- Department of Neurosurgery, Kobe City Nishi-Kobe Medical Center, Kobe, Japan
- Department of Neurosurgery, Graduate School of Medicine, Kobe University, Kobe, Japan
- Department of Neurosurgery, Jyunshin Hospital, Kakogawa, Japan
- Department of Neurosurgery, Myodani Hospital, Kobe, Japan.
Correspondence Address:
Noriaki Ashida, Department of Neurosurgery, Kobe City Nishi-Kobe Medical Center, Kobe, Japan.
DOI:10.25259/SNI_44_2024
Copyright: © 2024 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Noriaki Ashida1, Atsushi Fujita2, Hideya Hayashi1, Masashi Higashino3, Yusuke Ikeuchi2, Hirofumi Iwahashi1, Masamitsu Nishihara1, Kohkichi Hosoda4, Takashi Sasayama2. Successful shrinkage of a recurrent partially thrombosed symptomatic large basilar tip aneurysm using a Target 3D Coil. 22-Mar-2024;15:103
How to cite this URL: Noriaki Ashida1, Atsushi Fujita2, Hideya Hayashi1, Masashi Higashino3, Yusuke Ikeuchi2, Hirofumi Iwahashi1, Masamitsu Nishihara1, Kohkichi Hosoda4, Takashi Sasayama2. Successful shrinkage of a recurrent partially thrombosed symptomatic large basilar tip aneurysm using a Target 3D Coil. 22-Mar-2024;15:103. Available from: https://surgicalneurologyint.com/surgicalint-articles/12809/
Abstract
Background: Standalone coil embolization is often less effective for partially thrombosed intracerebral aneurysms (PTIA) because of the risk of frequent recurrence if the coil migrates into the thrombus. This report describes a case of PTIA at the basilar tip in which simple coil embolization using a Target 3D Coil resulted in sustained remission without recurrence during long-term follow-up.
Case Description: The patient was a 63-year-old male who presented with right oculomotor nerve palsy after having undergone direct surgery for a basilar artery aneurysm 15 years earlier. Recurrence with partial thrombosis of the basilar artery aneurysm was diagnosed. Target 3D Coil embolization with frame construction in the aneurysmal sac was performed, resulting in the complete disappearance of the aneurysm and