- Department of Neurosurgery, All India Institute of Medical Sciences, Patna, Bihar, India.
Correspondence Address:
Vikas Chandra Jha, Department of Neurosurgery, All India Institute of Medical Sciences, Patna, Bihar, India.
DOI:10.25259/SNI_805_2023
Copyright: © 2024 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Vikas Chandra Jha, Rahul Jain, Vivek Saran Sinha, Nitish Kumar, Gaurav Verma, Nikhil Dhage. Spontaneous thrombosis and calcification of giant cavernous carotid artery aneurysm: A rare case and management insights. 22-Mar-2024;15:98
How to cite this URL: Vikas Chandra Jha, Rahul Jain, Vivek Saran Sinha, Nitish Kumar, Gaurav Verma, Nikhil Dhage. Spontaneous thrombosis and calcification of giant cavernous carotid artery aneurysm: A rare case and management insights. 22-Mar-2024;15:98. Available from: https://surgicalneurologyint.com/surgicalint-articles/12816/
Abstract
Background: Giant cavernous carotid artery aneurysms (>25 mm) are rare (3–5%), with some prone to spontaneous thrombosis (10–20% complete). We present a unique case of one of the largest aneurysms spontaneously thrombosing and calcifying.
Case Description: A 57-year-old with persistent right-sided headaches had a substantial hyperdense mass in the right middle cranial fossa, eroding petrous bone. Magnetic resonance imaging and digital subtraction angiography revealed a giant cavernous segment fusiform aneurysm of the right internal carotid artery (ICA) with spontaneous thrombosis and distal ICA occlusion. Collateral circulation maintains the cerebral blood supply. Despite anatomical challenges, conservative management was chosen due to the patient’s stability.
Conclusion: This case highlights the complex interplay between thrombosed giant aneurysms and affected vessels, with unique features such as cross-flow, calcification, and bone erosion. We advocate conservative management for stable cases, supported by literature, emphasizing vigilant follow-up. This expands the spectrum of aneurysm presentations and encourages further research into their dynamics.
Keywords: Aneurysm calcification, Follow-up monitoring, Giant cavernous carotid artery aneurysms, Spontaneous thrombosis
INTRODUCTION
Giant cavernous carotid artery aneurysms are defined as those exceeding a size of 25 mm. According to existing literature, the incidence of cavernous carotid aneurysms is estimated to be in the range of 3–5%.[
