- Department of Neurosurgery, Toho University Graduate School of Medicine, Tokyo, Japan.
- Department of Neurosurgery, Seisho Hospital, Kanagawa, Japan.
- Department of Neurosurgery, Toho University Ohashi Medical Center, Tokyo, Japan.
Correspondence Address:
Takashi Matsumoto, Department of Neurosurgery, Toho University Graduate School of Medicine, Tokyo, Japan.
DOI:10.25259/SNI_749_2022
Copyright: © 2022 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Takashi Matsumoto1, Masataka Takeuchi2, Atsushi Uyama2, Yoshifumi Konishi2, Satoshi Iwabuchi3. Support and guide performance comparison of balloon guide catheters. 28-Oct-2022;13:490
How to cite this URL: Takashi Matsumoto1, Masataka Takeuchi2, Atsushi Uyama2, Yoshifumi Konishi2, Satoshi Iwabuchi3. Support and guide performance comparison of balloon guide catheters. 28-Oct-2022;13:490. Available from: https://surgicalneurologyint.com/surgicalint-articles/support-and-guide-performance-comparison-of-balloon-guide-catheters/
Abstract
Background: Several types of balloon guide catheters (BGCs) are used in mechanical thrombus retrieval. However, direct comparisons of their supporting and guiding performance have not been reported. We compared the supporting and guiding performance of the Branchor, Flowgate, and Optimo BGCs using a type 3 aorta artificial vascular model.
Methods: An inner catheter was pushed into the artificial vascular model using a linear actuator for the supporting performance evaluation. A previously placed BGC in the internal carotid artery was then intentionally caused to slip. Supporting performance was evaluated by measuring the distance the BGC slipped and generated maximum resistance during Inner catheter insertion. For the guiding performance experiment, a linear actuator was used to guide the BGC into the internal carotid artery of the artificial vessel model. The guiding performance was evaluated by measuring the distance reached by the BGC, maximum resistance generated during insertion of the guiding catheter, and distance the inner catheter slipped. Each experiment was replicated 5 times.
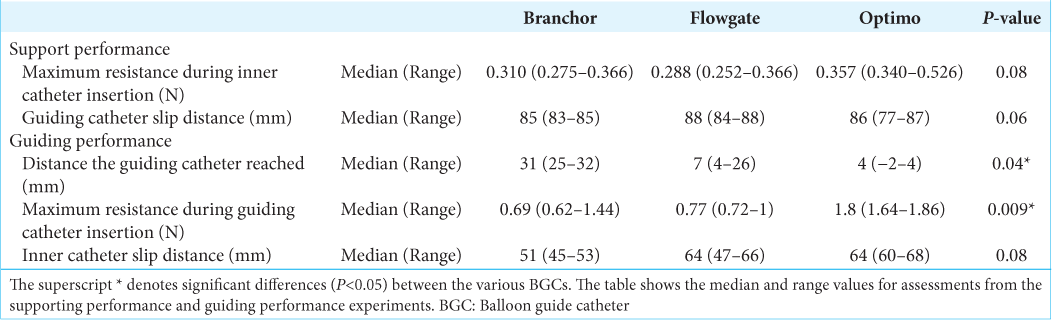
Results: No statistically significant differences were observed in the results of the five supporting performance experiments. However, the results of the first and second experiments suggested that the Optimo offers better supporting performance. In the guiding performance experiment, significant differences were observed, suggesting that the Branchor and Flowgate have superior guiding performance in comparison with the Optimo.
Conclusion: The Optimo offered superior supporting performance, while the Branchor and Flowgate showed better guiding performance than the Optimo.
