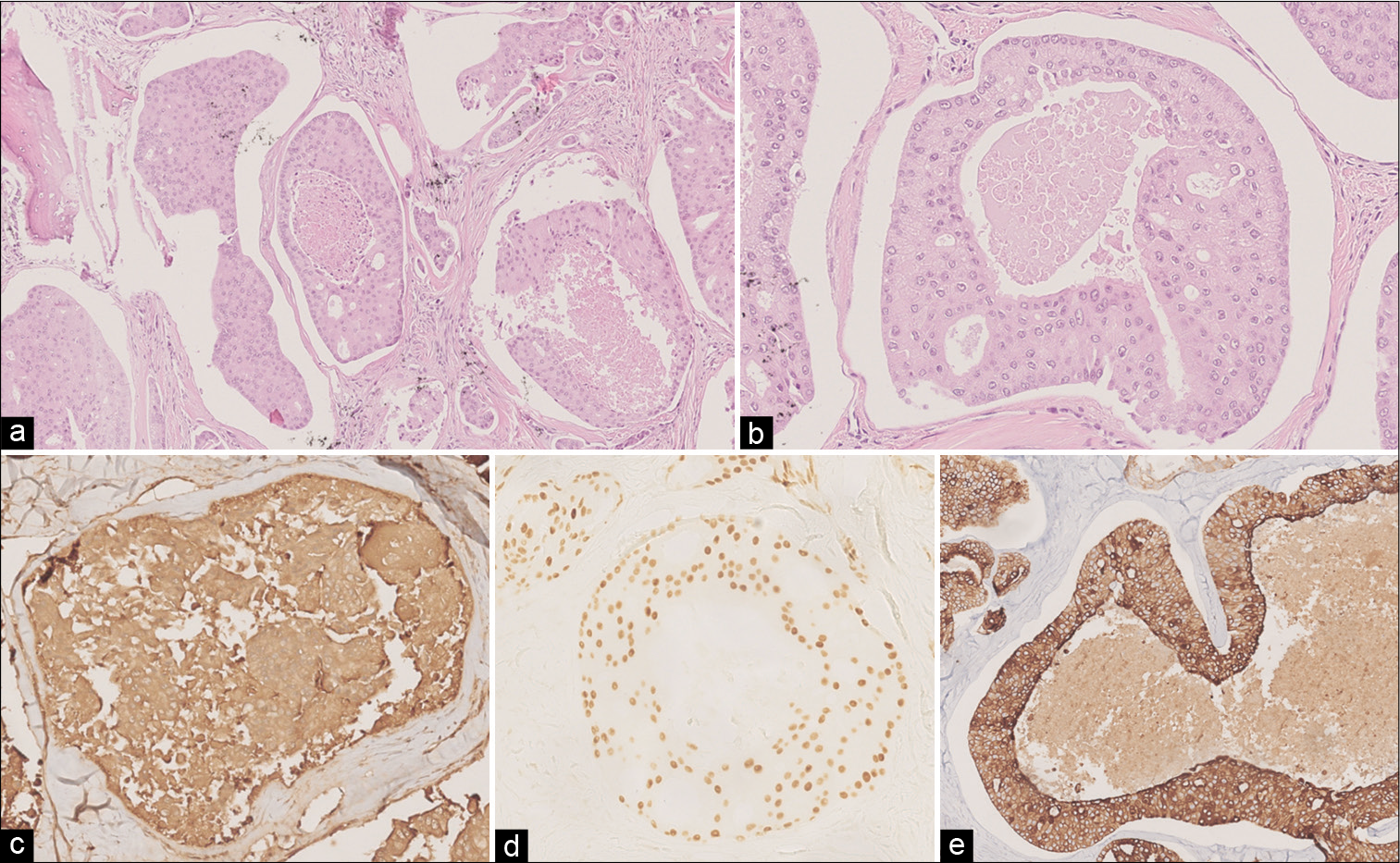
Figure 3:
Histological findings after vertebral lesion resection. H and E image (a: 100X, b: 200X) shows a bone infiltrating metastatic carcinoma with duct structures and comedonecrosis. Neoplastic cells were mildly pleomorphic with an eosinophilic cytoplasm and features consistent with apocrine differentiation. Immunohistochemical stainings showed a diffuse positivity for GCDFP15 (c) and androgen receptor (d), while cytokeratins 8/18 were focally positive (e).
Adjuvant radiation therapy
Subsequently, patient underwent adjuvant local radiotherapy and received 30 Gy in 10 fractions within 2 postoperative weeks. One-month post-operatively, the MRI confirmed adequate epidural decompression without tumor recurrence [
DISCUSSION
SDC are rare, high-grade neoplasms involving major salivary glands, and represent approximately 6% of all salivary gland cancers.[
Prognosis with surgery and radiation therapy
Although there is no specific management algorithm for the treatment of vertebral metastases from SDCs, those with single osteolytic lesions and high-grade epidural compression may be successfully treated with aggressive gross-total surgical resection followed by radiation therapy.[
Histology
Apocrine phenotype (histological presence of decapitation secretions) and androgen receptor expression defines SDC that may metastasize to the spine (e.g., to the T11 level in this case) warranting surgical extirpation followed by adjuvant radiation therapy.[
CONCLUSION
Three-year delayed metastatic parotid carcinoma of the T11 vertebral body was adequately managed with separation surgery achieved through an extracavitary transpedicular partial anterior corpectomy and posterior pedicle screw fusion, followed by radiation therapy.