improvement of the oculomotor nerve palsy. Magnetic resonance imaging at five years postoperatively confirmed that the thrombus had completely disappeared, and there was no recurrence of the aneurysm. The closed loops in the Target 3D Coil may have contributed to the cohesive mass of coils remaining in the sac of the PTIA, potentially leading to healing.
Conclusion: The characteristics of the Target 3D Coil may have prevented migration of the coil into the thrombus, potentially contributing to the successful resolution of the aneurysm.
Keywords: Coil embolization, Partially thrombosed intracranial aneurysm, Target 3D Coil
INTRODUCTION
Partially thrombosed intracerebral aneurysm (PTIA) at the basilar tip is a progressive condition that is often associated with neurological symptoms because of mass effect.[
CASE PRESENTATION
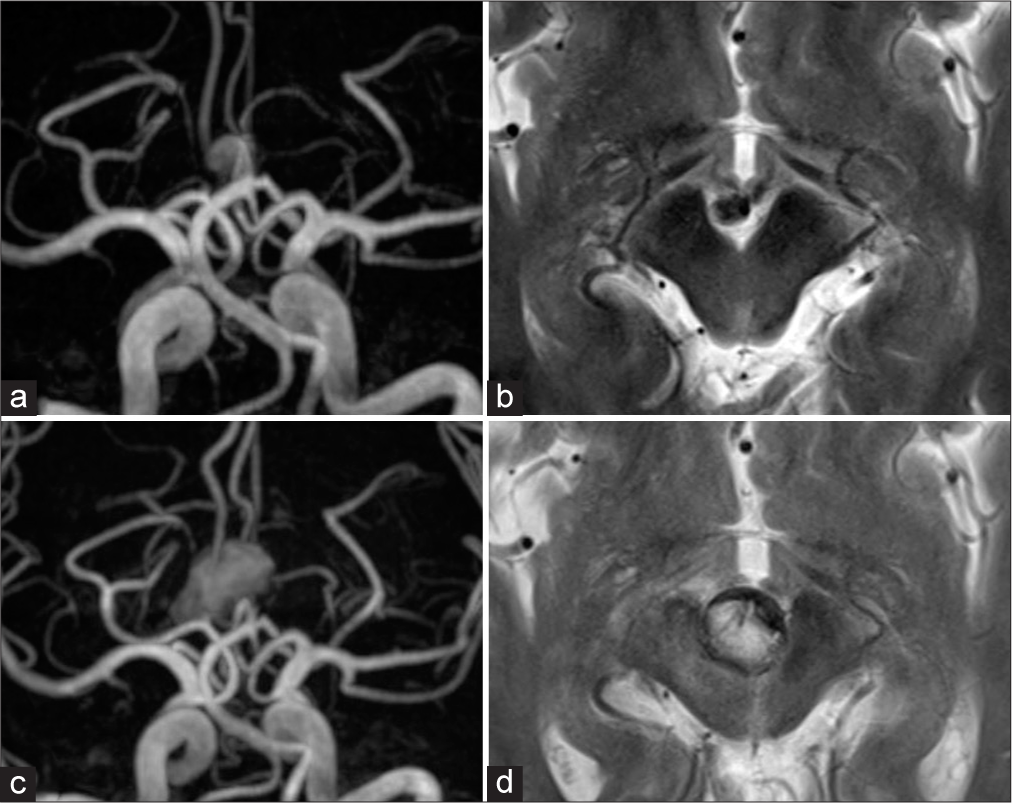
A 48-year-old male underwent open surgical wrapping of an unruptured intracranial aneurysm at the basilar tip in 2003. Ten years later, final postoperative follow-up magnetic resonance angiography (MRA) and T2-weighted imaging (T2WI) of the head revealed blood flow within the aneurysmal sac, but no evidence of enlargement [
Figure 1:
Radiological findings after the initial treatment (a and b) and at the time of recurrence (c and d). (a) MRA image demonstrating blood flow in the aneurysm at the basilar tip. (b) T2WI shows a flow void in the aneurysm, but no thrombus is observed. (c) MRA image showing enlargement of both blood flow in the aneurysm and the surrounding thrombus. (d) T2WI illustrates midbrain compression as a result of the thrombus in the basilar tip. MRA: Magnetic resonance angiography, T2WI: T2-weighted image.
Cerebral angiography demonstrated an inflow of contrast agent into the neck portion of the PTIA (presumed to be the aneurysmal sac) measuring 3.4 mm × 3.0 mm and extending from the basilar tip to the left posterior cerebral artery [
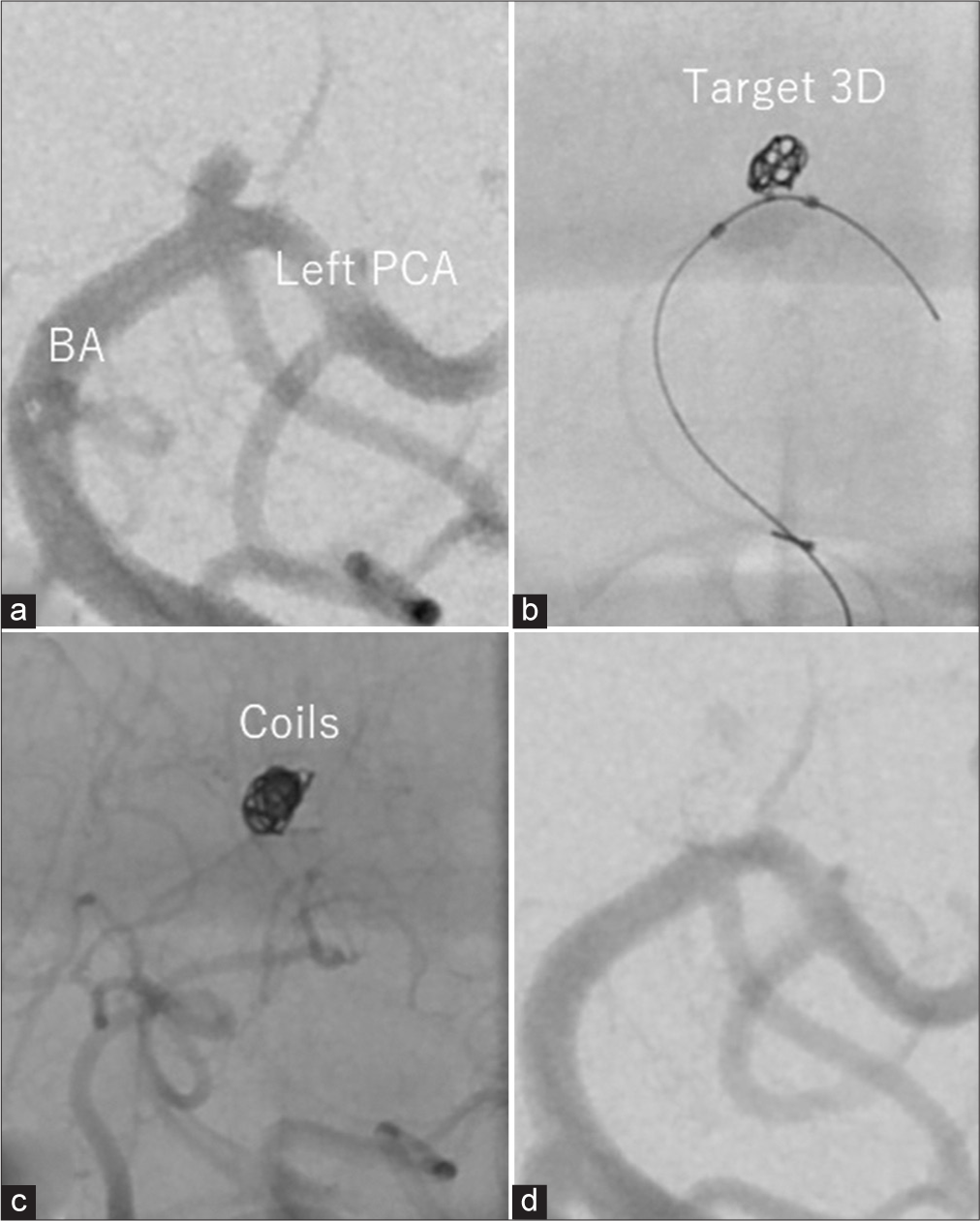
Figure 2:
Cerebral angiographic findings after coil embolization. (a) Angiographic image showing the region of blood flow in the basilar tip aneurysm. (b) The frame was created by the Target 3D Coil with balloon assistance. (c) Final image obtained after coil embolization. (d) Final angiographic image showing the disappearance of the aneurysmal sac. BA: Basilar artery, PCA: Posterior cerebral artery.
The procedure was performed through the right femoral artery. A 6-Fr guiding catheter (Envoy; Codman Neuro, Raynham, MA, USA) was introduced at the C3 vertebral body level through the right vertebral artery. With the assistance of a 4-mm × 7-mm balloon catheter (HyperForm; Micro Therapeutics, Irvine, CA, USA), coil embolization was initiated using a coil delivery microcatheter (SL10; Stryker Neurovascular, Fremont, CA, USA). To prevent embedding of the coil in the thrombus, we used a soft 3-mm × 6-cm Target 3D device as the framing coil; this consisted of closed-loop secondary coils, allowing the formation of a well-constructed frame within the aneurysmal sac [
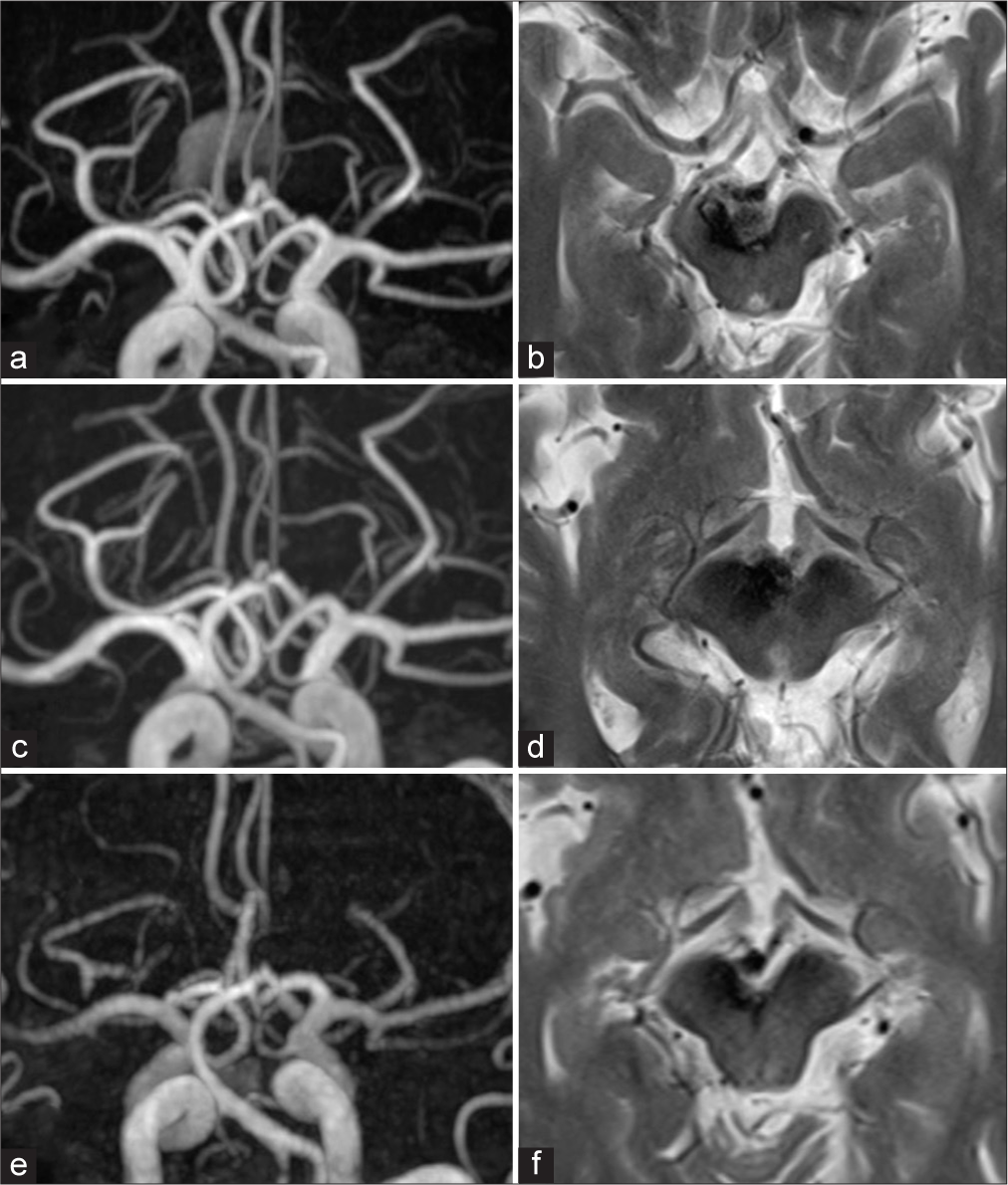
Follow-up MRA two months after surgery revealed residual thrombus but no restoration of blood flow to the sac [
Figure 3:
Radiological findings at 2 months (a and b), 6 months (c and d), and 5 years (e and f) after coil embolization. (a) MRA image revealing residual thrombus at the basilar tip. (b) T2WI showing that the thrombus has reduced in size, as has the midbrain compression. (c) MRA image indicating a significant reduction in thrombus at the basilar tip. (d) T2WI confirms the further reduction in midbrain compression as a result of thrombus. (e) MRA image demonstrating the complete disappearance of the thrombus. (f) T2WI shows complete resolution of the midbrain compression. MRA: Magnetic resonance angiography, T2WI: T2-weighted image.
DISCUSSION
In the treatment of symptomatic large PTIA, coil embolization into the aneurysmal sac resulted not only in the disappearance of intra-aneurysmal blood flow but also in the complete resolution of PTIA itself. Ultimately, the patient’s neurological symptoms improved, and his course has been recurrence-free in the long term.
