- Department of Neurosurgery, Hospital Central Sur de Alta Especialidad PEMEX, Tlalpan, Ciudad de Mexico, Mexico,
- Department of Imaging, Hospital Central Sur de Alta Especialidad PEMEX, Tlalpan, Ciudad de Mexico, Mexico,
- Department of Anatomical Pathology, Hospital Central Sur de Alta Especialidad PEMEX, Tlalpan, Ciudad de Mexico, Mexico,
- Department of Neurosurgery, Hospital Central Norte PEMEX, Azcapotzalco, Estado de Mexico, Mexico,
- Department of Neurosurgery, Hospital Vossan, Campeche, Mexico.
Correspondence Address:
Diego Ochoa-Cacique
Department of Neurosurgery, Hospital Central Sur de Alta Especialidad PEMEX, Tlalpan, Ciudad de Mexico, Mexico,
DOI:10.25259/SNI_649_2020
Copyright: © 2020 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Diego Ochoa-Cacique1, María Córdoba-Mosqueda1, José Ramón Aguilar-Calderón1, Martha Cristina Sánchez-Silva2, Rosa María Vicuña-González3, Abraham Ibarra-de la Torre1, Victor Andrés Reyes-Rodríguez4, José de Jesús Lomelí-Ramírez5, Óscar Medina-Carrillo1, Mauricio Daniel Sánchez-Calderón1, Erick Alberto Castañeda-Ramírez1, Ulises García-González1. Spinal drop of atypical choroid plexus papilloma in an adult patient: A case report and literature review. 25-Nov-2020;11:409
How to cite this URL: Diego Ochoa-Cacique1, María Córdoba-Mosqueda1, José Ramón Aguilar-Calderón1, Martha Cristina Sánchez-Silva2, Rosa María Vicuña-González3, Abraham Ibarra-de la Torre1, Victor Andrés Reyes-Rodríguez4, José de Jesús Lomelí-Ramírez5, Óscar Medina-Carrillo1, Mauricio Daniel Sánchez-Calderón1, Erick Alberto Castañeda-Ramírez1, Ulises García-González1. Spinal drop of atypical choroid plexus papilloma in an adult patient: A case report and literature review. 25-Nov-2020;11:409. Available from: https://surgicalneurologyint.com/surgicalint-articles/10414/
Abstract
Background: Choroid plexus papillomas (CPPs) are benign extra-axial tumors that originate from the choroid plexus; these tumors rarely have metastases, being at the spinal level the location with few reported cases.
Case Description: We report the case of a 48-year-old man with a history of atypical fourth ventricular CPP and gross total resection (GTR) in 2008. In 2015, he presented with radicular pain, decreased strength, and paresthesia in the left leg. Magnetic resonance imaging revealed a well-defined intradural ovoid lesion in the vertebral canal at the level of the L3-L4 intersomatic space. Subtotal resection of the tumor was performed. The patient recovered well, with relief of pain and no neurological deficit. A literature research few cases of CPP metastasis in adults. We describe here a fifth-decade male patient with a lumbar neoplasm, which according to the histopathologic characteristics and location is the first case of an atypical papilloma implant of the choroid plexus at this spinal level.
Conclusion: GTR of primary lesions and associated implants remains the gold standard for surgical treatment of CPP. Radiotherapy, stereotactic radiosurgery, and chemotherapy are adjuvant therapies for CPP but there is no definitive protocol for the management of implants. Proper follow-up of these patients is essential since spinal drop can appear many years after the initial presentation of CPP.