Keywords: Balloon guide catheter, Mechanical thrombectomy, Performance
INTRODUCTION
Mechanical thrombus retrieval for acute cerebral infarction is an established therapeutic procedure.[
MATERIALS AND METHODS
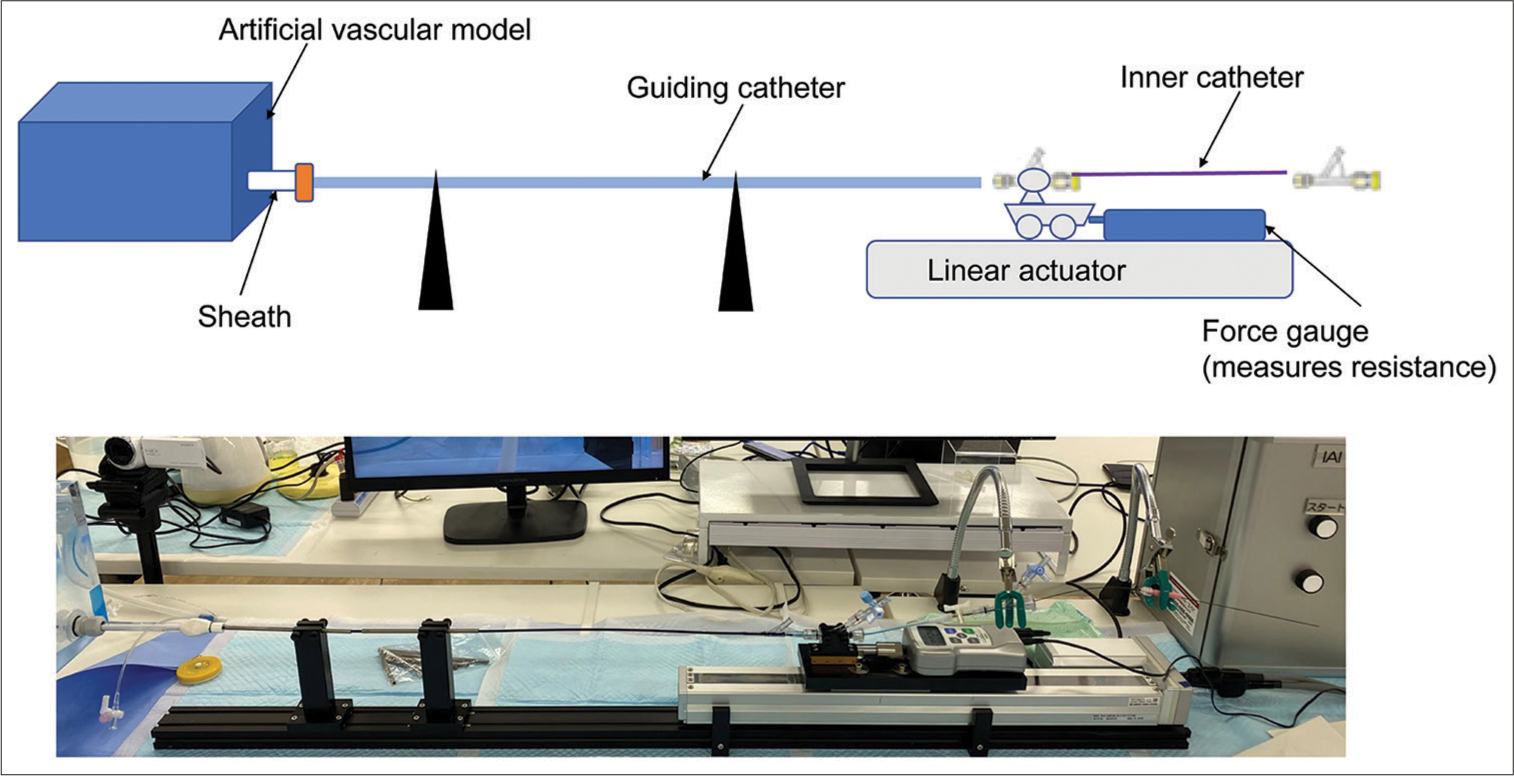
Experimental device
A silicone vascular model of the aortic arch to the neck’s internal carotid artery was created to represent a type 3 aorta [
Support and guide performance evaluation of BGC
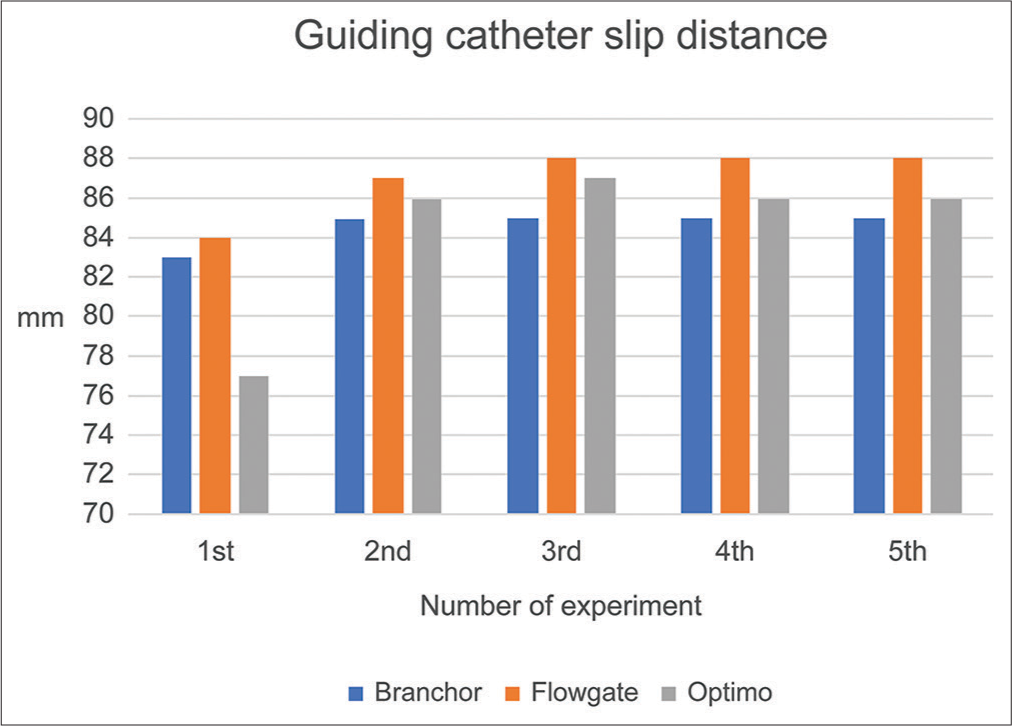
For the supporting performance, a BGC was placed in the internal carotid artery, and an inner catheter and a guidewire were passed through it until they touched the wall of the artificial vascular model. Then, the 12-cm inner catheter was pushed forward at a rate of 1 cm/s, which caused the BGC to slip. The distance the BGC slipped and maximum resistance generated when inserting the inner catheter was evaluated. The supporting performance was considered more significant when the distance the BGC slipped was shorter and when the maximum resistance of the inner catheter was higher.
For the guiding performance, a BGC was guided 12cm into the internal carotid artery from the aortic arch at a rate of 1cm/s with an inner catheter and a guidewire placed in the internal carotid artery. The reached distance of the BGC, maximum resistance generated during insertion of the guiding catheter, and distance the inner catheter slipped was evaluated. Guiding performance was considered greater when the distance the BGC reached into the internal carotid artery was longer, the maximum resistance during insertion was smaller, and the distance the inner catheter slipped was shorter.
Materials used
The BGCs, 9Fr Branchor (Asahi Intecc, Aichi, JAPAN), 8Fr Flowgate (Stryker, Kalamazoo, MI, USA), and 9Fr Optimo (Tokai Medical Products, Aichi, JAPAN) were used, while for the inner catheter and guidewire, 6Fr JB2 130 cm (Medikit, Tokyo, JAPAN) and RADIFOCUS 0.035 in stiff 180 cm (TERUMO, Tokyo, JAPAN) were used, respectively.
Study design
The BGCs’ guiding and supporting performance were replicated 5 times each. The same catheter and wire were used in all five supporting performance experiments, after which a new catheter and wire were used for the guiding performance experiments.
Statistical analysis
The results for the three catheter types were examined with the Kruskal–Wallis test, and if significant differences were observed among the three groups, the Steel-Dwass test was added for multiple comparisons. The statistical analysis software used was JMP version 14.2.0 (SAS Institute, Inc., North Carolina, USA).
