- Department of Neuroscience, Winthrop University Hospital, 200 Old Country Road, Suite 485, Mineola, NY, USA
- Department of Radilogy, New York University, Langone Medical Center, New York, USA
Correspondence Address:
Nancy E. Epstein
Department of Radilogy, New York University, Langone Medical Center, New York, USA
DOI:10.4103/2152-7806.159379
Copyright: © 2015 Epstein NE. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.How to cite this article: Epstein NE, Hollingsworth RD, Silvergleid R. Spinal surgeons need to read patients’ studies to avoid missing pathology. Surg Neurol Int 25-Jun-2015;6:
How to cite this URL: Epstein NE, Hollingsworth RD, Silvergleid R. Spinal surgeons need to read patients’ studies to avoid missing pathology. Surg Neurol Int 25-Jun-2015;6:. Available from: http://surgicalneurologyint.com/surgicalint_articles/spinal-surgeons-need-to-read-patients-studies-to-avoid-missing-pathology/
Abstract
Background:Many spine surgeons rely on reports of radiological studies for patients seen routinely in consultation. However, “best practice” should include the spine surgeon's individual assessment of the images themselves to better determine whether the diagnoses rendered were/are correct.
Methods:A now 54-year-old male had an original enhanced magnetic resonance imaging (MR) scan of the cervical spine performed in 2012 that was read as showing mild spondylotic changes at multiple levels.
Results:In 2015, the patient presented with a severe spastic quadriparesis, right greater than left, which had markedly worsened over the prior 3 months. Review of the original enhanced MR from 2012 revealed a right-sided C5–C6 tumor (e.g., likely meningioma) filling the right neural foramen with extension into the spinal canal (7 mm × 8 mm × 11 mm): The tumor was originally “missed”. The new 2015 enhanced MR scan documented the tumor had enlarged 6.7 fold (measuring 17 mm × 11 mm × 2.2 cm), and now filled 2/3 of the spinal canal, markedly compressing the cord and right C6 nerve root. Following a C4–C6 laminectomy, and a challenging tumor removal, and the patient was neurologically intact.
Conclusion:This case underscores the need for spine surgeons to carefully review both images and reports of prior diagnostic studies that accompany patients. In this case, the original failure to recognize the tumor led to a 2.5-year delay in surgery that resulted in the patient's severe preoperative quadriparesis, and a much more challenging surgery.
Keywords: Computed tomography, enhanced magnetic resonance imaging, magnetic resonance imaging, meningioma, radiology, read studies, spinal surgery, surgeon, quadriparesis
EDITORIAL
As spine surgeons, we see a lot of patients who come in with prior diagnostic studies. We typically read the outside images ourselves in conjunction with the radiological reports. This “best practice” helps us determine whether or not a surgical lesion is present, and enables us to document the significance, location, side, and size of the problem. Here, we present a patient whose surgery was delayed by 2.5-years due to the original neuroradiologist's failure to diagnose a right-sided C5–C6 meningioma on an enhanced magnetic resonance imaging (MR) scan.
Case study
A 54-year-old male presented with a progressive spastic quadriparesis, right greater than left that had markedly exacerbated over a 3-month duration. On examination, he exhibited severe motor deficits (right upper extremity [2–3/5], left upper extremity [3–4/5], and bilateral lower extremities [3–4/5 right]), accompanied by diffusely 3-4+ hyperactive reflexes with bilateral Babinski signs. Bilateral, and bilateral sensory loss from C6 downward was additionally associated with decreased vibration and position appreciation in the right greater than the left lower extremities.