The recurrence rate following intracranial surgery for large PTIA is typically higher than that for conventional large arterial aneurysms, especially when aiming to achieve a cure by simple coil embolization into the aneurysmal sac of PTIA, as documented in the literature.[
One factor determining the nature of coils in relation to the aneurysm wall is the configuration of the coil loops. These loops, which form the secondary coil diameter, can be broadly categorized as open or closed. Open loops refer to coils where part of the loop remains unclosed. These loops tend to spread outward when adhering to the aneurysmal wall. When using such coils in a PTIA, the coil loops may embed into the thrombus in the long term, potentially leading to restoration of blood flow. In contrast, closed loops are fully closed and remain in place, solidifying inward due to internal forces. The Target 3D Coil used for framing in this case consists entirely of closed loops; this coil remains in place and achieves more than 90% closure.[
Flow diverter stents with flow-altering effects are now widely used in the treatment of difficult-to-manage cerebral aneurysms.[
Disappearance of the aneurysm itself following treatment of PTIA has been observed occasionally, but the precise mechanism remains unclear. The mechanism possibly includes interruption of the blood supply to the vasa vasorum as a result of the disappearance of intra-aneurysmal blood flow,[
CONCLUSION
Our case demonstrates the effectiveness of straightforward coil embolization for a symptomatic large PTIA. The characteristics of the Target 3D Coil may have prevented coil migration into the thrombus, potentially contributing to the successful resolution of the aneurysm. The patient has remained recurrence-free in the long term. This highlights the potential for healing with a simplified coil treatment approach, considering the coil characteristics for a large PTIA.