Although spontaneous thrombosis of the parent artery is a rare occurrence, it poses a potentially catastrophic complication for these patients. In this current article, we present a unique case involving one of the largest aneurysms known to undergo spontaneous thrombosis and subsequent calcification. We provide a detailed clinical analysis of this case and conduct a comprehensive literature review of similar cases reported over the years. Furthermore, we explore potential management options in light of the limited existing literature on this subject.
CASE DETAIL
A 57-year-old male presented to our department, complaining of persistent right-sided headaches lasting two years. Physical examination revealed that the right temporalis muscles were less prominent, which may be an incidental finding in this patient. A prior non-contrast computed tomography scan showed a hyperdense mass in the right middle cranial fossa, eroding the right petrous bone [
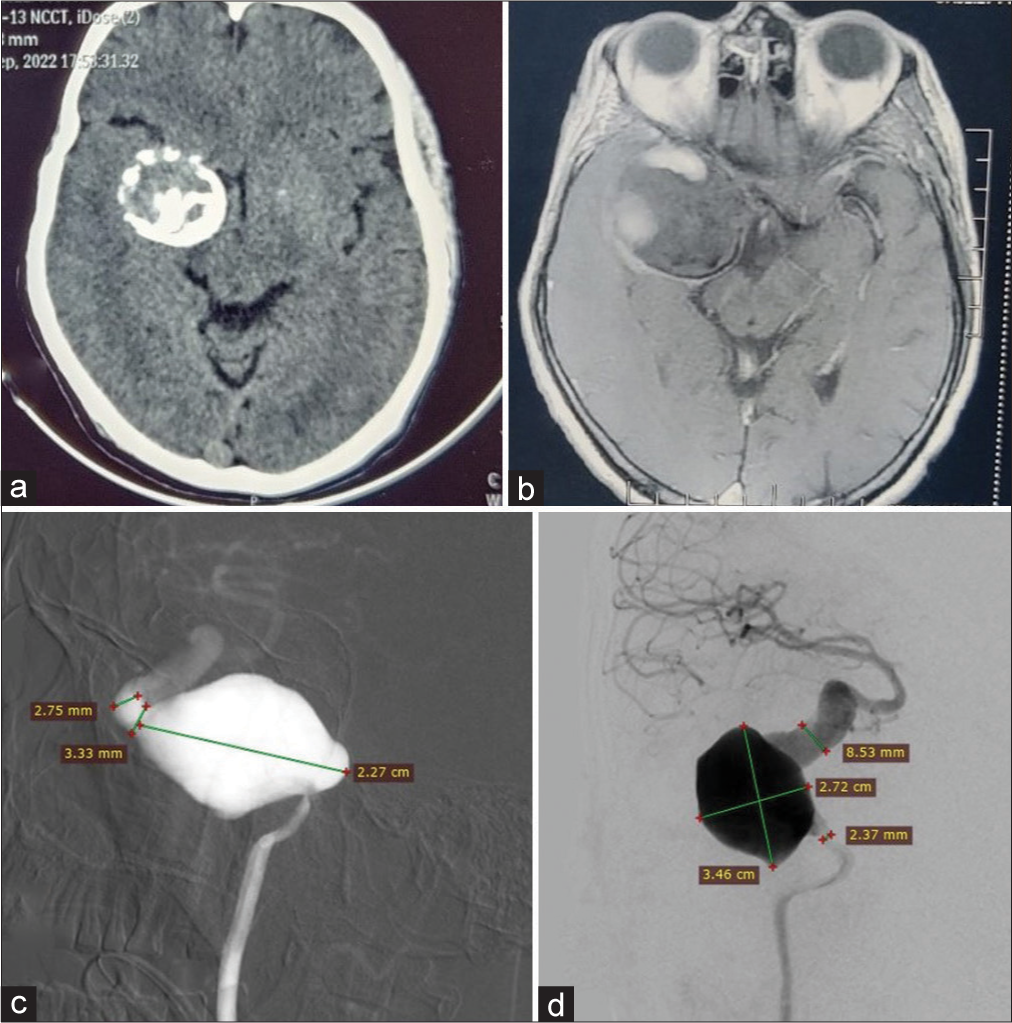
Figure 1:
(a) Axial non-contrast computed tomography head demonstrates a round, heterogeneously hyperdense mass lesion in the right middle cranial fossa with calcifications in both the center and its periphery. (b) Axial gadolinium-contrast magnetic resonance imaging brain reveals a well-defined, round lesion that is iso- or even hypo-intense and is partially enhanced by intravenous gadolinium, and it is extra-axial mass along the right medial temporal convexity, closely adjacent to the right sphenoid wing. (c) Cerebral digital subtraction angiography (DSA) – Lateral view illustrates a fusiform giant aneurysm in the cavernous segment of the right internal carotid artery (ICA); the aneurysm seems to originate in the cavernous segment and likely terminates at the transition of cavernous to ophthalmic segment with measurements shown in the image. In addition, it displays the dilated and tortuous supra clinoid portion of the right ICA. (d) Cerebral DSA – Anteroposterior view shows the same fusiform giant aneurysm in the cavernous segment of the right ICA with measurements. It also highlights the dilated and tortuous supra clinoid portion of the right ICA, which is compared to the lumen size of the proximal ICA. Furthermore, it points out the hypoplastic right A1 anterior cerebral artery and normal right middle cerebral artery supply.