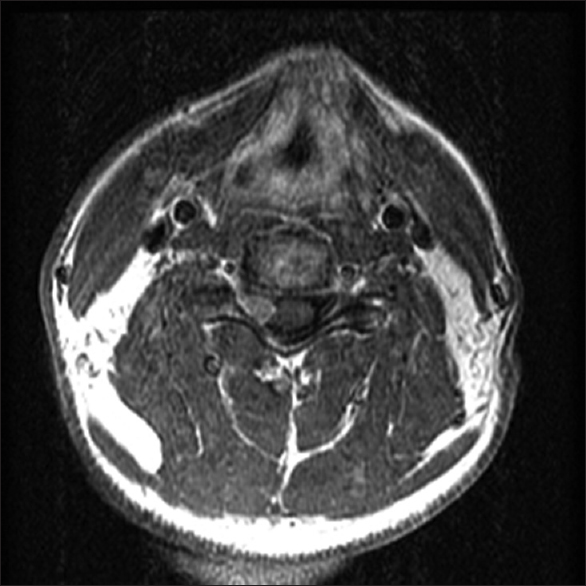
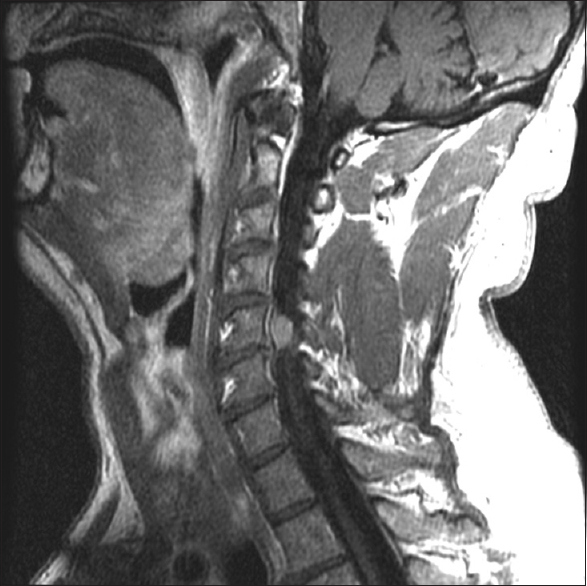
Original 2012 enhanced T1 magnetic resonance imaging: missed right C5–C6 intradural extramedullary tumor
The original report of the enhanced MR from 2012 read; small central/right paracentral disc herniation at C4–C5, and moderate size broad based bony ridge at the C5–C6 level causing mild cord compression/moderate bilateral foraminal stenosis. Review of the original enhanced T1 MR study clearly revealed a right-sided uniformly enhancing intradural extramedullary tumor extending from the C5-C6 neural foramen, into the neural foramen and into the spinal canal (7 mm × 8 mm × 11 mm) contributing to marked C6 root and mild/moderate cord compression [Figures
Figure 1
The original October 2012 axial T1 enhanced magnetic resonance imaging (MR) showing smaller right C5–C6. On the original October 2012 axial T1 enhanced MR study. The right-sided C5-6 intradural extramedullary tumor was clearly visualized extending into the right neural foramen where it markedly compressed the right C6 root and the spinal cord
Figure 2
The original sagittal T1 enhanced magnetic resonance imaging (MR) from 10/2012 showing smaller right C5–C6 meningioma study. The right-sided C5–C6 intradural extramedullary tumor was clearly visualized extending into the right neural foramen. It already markedly compressed the right C6 root and there was mild/moderate cord compression
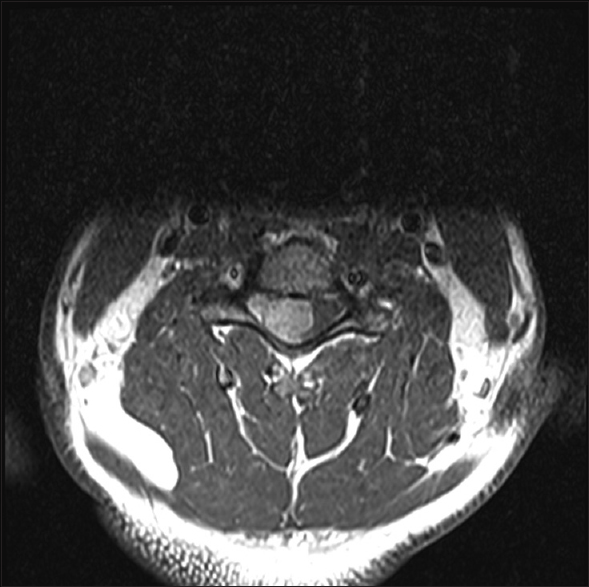
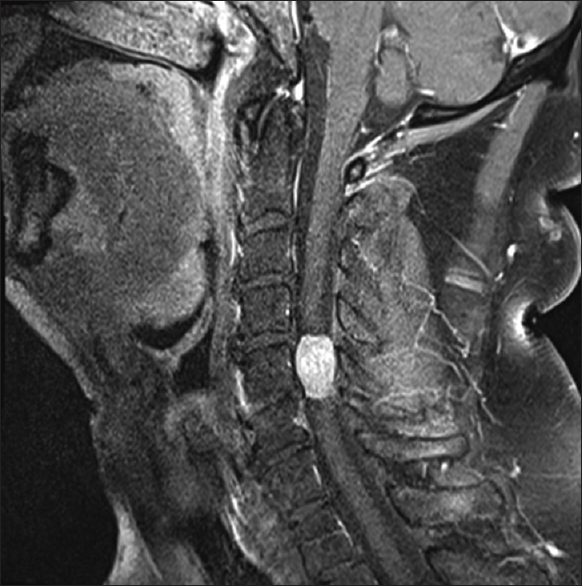
2015 enhanced T1 magnetic resonance imaging showed massive right C4–C6 intradural extramedullary tumor
The subsequent enhanced T1 MR from 2015 showed a 6.7-fold larger intradural/extramedullary tumor (17 mm × 11 mm × 2.2 cm) that now filled 2/3 of the spinal canal, markedly compressing the cord to the left [Figures
Figure 3
Axial T1 enhanced magnetic resonance imaging from 2015 showing a much larger right C4–C6 meningioma filling 2/3 of the spinal. On the axial T1 postcontrast study of March 2015. The tumore now had substantially gorwn in size; measuring 11 mm × 17 mm in diameter. It not only filled the C5–C6 foramen but also occupied 2/3 of the spinal canal. This resulted in marked cord compression, with the cord displaced now markedly toward the left side of the spinal canal
Figure 4
Sagittal T1 enhanced magnetic resonance imaging from 2015 showing much larger right C4–C6 meningioma filling 2/3 of the spinal. On the sagittal T1 postcontrast study of March 2015. The tumor now had substantially grown in size; measuring in addition to 11 mm × 17 mm in diameter, 2.2 cm in cephalad/caudad dimension. On this midline sagittal image, it filled the spinal canal at the C6 level (e.g., note the spinal cord is fully compressed toward the left) and resulted in marked spinal cord compression
No intraoperative changes in poor baseline somatosensory evoked potentials/motor evoked potentials
Poor baseline preoperative and intraoperative somatosensory evoked potentials and motor evoked potentials (MEPs) (no electromyographic changes) did not decline or drop out during surgery. Notably, baseline data were also obtained, and maintained throughout awake intubation/awake positioning, and surgery. In addition, we specifically avoided using any muscle relaxants during the procedure to avoid losing the ability to continually monitor the tenuous MEPs.
Use of steroids
Of interest, the patient had been on decadron 4 mg orally every 6 h for a week prior to surgery (our prescription), and was prophylactically given 1 g of methylprednisolone immediately preoperatively.