RESULTS
Figure 4:
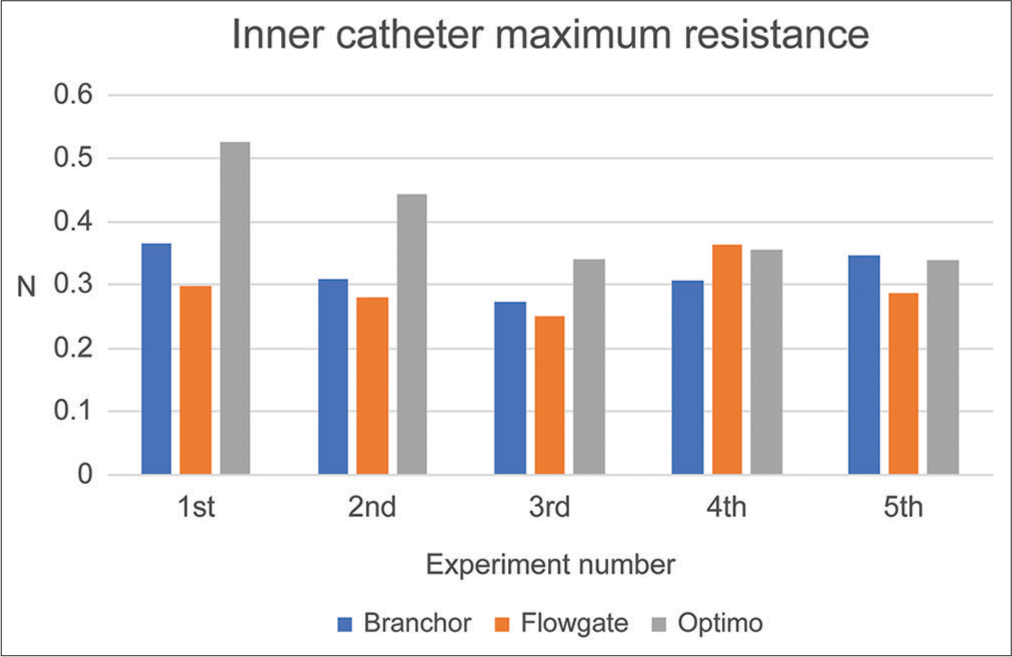
Maximum resistance on the inner catheter in the support performance experiment in the first and second experiments, the maximum resistance on the inner catheter was greater in the Optimo than in the others. Statistical analysis of all five results yielded no statistically significant differences.
DISCUSSION
During mechanical thrombectomy for acute cerebral infarction, difficulties in BGC guiding or slippage during treatment may occur occasionally. Such problems with BGCs can delay the start of treatment until vessel reopening, which could significantly influence patient prognosis. In our experiments, we observed differences in the supporting and guiding performance of different BGC types. In general, a BGC has a soft tip and a body that becomes more rigid as it approaches the hand. Factors that regulate BGC support and guiding performance include rigidity, the pattern of rigidity changes from the tip to the hand, and the coating of the catheter’s outer wall.
No statistically significant differences (P < 0.05) were observed when comparing the five supporting performance experiments. However, in the first and second experiments, the Optimo had a shorter BGC slip distance than the others, and its resistance during inner catheter insertion was higher, suggesting that it offered superior support. The rigidity of the catheter is also critical for evaluating the supporting performance of BGCs; however, the five experiments placed repeated mechanical stress on the catheter, which could have damaged its original rigidity, causing the BGC slip distance to increase and resistance during inner catheter insertion to decrease in the third and subsequent experiments. Considering its original supporting performance, we believe that the Optimo would be effective for cases that require BGCs with a high degree of support performance, such as cases with a sharp bend in the internal carotid artery siphon or peripheral lesions.