Keywords: Choroid plexus papilloma, Metastasis, Spinal drop
INTRODUCTION
Choroid plexus papillomas (CPPs) are extra-axial benign tumors that originate from the neuroepithelial lining of the choroid plexus. These tumors represent about 0.4–0.6% of all intracranial neoplasms and 2–4% of brain tumors in children and 1% of those in adults.[
CASE EXAMINATION
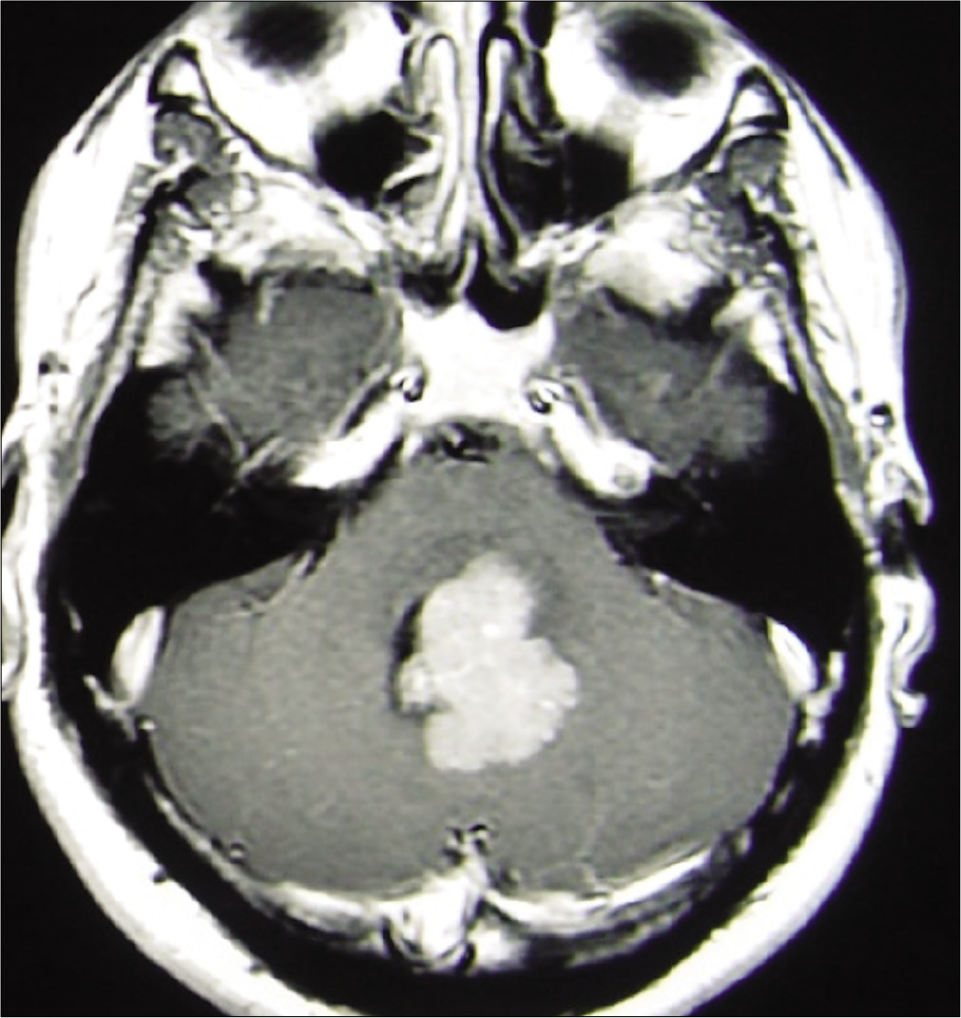
A 48-year-old male with a history of fourth ventricle atypical CPP diagnosed in 2008 underwent gross total resection (GTR) without any postoperative neurological deficits [
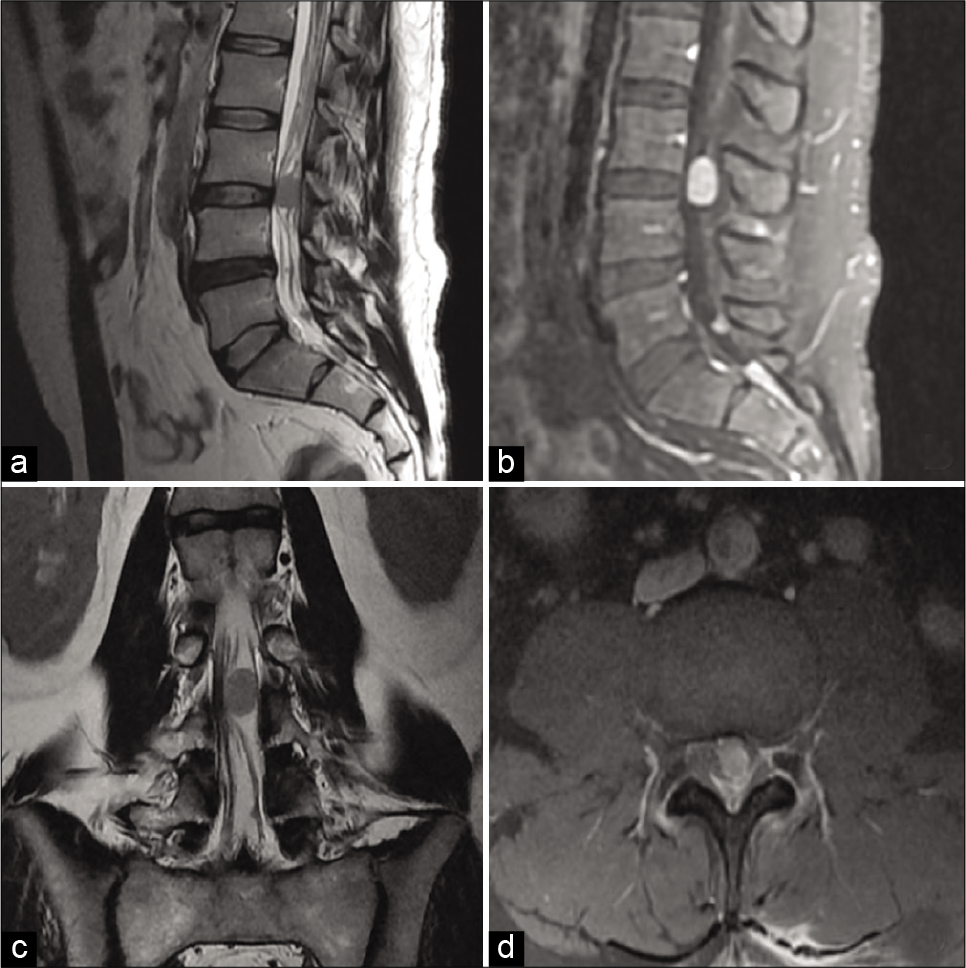
The neurological examination showed decreased left thigh strength quantified as 4+ according to the Royal Medical Research Council of Great Britain scale, left patellar reflex quantified as 1+ according to the muscle stretch reflex (deep tendon reflex) grading scale, and hypoesthesia in the third and fourth lumbar dermatomes. Spinal magnetic resonance imaging revealed a well-defined intradural ovoid lesion in the vertebral canal at the level of the L3-L4 intersomatic space with a length of 1.7 × 1.2 cm that was isointense on T1 and T2 sequences with homogeneous enhancement [
Figure 2:
Spinal magnetic resonance imaging. (a) Sagittal T2-weighted image (b) sagittal lumbar spine contrast material-enhanced T1-weighted image (c) coronal T2-weighted image (d) axial lumbar spine contrast material-enhanced T1-weighted image. In the vertebral canal at the level of the intersomatic space, L3L4 presents a well-defined ovoid lesion, isointense on T2 sequence, with a length of 1.7 × 1.2 cm, and intradural-extramedullary behavior that involves the spinal canal with homogeneous enhancement.
Differential diagnosis, investigations, and treatment
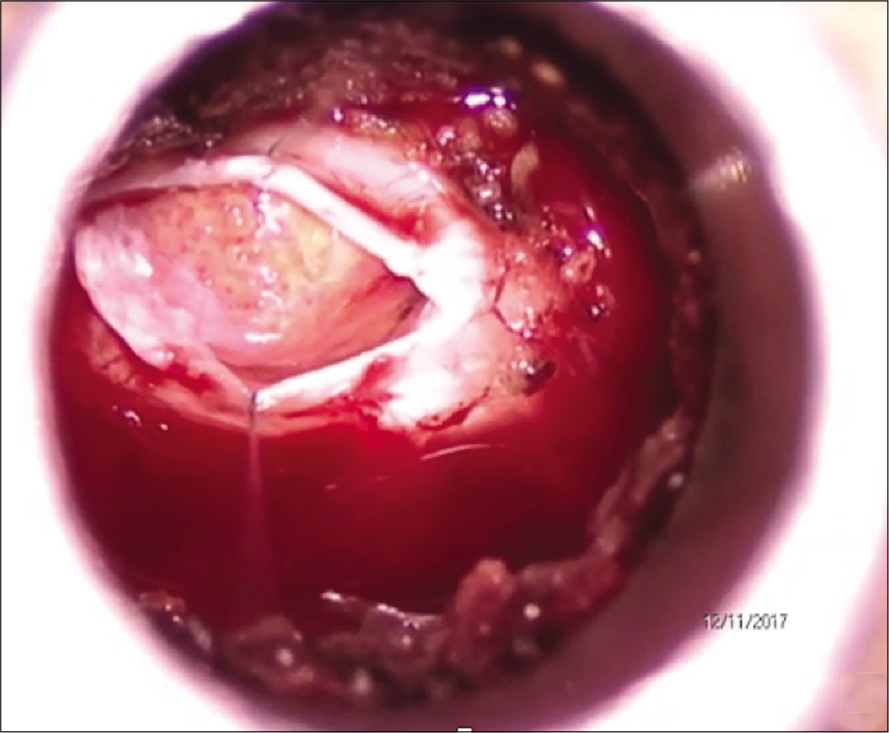
The patient underwent microscopy-assisted tubular microsurgery with neural decompression through left L3 hemilaminectomy with subtotal resection (STR). Intraoperatively, we found a soft yellow-colored lesion that was firmly adhered to the fourth lumbar nerve root [
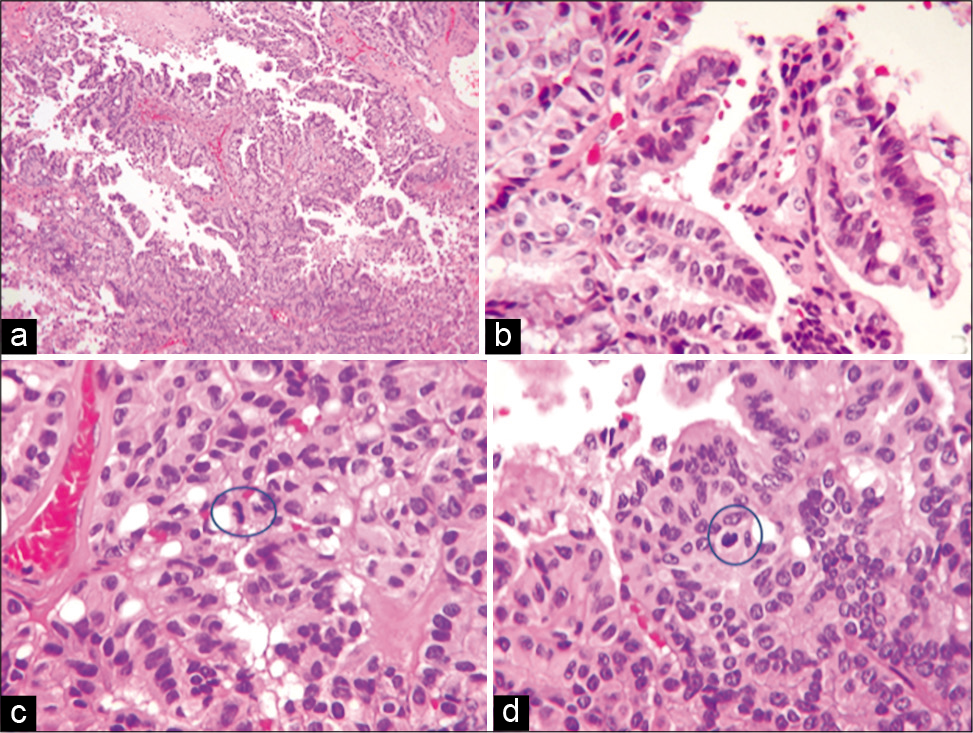
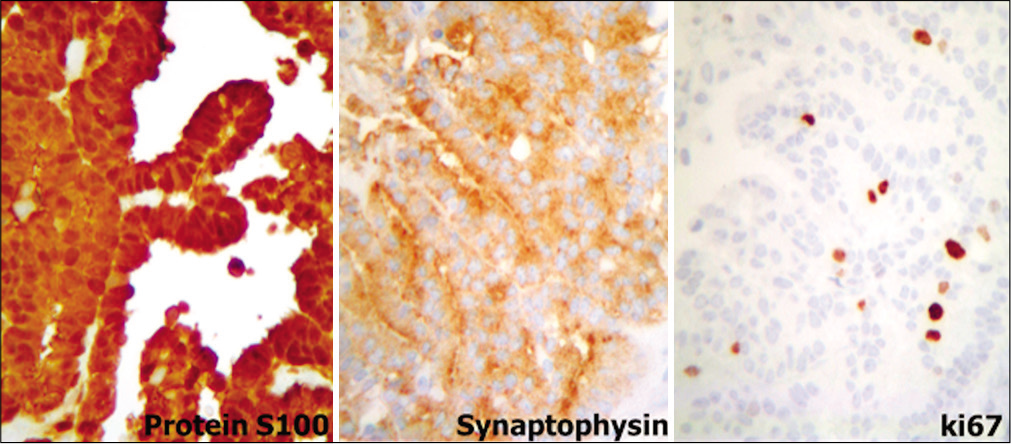
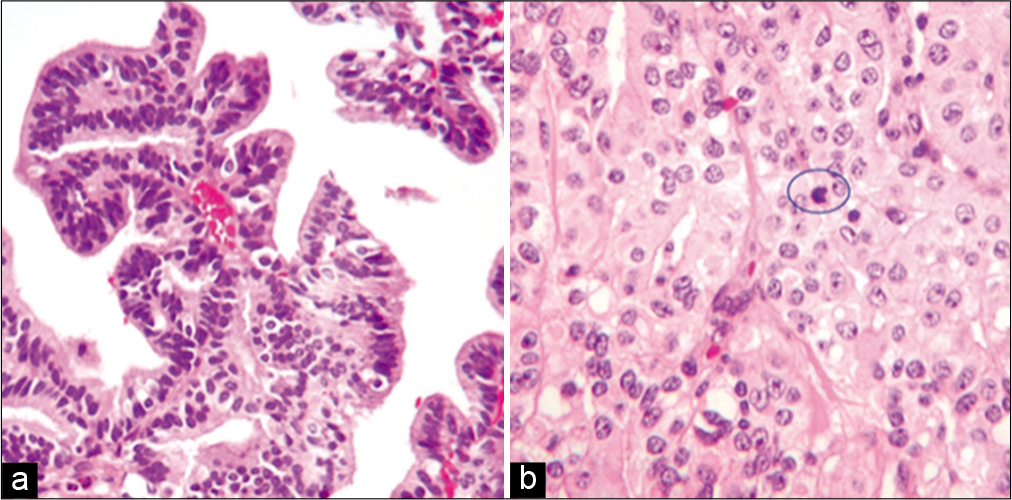
The final pathology report described a neoplasm with papillary architecture, with four mitoses in 10 high-power fields [
Outcome and follow-up
Postoperatively, there was improvement of the radicular pain and an increase in left leg strength, with no changes in left leg dermal sensation. 2 months later, the patient was treated with radiotherapy (RT).
DISCUSSION
Background
CPPs are rare central nervous system tumors that account for <1% of all intracranial neoplasms.[