A contrast-enhanced magnetic resonance imaging displayed a hypointense lesion on T2 and fluid-attenuated inversion recovery images with subtle T1 hyperintensity. The mass measured 5 × 4.5 cm, exerting mass effect on adjacent structures and showing susceptibility-weighted imaging (SWI) blooming, suggesting blood products or calcifications. Post-contrast imaging hinted at a large calcified aneurysm or calcified meningioma [
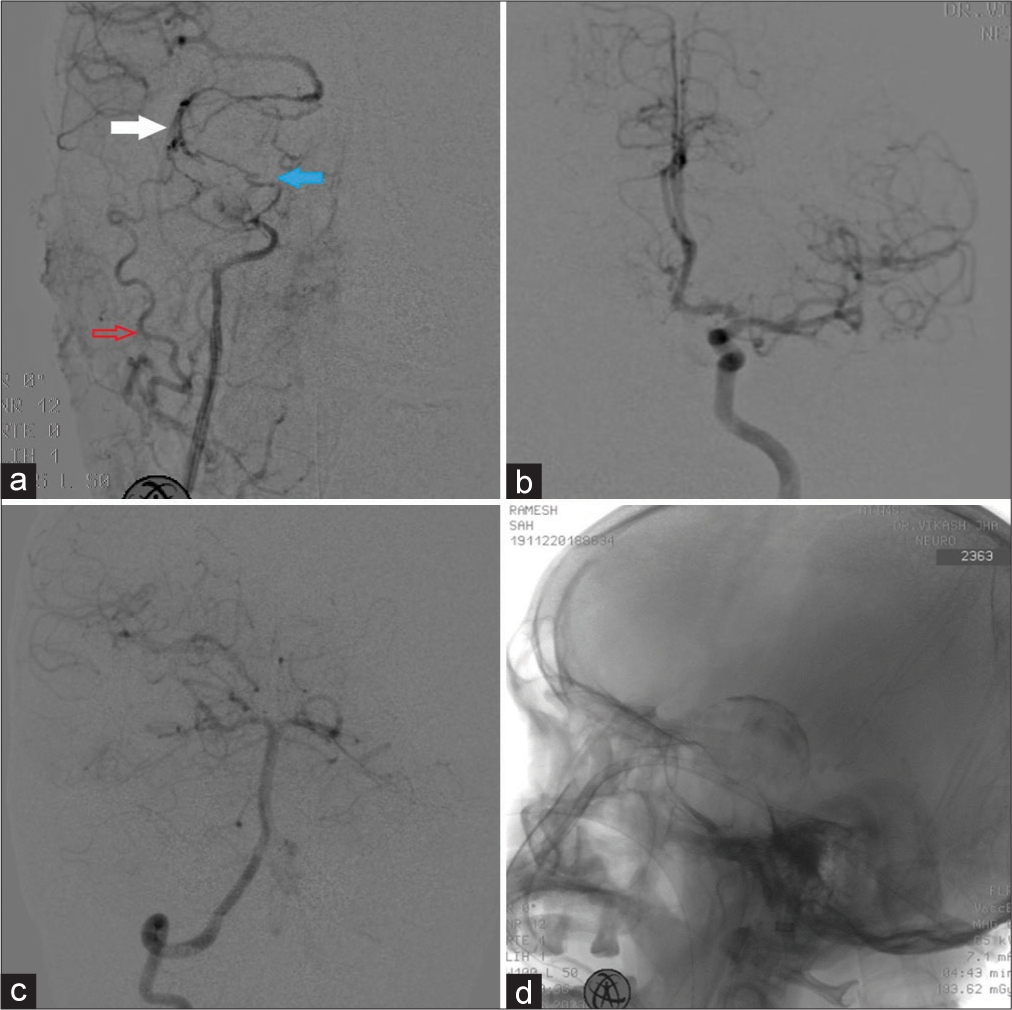
In May 2023, the patient returned for further evaluation and cerebral DSA. This showed complete spontaneous thrombosis of the cavernous ICA aneurysm, occluding the distal ICA. Collateral circulation from extracranial sources, contralateral circulation, and posterior circulation ensured sufficient supply to the right cerebral hemisphere [
Figure 2:
(a) Intraoperative right common carotid artery injection – anteroposterior view displays the proximal right internal carotid artery (ICA) and peripheral enhancement of vasa vasorum within the spontaneously thrombosed aneurysm (blue solid arrow). It also demonstrates the obliteration of the ICA distal to the aneurysm (white solid arrow) and collateral circulation provided by a branch of the right external carotid artery (red arrow) to the distal right ICA and subsequently to the right middle cerebral artery (MCA). (b) Intraoperative left ICA injection – anteroposterior view shows the filling of bilateral A2 ACA from the left ACA. Cross-compression demonstrates no cross-flow to the right A1 ACA. (c) Right vertebral artery injection, AP view, demonstrates collateral flow into the right MCA from the right posterior communicating artery. (d) Fluoroscopic image of the skull–lateral view reveals a relatively well-defined mass at the base of the skull with a clear superior margin, situated superior to the sphenoid wing. Peripheral radiopaque margins may indicate calcification. There is no evidence of sella widening, and the sphenoid sinus appears normal. ACA: Anterior cerebral artery