Ethical approval
Institutional Review Board approval is not required.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consents.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
Disclaimer
The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Journal or its management. The information contained in this article should not be considered to be medical advice; patients should consult their own physicians for advice as to their specific medical needs.
References
1. Chancellor B, Raz E, Shapiro M, Tanweer O, Nossek E, Riina HA. Flow diversion for intracranial aneurysm treatment: Trials involving flow diverters and long-term outcomes. Neurosurgery. 2020. 86: S36-45
2. Cloft HJ, Joseph GJ, Tong FC, Goldstein JH, Dion JE. Use of three-dimensional Guglielmi detachable coils in the treatment of wide-necked cerebral aneurysms. AJNR Am J Neuroradiol. 2000. 21: 1312-4
3. Ferns SP, van Rooij WJ, Sluzewski M, van den Berg R, Majoie CB. Partially thrombosed intracranial aneurysms presenting with mass effect: Long-term clinical and imaging follow-up after endovascular treatment. AJNR Am J Neuroradiol. 2010. 31: 1197-205
4. Hasan DM, Nadareyshvili AI, Hoppe AL, Mahaney KB, Kung DK, Raghavan ML. Cerebral aneurysm sac growth as the etiology of recurrence after successful coil embolization. Stroke. 2012. 43: 866-8
5. Hope JK, Byrne JV, Molyneux AJ. Factors influencing successful angiographic occlusion of aneurysms treated by coil embolization. AJNR Am J Neuroradiol. 1999. 20: 391-9
6. Hoppe AL, Raghavan ML, Hasan DM. Comparison of the association of sac growth and coil compaction with recurrence in coil embolized cerebral aneurysms. PLoS One. 2015. 10: e0123017
7. Kim SJ, Choi IS. Midterm outcome of partially thrombosed intracranial aneurysms treated with guglielmi detachable coils. Interv Neuroradiol. 2000. 6: 13-25
8. Kim YJ, Ko JH. Endovascular treatment of a large partially thrombosed basilar tip aneurysm. J Korean Neurosurg Soc. 2012. 51: 62-5
9. Krings T, Alvarez H, Reinacher P, Ozanne A, Baccin CE, Gandolfo C. Growth and rupture mechanism of partially thrombosed aneurysms. Interv Neuroradiol. 2007. 13: 117-26
10. Lawton MT, Quinones-Hinojosa A, Chang EF, Yu T. Thrombotic intracranial aneurysms: Classification scheme and management strategies in 68 patients. Neurosurgery. 2005. 56: 441-54 discussion-54
11. Lee J, Cho WS, Yoo RE, Yoo DH, Cho YD, Kang HS. The fate of partially thrombosed intracranial aneurysms treated with endovascular intervention. J Korean Neurosurg Soc. 2021. 64: 427-36
12. Matsuda Y, Terada T, Tetsuo Y, Tsumoto T. Partially thrombosed giant basilar tip aneurysm that remarkably decreased in size after stent-assisted coiling associated with the disappearance of neovascularization. Neuroradiology. 2022. 64: 837-41
13. Mitsui T, Nakagawa I, Kotsugi M, Park H, Yokoyama S, Myouchin K. Remarkable shrinkage of a thrombosed giant aneurysm by stent-assisted jam-packed coil embolization. Surg Neurol Int. 2021. 12: 328
14. Mizutani T, Miki Y, Kojima H, Suzuki H. Proposed classification of nonatherosclerotic cerebral fusiform and dissecting aneurysms. Neurosurgery. 1999. 45: 253-9 discussion 9-60
15. Okuda H, Hirokawa N, Saitoh M, Otani A, Someya M, Usami Y. Stiff coils enhance shape retention and pressure resistance in an aneurysm model even at low volume. Minim Invasive Ther Allied Technol. 2022. 31: 767-76
16. Roccatagliata L, Guedin P, Condette-Auliac S, Gaillard S, Colas F, Boulin A. Partially thrombosed intracranial aneurysms: symptoms, evolution, and therapeutic management. Acta Neurochir (Wien). 2010. 152: 2133-42
17. Sattur MG, Li Y, Almallouhi E, Lena J, Spiotta AM. Lessons learned from endovascular coil embolization of pericallosal artery aneurysms and adoption of flow diversion: A retrospective cohort assessment of the efficacy of coiling and flow diversion. World Neurosurg. 2019. 129: e444-51