- Department of Neurosurgery, Santa Ines Hospital, Cuenca, Azuay, Ecuador.
- Department of Family Medicine, System of Public Health, Cuenca, Azuay, Ecuador.
- Critical Care Unit, Santa Ines Hospital, Cuenca, Azuay, Ecuador.
Correspondence Address:
Sebastian Lopez
Critical Care Unit, Santa Ines Hospital, Cuenca, Azuay, Ecuador.
DOI:10.25259/SNI-46-2019
Copyright: © 2019 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Sebastian Lopez, Franklin Santillan, Juan Jose Diaz, Pedro Mogrovejo. Spinal cord compression by multiple cysticercosis. 07-Jun-2019;10:94
How to cite this URL: Sebastian Lopez, Franklin Santillan, Juan Jose Diaz, Pedro Mogrovejo. Spinal cord compression by multiple cysticercosis. 07-Jun-2019;10:94. Available from: http://surgicalneurologyint.com/surgicalint-articles/9352/
Abstract
Background:Neurocysticercosis (NCC) is the most common parasitic infection involving the central nervous system in endemic areas. Notably, spinal involvement occurs in only 0.7%–3% of patients.
Case Description:A 58-year-old female presented with progressive spinal cord compression attributed to multiple cystic intradural extramedullary thoracic lesions. She underwent laminectomy at two separate thoracic levels; this involved excision of the upper T4–T6, and just exploration of the lower T9–T11 lesions. One year postoperatively, she exhibited a residual paraparesis.
Conclusion:Spinal NCC must be considered among the differential diagnostic considerations for patients presenting with spinal intramedullary or subarachnoid/extramedullary cystic lesions. Although they are typically found in endemic regions, those who have traveled to these locations are also susceptible. Even though it is considered a benign condition, spinal NCC may cause permanent and irreversible neurological damage. Public health policies should, therefore, be developed to help control the spread and transmission of NCC.
Keywords: Intradural, paraparesis, Rare, Spinal cord compression, Spinal cysticercosis, Subarachnoid
INTRODUCTION