Surgery: Laminectomy C4–C6 for removal of meningioma
A C4–C6 laminectomy was performed. This involved coming up the lateral gutters with a 4 mm diamond burr in combination with a 1 mm Kerrison punch. Once the midline dura was opened, centered over the C5 level, tumor filled the right side of the spinal canal, and extended from the inferior aspect of C4 to the superior aspect of C6. Tumor clearly crossed the midline, and markedly compressed the spinal cord toward the left. Utilizing microscope visualization, micro instrumentation, and the ultrasonic surgical aspirator, the tumor was “decompressed” from the “inside-out”. The dura was closed with interrupted 7-0 Gortex (Gore-Creative Technologies Worldwide, Newark, Delaware, USA) sutures (every 3–4 mm) and micro dural staples (medium 1.4 mm) placed between the sutures. A thin layer of Tisseel (Baxter, Deerfield, IL, USA), followed by Duragen (Integra LifeSciences, Plainsboro, NJ, USA) were applied. Postoperatively, the patient was neurologically normal, and went home within less than 48 h. The final pathology was benign meningioma.
The point
The point of this editorial is to highlight the need for spine surgeons to read patients’ outside diagnostic studies, and not just rely on the radiological/neuro-radiological reports. Had surgery been performed in 2012, when the right-sided C5–C6 tumor clearly visible on the original enhanced T1 MR was substantially smaller, the patient would never had developed a severe quariparesis, and the surgery would have been more straightforward. By 2015, with a second enhanced MR showing a tumor now 6.7 fold larger, filling 2/3 of the spinal canal from C4–C6, the patient had to undergo a much more challenging procedure.
Commentary
Commentaries
COMMENTS FROM THE EDITORIAL BOARD OF SURGICAL NEUROLOGY INTERNATIONAL SPINE SUPPLEMENT
I am in full agreement with this editorial. I feel it is absolutely imperative the spine surgeon personally review the films when seeing the patient. I like to use the technique of reviewing the studies myself, then reviewing the radiologists’ reading of the same study. I think this gives the patient an “extra read” as part of the evaluation process and also provides me with a check and balance system. I think we, as treating surgeons, need to be aware of that the skill levels vary among radiologists. Often time I see studies that are read by a “generalist.” The specifics of that study may be substantially different from that of a specialty trained neuroradiologist's reading.
I feel this is a well written case report which effectively highlights an important aspect of being a careful clinician. Neurosurgeons should independently interpret their radiological studies in conjunction with the radiologists report. Likewise, sometimes the report calls something to the attention of the surgeon that he/she may have missed that can change their management.
My first questions in this case are who ordered the first scan, and why? The next question: Was the radiologist's interpretation consistent with the careful history and physical exam taken before the scan was ordered? Whether or not the surgeon has a long historical practice of deferring to the radiologist, he or she nevertheless should be satisfied with the answers to these questions. But, finally I agree with the underlying premise of this case: if one is going to assume the responsibility for the surgical outcome in the case, that surgeon, to be qualified, must be able to read these films. This would be an interesting case for residents.