Ethical committee approval
All clinical and radiological data were collected and retrospective analyzed. This study does not require any variations in patient’s treatment and no formal ethics committee approval was required.
Authors’ contributions
BMB writing and editing; FP conceptualization and revision; LB data collection; GD revision; MA data collection; writing and revision; FZ revision; DG conceptualization and supervision.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Barzilai O, Robin AM, O’Toole JE, Laufer I. Minimally invasive surgery strategies: Changing the treatment of spine tumors. Neurosurg Clin N Am. 2020. 31: 201-9
2. Bilsky M, Smith M. Surgical approach to epidural spinal cord compression. Hematol Oncol Clin North Am. 2006. 20: 1307-17
3. Cofano F, di Perna G, Alberti A, Baldassarre BM, Ajello M, Marengo N. Neurological outcomes after surgery for spinal metastases in symptomatic patients: Does the type of decompression play a role? A comparison between different strategies in a 10-year experience. J Bone Oncol. 2020. 26: 100340
4. Cofano F, di Perna G, Marengo N, Ajello M, Melcarne A, Zenga F. Transpedicular 3D endoscope-assisted thoracic corpectomy for separation surgery in spinal metastases: Feasibility of the technique and preliminary results of a promising experience. Neurosurg Rev. 2020. 43: 351-60
5. Cofano F, di Perna G, Monticelli M, Marengo N, Ajello M, Mammi M. Carbon fiber reinforced vs titanium implants for fixation in spinal metastases: A comparative clinical study about safety and effectiveness of the new “carbon-strategy”. J Clin Neurosci. 2020. 75: 106-11
6. Cofano F, di Perna G, Zenga F, Ducati A, Baldassarre B, Ajello M. The neurology-stability-epidural compression assessment: A new score to establish the need for surgery in spinal metastases. Clin Neurol Neurosurg. 2020. 195: 105896
7. Cofano F, Monticelli M, Ajello M, Zenga F, Marengo N, di Perna G. The targeted therapies era beyond the surgical point of view: What spine surgeons should know before approaching spinal metastases. Cancer Control. 2019. 26: 1-13
8. di Perna G, Cofano F, Mantovani C, Badellino S, Marengo N, Ajello M. Separation surgery for metastatic epidural spinal cord compression: A qualitative review. J Bone Oncol. 2020. 25: 100320
9. Gasbarrini A, Cappuccio M, Mirabile L, Bandiera S, Terzi S, Bròdano GB. Spinal metastases: Treatment evaluation algorithm. Eur Rev Med Pharmacol Sci. 2004. 8: 265-74
10. Hosal AS, Fan C, Barnes L, Myers EN. Salivary duct carcinoma. Otolaryngol Head Neck Surg. 2003. 129: 720-5
11. Jaspers HC, Verbist BM, Schoffelen R, Mattijssen V, Slootweg PJ, van der Graaf WT. Androgen receptor-positive salivary duct carcinoma: A disease entity with promising new treatment options. J Clin Oncol. 2011. 29: e473-6
12. Jayaprakash V, Merzianu M, Warren GW, Arshad H, Hicks WL, Rigual NR. Survival rates and prognostic factors for infiltrating salivary duct carcinoma: Analysis of 228 cases from the surveillance, epidemiology, and end results database. Head Neck. 2014. 36: 694-701
13. Laufer I, Rubin DG, Lis E, Cox BW, Stubblefield MD, Yamada Y. The NOMS framework: Approach to the treatment of spinal metastatic tumors. Oncologist. 2013. 18: 744-51
14. Roh JL, Lee JI, Choi SH, Nam SY, Kim SO, Cho KJ. Prognostic factors and oncologic outcomes of 56 salivary duct carcinoma patients in a single institution: High rate of systemic failure warrants targeted therapy. Oral Oncol. 2014. 50: e64-6
15. Salovaara E, Hakala O, Bäck L, Koivunen P, Saarilahti K, Passador-Santos F. Management and outcome of salivary duct carcinoma in major salivary glands. Eur Arch Otorhinolaryngol. 2013. 270: 281-5
16. Wee DT, Thomas AA, Bradley PJ. Salivary duct carcinoma: What is already known, and can we improve survival?. J Laryngol Otol. 2012. 126: S2-7
17. Williams L, Thompson LD, Seethala RR, Weinreb I, Assaad AM, Tuluc M. Salivary duct carcinoma: The predominance of apocrine morphology, prevalence of histologic variants, and androgen receptor expression. Am J Surg Pathol. 2015. 39: 705-13
18. Williams MD, Roberts D, Blumenschein GR, Temam S, Kies MS, Rosenthal DI. Differential expression of hormonal and growth factor receptors in salivary duct carcinomas: Biologic significance and potential role in therapeutic stratification of patients. Am J Surg Pathol. 2007. 31: 1645-52