In the guiding performance experiment, we observed significant differences (P < 0.05) in the reached distance of the BGC (P = 0.04*) and maximum resistance during BGC insertion (P = 0.009*), with the Branchor reaching significantly longer than the Optimo and the Branchor and Flowgate showing smaller maximum resistance values than the Optimo. This finding indicates that the Branchor and Flowgate would be effective when guiding is expected to be difficult, such as in a type 3 aorta or when the bend from the aortic arch to the internal carotid artery is sharp.
The catheters were inserted at a constant speed using a machine, which differs from the hand movements of actual surgeons. In addition, supporting performance could be improved through balloon inflation, while guiding performance could be enhanced by employing various techniques.[
CONCLUSION
This study suggests that the Optimo had superior supporting performance, while the Branchor and Flowgate had superior guiding performance. These experiments were conducted with a simple mechanical pushing motion that differs from the movements of an actual surgeon’s hands. Building a device that could reproduce an actual surgeon’s rotation and other movements could enable assessments that closely resemble a clinical setting. Evaluation of the supporting and guiding performance of different catheters could enable analysis of the elements required to achieve excellent supporting and guiding performance, which could facilitate the development of new catheters that perform well in both aspects.
Declaration of patient consent
Patient’s consent not required as there are no patients in this study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Disclaimer
The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Journal or its management. The information contained in this article should not be considered to be medical advice; patients should consult their own physicians for advice as to their specific medical needs.
Acknowledgments
We would like to thank Editage (www.editage.com) for English language editing. We received a great deal of contribution from Dr. Yu Shimizu and Dr. Kazuma Tsutou in creating the experimental model.
References
1. Berkhemer OA, Fransen PS, Beumer D, Van den Berg LA, Lingsma HF, Yoo AJ. A randomized trial of intraarterial treatment for acute ischemic stroke. N Engl J Med. 2015. 372: 11-20
2. Brinjikji W, Starke RM, Murad MH, Fiorella D, Pereira VM, Goyal M. Impact of balloon guide catheter on technical and clinical outcomes: A systematic review and meta-analysis. J Neurointerv Surg. 2018. 10: 335-9
3. Campbell BC, Mitchell PJ, Kleinig TJ, Dewey HM, Churilov L, Yassi N. Endovascular therapy for ischemic stroke with perfusion-imaging selection. N Engl J Med. 2015. 372: 1009-18
4. Chueh JY, Puri AS, Wakhloo AK, Gounis MJ. Risk of distal embolization with stent retriever thrombectomy and ADAPT. J Neurointerv Surg. 2016. 8: 197-202
5. Goyal M, Demchuk AM, Menon BK, Eesa M, Rempel JL, Thornton J. Randomized assessment of rapid endovascular treatment of ischemic stroke. N Engl J Med. 2015. 372: 1019-30
6. Goyal M, Menon BK, Van Zwam WH, Dippel DW, Mitchell PJ, Demchuk AM. Endovascular thrombectomy after large-vessel ischaemic stroke: A meta-analysis of individual patient data from five randomised trials. Lancet. 2016. 387: 1723-31
7. Jovin TG, Chamorro A, Cobo E, De Miquel MA, Molina CA, Rovira A. Thrombectomy within 8 hours after symptom onset in ischemic stroke. N Engl J Med. 2015. 372: 2296-306
8. Saver JL, Goyal M, Bonafe A, Diener HC, Levy EI, Pereira VM. Stent-retriever thrombectomy after intravenous t-PA vs. t-PA alone in stroke. N Engl J Med. 2015. 372: 2285-95
9. Saver JL, Goyal M, Van der Lugt A, Menon BK, Majoie CB, Dippel DW. Time to treatment with endovascular thrombectomy and outcomes from ischemic stroke: A meta-analysis. JAMA. 2016. 316: 1279-88
10. Sharashidze V, Nogueira RG, Al-Bayati AR, Grossberg JA, Haussen DC. Balloon anchoring technique for thrombectomy in hostile craniocervical arterial anatomy. J Neurointerv Surg. 2020. 12: 763-67
11. Yoshimura S, Enomoto Y, Kitajima H, Yamada J, Kaku Y, Iwama T. Carotid-compression technique for the insertion of guiding catheters. AJNR Am J Neuroradiol. 2006. 27: 1710-1