This case report reminds us of the importance of reviewing studies ourselves. While it is easy to overlook this simple rule, this patient's experience illustrates how critical it is to be compulsive in these matters.
There is no question that every spine surgeon should review the spinal imaging studies on each patient and not rely on the radiologist's report. The best practice is to review the films prior to reading the radiology report. Once the surgeon has finished assessing the images then the next important step is to review the radiologist's report. A radiologist may overlook an important detail, and so might a spine surgeon. This is the optimal way to avoid such an event. Finally, there will be times when the images are inadequate. If images are degraded by motion, do not have axial views over the pertinent regions, lack contrast when indicated, or suffer from another problem, then the studies may need to be repeated. At times, the decision to repeat a study can only be made following personal review and correlation with the patient's clinical presentation.
The obligation of the surgeon to study all the diagnostic studies should be considered part of the standard care. If there is any question, the surgeon needs to review the imaging studies personally with the radiologist. Our practice was always “four eyes are better than two” – in fact, four is twice as good. Doing medical reviews and precertifications, it is more common for the surgeon to be resistant to joint reviews. When there are discrepancies, a new report needs to dictated. Then, a clinical opinion is more likely to be honored.
Dr. Epstein's brief report is important and well presented. It highlights an often neglected aspect of NS practice.
I certainly agree that surgeons should evaluate all diagnostic tests relating to their patients, especially radiographic studies. I thought this was obvious, and was part of everyone's training. The surgery described in this scenario certainly would have been less technically challenging if the tumor had been discovered earlier. This case report could be enhanced by providing evidence that the patient was in fact seen by a spine surgeon, who did not read the films him/herself, or failed to adequately diagnose the lesion. Looking at this, it is not clear how the original tumor was missed.
The general principle that in a multi-specialties hospital, the physician is the final legal person responsible for the therapy he/she suggests and performs, is particularly consistent for brain and spine surgeons. Among recent examples of missed diagnosis both in brain and spine surgeries,[
Although the spine surgeon appears to be so strongly conditioned and dependent on neuro-radiological and neuro-physiological patient's assessments, the global final success of the surgical strategy adopted is up to his special global and final intuition: Diagnostic and surgical. In other words, it means that the doubt is the scientific method to be always adopted by the surgeon, who is the leader of the physician team, mainly in such challenging circumstances. Although a missed cervical spine injury can have quite immediate devastating consequences, fortunately in the present case, following a challenging tumor removal, the patient was neurologically intact and permanently cured due to the benign nature of the lesion.
The take home message of this experience is that the surgeon should always discuss the neuro-radiological examination with a radiologist different from the one who has performed the exam and finally critically review the diagnostic conclusions.
I’m personally underwhelmed with the overall significance of this report. I perhaps incorrectly assumed that every spine surgeon looks at and interprets his/her own imaging studies, whether he/she orders them him/herself or not. As opposed to “best practice,” I have always assumed this was standard and basic practice. Based on this report, I think a much better, more informative, and interesting article would be a survey of practicing spine surgeons to determine how many actually base their reports, opinions, and judgments on radiology reports rather than their own personal review. I would have thought the answer would be none or next to none.
I didn’t even see anything that actually says what happened during the first encounter in 2012. Who actually saw this patient? And what was or was not done during that encounter? Was it the same doctor both in 2012 and again in 2015? Was there a reason the doctor in 2012 did not either see or think he/she did not need to see the actual studies in 2012? All of these sort of essential specifics of the first encounter are completely missing from the report/abstract/report.
This report documented a benign meningioma that was not reported on an adequate imaging study 3 years earlier. Consequently, it resulted in progression of the tumor growth, and the tumor remained untreated leading to further diminished neurological function.[
References
1. Dho YS, Kim SK, Wang KC, Phi JH. Catastrophic intramedullary abscess caused by a missed congenital dermal sinus. J Korean Neurosurg Soc. 2015. 57: 225-8
2. Ramgren B, Siemund R, Nilsson OG, Höglund P, Larsson EM, Abul-Kasim K. CT angiography in non-traumatic subarachnoid hemorrhage: The importance of arterial attenuation for the detection of intracranial aneurysms. Acta Radiol. 2014. p.
3. Resnick S, Inaba K, Karamanos E, Pham M, Byerly S, Talving P. Clinical relevance of magnetic resonance imaging in cervical spine clearance: A prospective study. JAMA Surg. 2014. 149: 934-9






Suresh Ramnath
Posted July 30, 2015, 7:38 pm
I am surprised that there was a need to conduct a survey about reading films (especially pre-operatively). I imagined (naively) that a surgeon obtained a history, examinedthe poatient, and then looked at the studies to see if they matched the problem,rather than the other way around. We do not seem to have enough time for that and instead rely overly on what we are told.