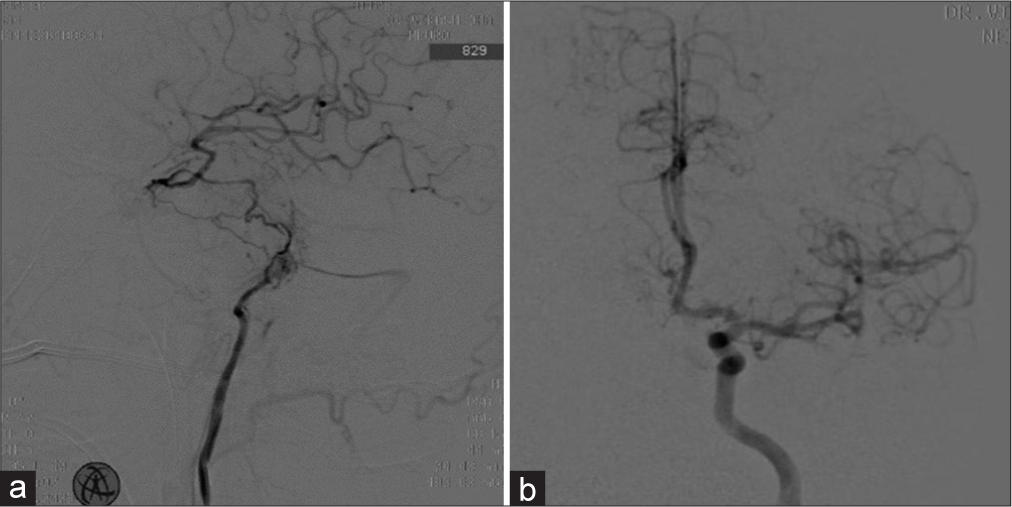
Despite the challenging decision not to proceed with invasive procedures such as superficial temporal artery and middle cerebral artery anastomosis on the right side, the patient remained asymptomatic at the 12-month follow-up, with stable thrombosed and calcified aneurysm [
Figure 3:
(a) Follow-up cerebral digital subtraction angiography – Right internal carotid artery (ICA) injection – lateral view depicts the truncated proximal ICA with minimal contrast blush within the proximal portion of the thrombosed aneurysm lumen with filling of distal ICA by right posterior communicating artery. (b) Follow-up left ICA injection – AP view shows the filling of bilateral A2 ACA from the left ACA, with no cross flow to the right A1 ACA observed during cross-compression. AP: Antero=posterior, ACA: Anterior cerebral artery
DISCUSSION
This case report delves into a remarkable instance involving a giant cavernous segment fusiform aneurysm of the right ICA.[
It is important to note that giant aneurysms commonly experience partial thrombosis, with a frequency of 60%. However, complete spontaneous thrombosis is a rare occurrence and only happens in 20% of cases.[
This case posed a considerable technical challenge due to a substantial disparity between the proximal and distal segments of the right ICA. The deployment of a flow diverter could have compromised distal flow from the right PCom artery and resulted in improper placement of the device. Distal artery bypass surgery, such as the superficial temporal artery to MCA (STA-MCA), could have augmented collateral flow. However, the patient remained neurologically stable, with a thrombosed right cavernous artery and collaterals from the anterior circulation through the anterior communicating artery to bilateral A2 segments and right distal ICA supplied by the right PCom artery. This led us to opt for conservative treatment.