Clinical features
The clinical presentation of CPP depends on the site of metastasis. The principal clinical manifestation was radicular pain accompanied by paresis of the affected myotome.[
Treatment
The decision to perform surgery on this patient was based on the presentation of neurological deficit and pain. According to the previous reports, the indications for surgical intervention of spinal metastasis are intractable pain; the presence of a growing tumor resistant to RT, chemotherapy, or hormonal therapy; evidence of spinal instability, progressive deformity, or neurological deficit; and clinically significant neural compression.[
Diverse scales have been proposed for evaluating the spinal metastasis treatment; the most popular are the Tokuhashi[
In recent years, minimally invasive surgery (MIS) has become increasingly popular for the treatment of spinal pathology. This approach is associated with lower operative blood loss, diminished narcotic use, shorter postoperative stay, and lower costs of hospitalization compared to open surgery. Although the mortality associated with MIS is 0%, there are other complications including CSF leakage (3%), asymptomatic pseudomeningocele (3%), superficial surgical site infections (2%), sinus vein thrombosis (1%), and neurological deterioration (4%).[
Neurological deterioration is expected in cases of GTR.[
Histology
The world health organization (WHO) classifies ventricular papillary neoplasms into different grades: Grade I CPPs resemble normal papillary tissue, Grade II or atypical tumors are benign CPPs transforming to a malignant type, and Grade III or choroid plexus carcinoma (CPC) is characterized by anaplasia, increased mitotic activity, nuclear pleomorphism, and necrosis.[
Prognostic
For CPPs, GTR is considered to be curative while STR produces good long-term results. Although GTR resulted in excellent tumor control at the 1- and 5-year follow-ups compared to STR, this advantage was lost after 10 years.[
Given the unpredictable behavior of CPPs, even with total resection, patients should be closely monitored during the post-surgical period as spinal implants have been reported 10 years after the primary tumor; in the present case, it was detected 7 years later. It is also important to consider the atypical location of CPP implants, which are very rare at the spinal level. In fact, ours is the first case reported at the lumbar level. CPP cases should be monitored for at least 10 years by routine cranial and spinal imaging, after which imaging should be performed in the event that there are root symptoms or the presence of spinal syndrome.