Neurocysticercosis (NCC) is a major public health issue in developing countries that are also becoming increasingly prevalent in developed nations.[
Here, we report a 58-year-old female who presented with progressive thoracic myelopathy. Although spinal NCC was not considered among the preoperative differential diagnostic considerations, surgery provided both neurological decompression and pathological confirmation of spinal NCC.
CASE REPORT
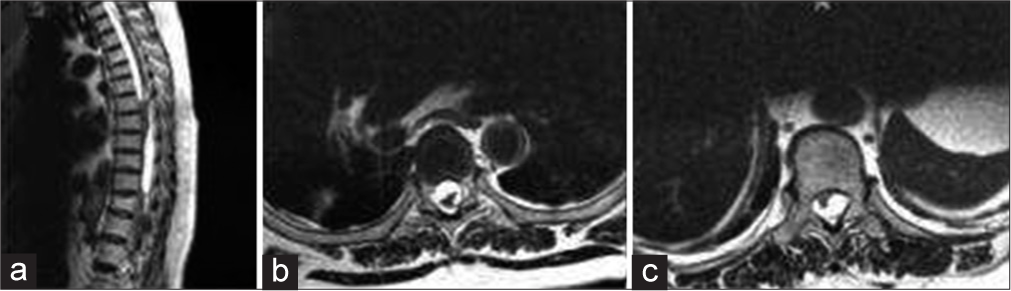
A 58-year-old female presented with a 6-month history of progressive paraparesis, sensory disturbance, and sphincter dysfunction. Ultimately she became paraplegic (motor function 0/5 in both lower extremities), was severely spastic, and exhibited a T4 sensory level to pin appreciation. The thoracic magnetic resonance (MR) imaging revealed multiple intradural extramedullary cystic lesions at the T4–6 and T9–11 levels [
Surgical procedure
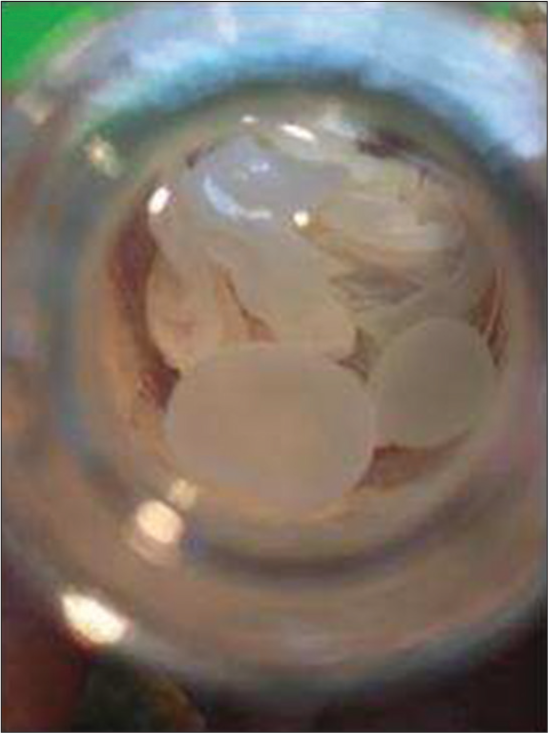
The patient required two separate laminectomies involving both the T4–T6 and T9–T11 levels for resection of spinal NCC cysts. At the T4–T6 levels, once the dura was opened, 12 cystic lesions of different sizes protruded through the arachnoid gap and were removed along with the arachnoid membrane [
Pathology and follow-up
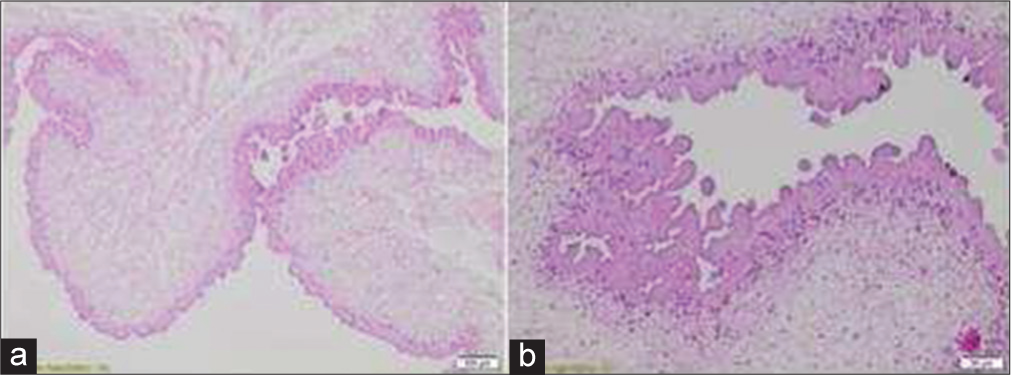
The histopathological report confirmed spinal NCC [
Immediately postoperatively, the patient regained 2/5 motor function in the lower extremities. The MR 1 month later showed partial resolution of the preoperative cord compression at both the T4–6 and T9–11 levels. In addition, corticosteroids were continued for the next 3 months to reduce inflammation [
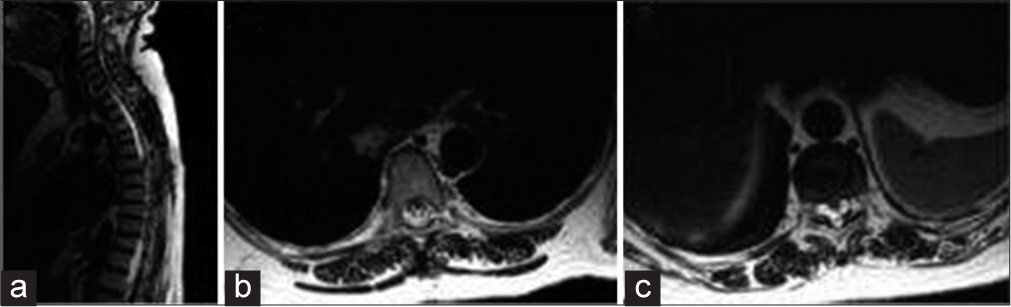
Figure 4
Postoperative T2-weighted magnetic resonance imaging. (a) Sagittal view showing the signal intensity of cerebrospinal fluid without septations in the upper thoracic level and partial resolution in the low level. (b and c) Axial view with a resolution of the compression in the upper level (b) and persistence of the cysts in the low level.