The mechanisms underlying vessel obliteration alongside spontaneous luminal thrombosis in giant aneurysms are not fully understood.[
Beyond thrombosis, the journey of intracranial aneurysms continues.[
While acute vessel occlusion by a thrombosed aneurysm can lead to neurological deficits and ischemic changes, especially in the presence of adequate collateral flow, our case, along with 18 others from the literature, did not exhibit clinical ischemic manifestations despite complete ICA thrombosis.[
In the majority of the series reported to date, patients improve at the 6-month mark or remain stable if not deteriorated.[
Through this case report, we aim to provide insights and valuable information regarding this rare phenomenon. The presence of unique features, such as cross-flow, calcification, and petrous bone erosion with intracranial and extracranial collaterals to supplement distal ICA flow in one of the largest giant calcified aneurysms reported to date, suggests a chronic nature of the disease not previously documented. We advocate for conservative management in cases with stable hemodynamics, as supported by existing literature, and emphasize the importance of close follow-up for the early detection of disease progression.[
CONCLUSION
This case report contributes valuable insights into a rare phenomenon, offering a comprehensive understanding of the intricate interplay between thrombosed giant aneurysms and the vessels they affect. The presence of unique features, such as cross-flow, calcification, and petrous bone erosion, adds further depth to our understanding of this complex condition. We advocate for conservative management in cases with stable hemodynamics, supported by existing literature, and emphasize the need for vigilant follow-up to detect any signs of disease progression. This case adds to the myriad presentations of this entity and opens doors for further research and exploration of the intricate dynamics involved in thrombosed aneurysms.
Ethical approval
Institutional review board approval is not required.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Use of Artificial Intelligence (AI)-Assisted Technology for manuscript preparation
The authors confirm that there was no use of Artificial Intelligence (AI)-Assisted Technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
Disclaimer
The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Journal or its management. The information contained in this article should not be considered to be medical advice; patients should consult their own physicians for advice as to their specific medical needs.
References
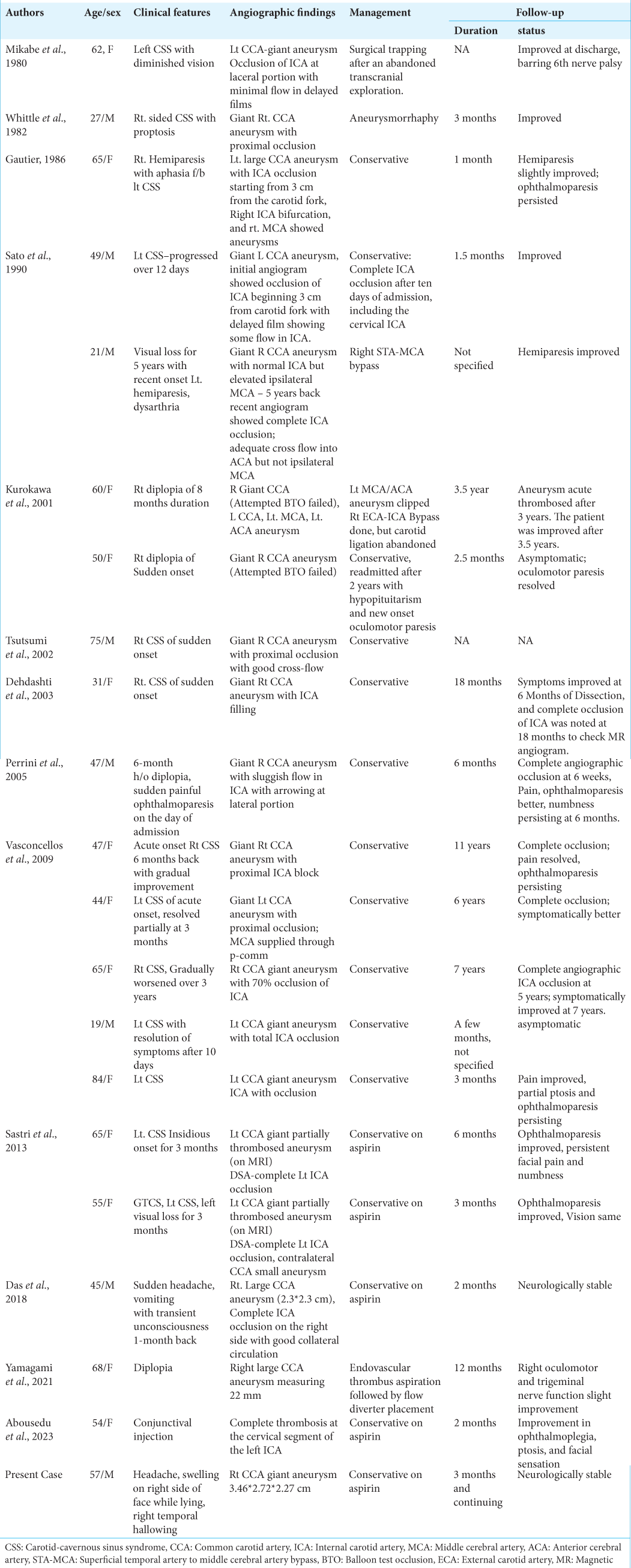
1. Abousedu YA, Saleem A, Alenezi S, Bosnjakovic P, Lazovic L, Alsheikh TM. Spontaneous thrombosis of a giant cavernous-carotid aneurysm with simultaneous ipsilateral complete parent artery occlusion: A rare phenomenon and review of the literature. Arch Clin Cases. 2023. 10: 21-8