CONCLUSION
CPPs are extra-axial benign tumors that account for < 1% of intracranial tumors in adults. The presence of spinal metastasis or implants is very rare; only three cases of spinal metastasis have been reported to date. An adequate follow-up of these patients is essential since implants can occur up to 10 years after the initial presentation of CPP.
The clinical manifestation of compression of nerve root or spinal syndrome in patients with a history of CPP precludes spinal metastasis, necessitating a complete study protocol as in the present case.
GTR of primary lesions and associated implants remains the gold standard for surgical treatment of CPP. There are no prophylactic treatments for preventing CPP metastasis. RT, stereotactic radiosurgery, and chemotherapy are adjuvant therapies for CPP but there is no definitive protocol for the management of implants.
Declaration of patient consent
Patient’s consent not required as patients identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Acknowledgments
Primary authorship was the responsibility of D. Ochoa was responsible for all statistical aspects, analysis, editing, and writing. ME. Cordoba was responsible for data analysis, translation, preparation of figures, and verification. The team coordinating consisted of JR. Aguilar, U. García, A. Ibarra, VA. Reyes, JJ. Lomelí, MC. Sánchez, and RM. Vicuña. The group writing was O. Medina, MD. Sánchez, and EA. Castañeda.
References
1. Abdulkader MM, Mansour NH, van Gompel JJ, Bosh GA, Dropcho EJ, Bonnin JM. Disseminated choroid plexus papillomas in adults: A case series and review of the literature. J Clin Neurosci. 2016. 32: 148-54
2. Akil H, Coupe NJ, Singh J. Spinal deposits of a benign choroid plexus papilloma. J Clin Neurosci. 2008. 15: 708-12
3. Babu ML, Gupta R, Kumar A, Gupta M. Spinal intradural metastasis. JK Sci. 2004. 6: 48-50
4. Bahar M, Hashem H, Tekautz T, Worley S, Tang A, de Blank P. Choroid plexus tumors in adult and pediatric populations: The cleveland clinic and University Hospitals experience. J Neurooncol. 2017. 132: 427-32
5. Deng Z, Xu B, Jin J, Zhao J, Xu H. Strategies for management of spinal metastases: A comprehensive review. Cancer Transl Med. 2015. 1: 94-100
6. Halvorsen CM. Minimally invasive microsurgical resection of primary, intradural spinal tumors is feasible and safe: A consecutive series of 83 patients. Neurosurgery. 2017. 1: 1-7
7. Jinhu Y, Jianping D, Jun M, Hui S, Yepeng F. Metastasis of a histologically benign choroid plexus papilloma: Case report and review of the literature. J Neurooncol. 2007. 83: 47-52
8. Morshed RA, Lau D, Sun PP, Ostling LR. Spinal drop metastasis from a benign fourth ventricular choroid plexus papilloma in a pediatric patient: Case report. J Neurosurg Pediatr. 2017. 20: 471-9
9. Safaee M, Clark AJ, Bloch O, Oh MC, Singh A, Auguste KI. Surgical outcomes in choroid plexus papillomas: An institutional experience. J Neurooncol. 2013. 113: 117-25
10. Safaee M, Oh MC, Sughrue ME, Delance AR, Bloch O, Sun M. The relative patient benefit of gross total resection in adult choroid plexus papillomas. J Clin Neurosci. 2013. 20: 808-12
11. Stuivenvolt M, Mandl E, Verheul J, Fleischeuer R, Tijssen CC. Atypical transformation in sacral drop metastasis from posterior fossa choroid plexus papilloma. BMJ Case Rep. 2012. p. 2-5
12. Tokuhashi Y, Matsuzaki H, Oda H, Oshima M. A revised scoring system for preoperative evaluation of metastatic spine tumor prognosis. Spine (Phila Pa 1976). 2005. 30: 2186-91
13. Tomita K, Kawahara N, Kobayashi T. Surgical strategy for spinal metastases. Spine (Phila Pa 1976). 2001. 26: 298-306
14. Wong AP, Lall RR, Dahdaleh NS, Lawton CD, Smith ZA, Wong RH. Comparison of open and minimally invasive surgery for intradural-extramedullary spine tumors. Neurosurg Focus. 2015. 39: E11