DISCUSSION
Frequency of spinal NCC
Spinal NCC is rare (0.7%–3%) and occurs either in intradural intramedullary or intradural extramedullary locations. In most cases it is associated with intracranial lesions, suggesting that the predominant etiology is direct dissemination (e.g., drop lesions) through the cerebrospinal fluid (CSF). However, there are other cases of isolated spinal involvement, attributed to retrograde migration of NCC through the epidural venous plexus. In a prior study, 31.5% of patients in Mexico with spinal cysticerci had a previous history of subarachnoid cysticercosis.[
Location of spinal NCC
Spinal NCC most frequently involves the lumbar and thoracic regions. In two studies, Indian patients typically presented with solitary intramedullary NCC lesions, while Mexican patients mostly had multiple extramedullary NCC (e.g., 68.4%).[
Clinical presentation
Spinal NCC may result in spinal cord and/or nerve roots symptoms/signs attributed to arachnoiditis, and/or focal compression.[
Surgery for spinal NCC
Surgical treatment is indicated where patients exhibit severe neurological deficits or when definitive histopathology is required to establish the diagnosis. When removing these lesions microsurgically, it is best to preserve the cyst wall; nevertheless, sometimes this is difficult to achieve, particularly when severe arachnoiditis is present (e.g., as in this case at the T9–11 levels where the cyst ruptured). Medical treatment includes a combination of Albendazole and corticosteroids. Despite adequate therapy, some patients may still continue to exhibit severe perioperative/postoperative morbidity (i.e., resultant neurological sequelae), or mortality.[
CONCLUSION
Spinal cysticercosis NCC is rare occurring in only 0.7%–3% of NCC in endemic regions. It should be considered among the differential diagnoses for patients with spinal cystic lesions, located either intradural/intramedullary or intradural/extramedullary (i.e., in the subarachnoid space). Although these lesions are benign, gross total excision resulting in resolution of the attendant neurological deficits is not always achieved. Major future efforts should be made to provide primary prevention of NCC and spinal NCC through improved sanitary measures.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form the patient(s) has/have given his/her/their consent for his/her/their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Bansal S, Suri A, Sharma MC, Kakkar A. Isolated lumbar intradural extra medullary spinal cysticercosis simulating tarlov cyst. Asian J Neurosurg. 2017. 12: 279-82
2. Callacondo D, Garcia HH, Gonzales I, Escalante D, Nash TE. High frequency of spinal involvement in patients with basal subarachnoid neurocysticercosis. Neurology. 2012. 78: 1394-400
3. Cantey PT, Coyle CM, Sorvillo FJ, Wilkins PP, Starr MC, Nash TE. Neglected parasitic infections in the United States: Cysticercosis. Am J Trop Med Hyg. 2014. 90: 805-9
4. Cárdenas G, Guevara-Silva E, Romero F, Ugalde Y, Bonnet C, Fleury A. Spinal Taenia solium cysticercosis in Mexican and Indian patients: A comparison of 30-year experience in two neurological referral centers and review of literature. Eur Spine J. 2016. 25: 1073-81
5. Fleury A, Sciutto E, Larralde C. Neurocysticercosis is still prevalent in Mexico. Salud Publica Mex. 2012. 54: 632-6
6. Gupta S, Singh PK, Gupta B, Singh V, Azam A. Isolated primary intradural extramedullary spinal neurocysticercosis: A case report and review of literature. Acta Neurol Taiwan. 2009. 18: 187-92
7. Muralidharan V, Nair BR, Patel B, Rajshekhar V. Primary intradural extramedullary cervical spinal cysticercosis. World Neurosurg. 2017. 106: 1052.e5-000000-
8. Qi B, Ge P, Yang H, Bi C, Li Y. Spinal intramedullary cysticercosis: A case report and literature review. Int J Med Sci. 2011. 8: 420-3
9. Sharma R, Garg K, Agarwal D, Garg A, Sharma MC, Sharma BS. Isolated primary intradural extramedullary spinal cysticercosis. Neurol India. 2017. 65: 882-4