2. Das KK, Singh G, Pandey S, Bhaisora KS, Jaiswal A, Behari S. Completely thrombosed giant intracranial aneurysm with spontaneous thrombosis of the parent artery: Is it nature’s divine intervention and a self-cure?. World Neurosurg. 2018. 118: 132-8
3. Dehdashti AR, de Tribolet N. Giant intracavernous aneurysm thrombosis by spontaneous carotid occlusion. Cerebrovasc Dis. 2003. 15: 301-2
4. Gautier JC, Awada A, Majdalani A. Ophthalmoplegia with contralateral hemiplegia. Occlusion of the internal carotid artery due to thrombosis of an intracavernous aneurysm. Stroke. 1986. 17: 1321-2
5. Hahn CD, Nicolle DA, Lownie SP, Drake CG. Giant cavernous carotid aneurysms: Clinical presentation in fifty-seven cases. J Neuroophthalmol. 2000. 20: 253-8
6. Kurokawa R, Kuroshima Y, Yoshida K, Kawase T. Spontaneous thrombosis of intracavernous internal carotid artery aneurysm and parent artery occlusion in patients with positive balloon test occlusion: Two case reports. Neurol Med Chir (Tokyo). 2001. 41: 436-41
7. Menon S, Menon RG. Cavernous carotid aneurysms: To do or not to do?. J Neurosci Rural Pract. 2017. 8: 284-7
8. Mikabe T, Ogihara R, Tomita S, Kin H, Karasawa H, Watanabe S. Giant intracranial aneurysm visualized by prolonged injection angiography-case report (author’s transl). No Shinkei Geka. 1980. 8: 749-53
9. Perrini P, Bortolotti C, Wang H, Fraser K, Lanzino G. Thrombosed giant intracavernous aneurysm with subsequent spontaneous ipsilateral carotid artery occlusion. Acta Neurochir (Wien). 2005. 147: 215-7
10. Sastri SB, Sadasiva N, Pandey P. Giant cavernous carotid aneurysm with spontaneous ipsilateral ICA occlusion: Report of 2 cases and review of literature. J Neurosci Rural Pract. 2013. 4: S113-6
11. Sato K, Fujiwara S, Yoshimoto T, Onuma T. Two cases of spontaneous internal carotid artery occlusion due to giant intracranial carotid artery aneurysm. Stroke. 1990. 21: 1506-9
12. Tsutsumi M, Kazekawa K, Tanaka A, Ueno Y, Nomoto Y. Spontaneous thrombosis of a giant intracavernous internal carotid artery aneurysm and ipsilateral internal carotid artery occlusion. Radiat Med. 2002. 20: 261-3
13. Vasconcellos LP, Flores JA, Conti ML, Veiga JC, Lancellotti CL. Spontaneous thrombosis of internal carotid artery: A natural history of giant carotid cavernous aneurysms. Arq Neuropsiquiatr. 2009. 67: 278-83
14. Whittle IR, Williams DB, Halmagyi GM, Besser M. Spontaneous thrombosis of a giant intracranial aneurysm and ipsilateral internal carotid artery. Case report. J Neurosurg. 1982. 56: 287-9
15. Yamagami K, Hatano T, Ando M, Chihara H, Ogura T, Suzuki K. Symptomatic cavernous internal carotid artery aneurysm complicated by simultaneous rapid growth of the intra-aneurysmal and parent artery thromboses. NMC Case Rep J. 2021. 8: 177-82