- Rose Ella Burkhardt Brain Tumor and Neuro-oncology Center, Neurological Institute, Cleveland Clinic, Cleveland, Ohio, USA
- Minimally Invasive Cranial Base and Pituitary Surgery Program, Cleveland Clinic, Cleveland, Ohio, USA
- Department of Endocrinology, Diabetes, and Metabolism, Cleveland Clinic, Cleveland, Ohio, USA
- Department of Anesthesiology, Cleveland Clinic, Cleveland, Ohio, USA
- Section of Skull Base Rhinology and Sinus Surgery, Head and Neck Institute, Cleveland Clinic, Cleveland, Ohio, USA
Correspondence Address:
Pablo F. Recinos
Rose Ella Burkhardt Brain Tumor and Neuro-oncology Center, Neurological Institute, Cleveland Clinic, Cleveland, Ohio, USA
Minimally Invasive Cranial Base and Pituitary Surgery Program, Cleveland Clinic, Cleveland, Ohio, USA
Section of Skull Base Rhinology and Sinus Surgery, Head and Neck Institute, Cleveland Clinic, Cleveland, Ohio, USA
DOI:10.4103/2152-7806.170472
Copyright: © 2015 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Abbassy M, Kshettry VR, Hamrahian AH, Johnston PC, Dobri GA, Avitsian R, Woodard TD, Recinos PF. Surgical management of recurrent Cushing's disease in pregnancy: A case report. Surg Neurol Int 25-Nov-2015;6:
How to cite this URL: Abbassy M, Kshettry VR, Hamrahian AH, Johnston PC, Dobri GA, Avitsian R, Woodard TD, Recinos PF. Surgical management of recurrent Cushing's disease in pregnancy: A case report. Surg Neurol Int 25-Nov-2015;6:. Available from: http://surgicalneurologyint.com/surgicalint_articles/surgical-management-of-recurrent-cushings-disease-in/
Abstract
Background:Cushing's disease is a condition rarely encountered during pregnancy. It is known that hypercortisolism is associated with increased maternal and fetal morbidity and mortality. When hypercortisolism from Cushing's disease does occur in pregnancy, the impact of achieving biochemical remission on fetal outcomes is unknown. We sought to clarify the impact of successful surgical treatment by presenting such a case report.
Case Description:A 38-year-old pregnant woman with recurrent Cushing's disease after 8 years of remission. The patient had endoscopic transsphenoidal of her pituitary adenoma in her 18th week of pregnancy. The patient had postoperative biochemical remission and normal fetal outcome with no maternal complications.
Conclusion:Transsphenoidal surgery for Cushing's disease can be performed safely during the second trimester of pregnancy.
Keywords: Cushing's syndrome, endonasal, endoscopic, fetal complications, minimally invasive skull base surgery, operative timing, pituitary
INTRODUCTION
The first published case of Cushing's syndrome in pregnancy was reported by Hunt and McConahey in 1953.[
The diagnosis of Cushing's disease in pregnancy presents a challenge due to up regulation of the hypothalamic-pituitary axis (HPA) and associated hypercortisolism of pregnancy.[
CASE DESCRIPTION
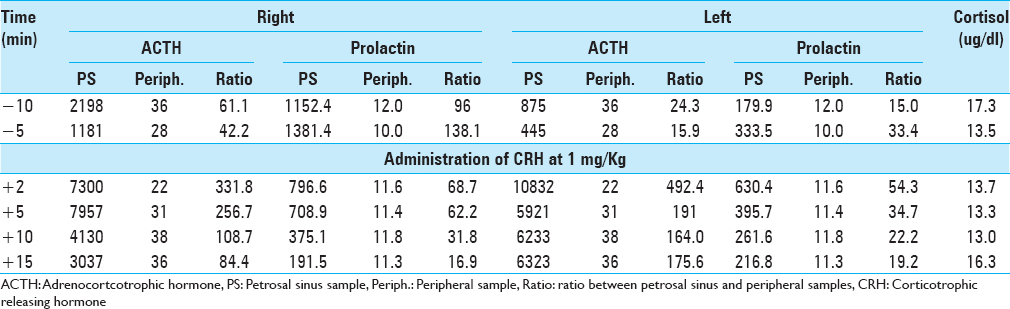
A 38-year-old pregnant woman presented with recurrent Cushing's disease. She had initially presented 8 years prior with weight gain, truncal obesity, and hypertension. Adrenocorticotrophic hormone (ACTH)-dependent hypercortisolism was confirmed. Urinary free cortisol (UFC) was 105.1 ug/24 h (reference range <45 ug/24 h), midnight serum cortisol was 249 ng/dl (reference range <100 ng/dl), and ACTH was 60 pg/ml (reference range 5–27 pg/ml). The patient had a negative high-resolution brain magnetic resonance imaging (MRI) with contrast. Inferior petrosal sinus sampling (IPSS) results were consistent with a pituitary source of her ACTH-dependent Cushing's syndrome [
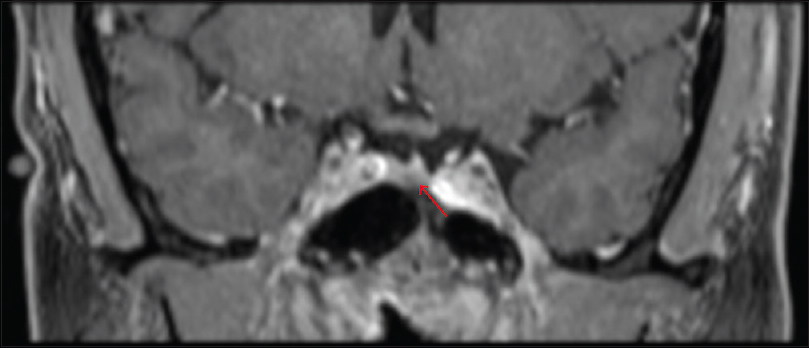
The patient remained in remission and re-presented after 8 years with symptoms of fatigue, diaphoresis, 50 pound (22.7 kg) weight gain over 3–4 months, easy bruising, hair loss, hypertension, and headaches. Biochemical workup was as follows: 24-h UFC 172.1 μg (reference range <45 ug/24 h), ACTH 56.3 pg/ml (reference range 6–48 pg/ml); 8 a.m. serum cortisol 26.9 μg/dl. Two consecutive midnight salivary cortisol samples were 245 and 262 ng/dl (reference range <100 ng/dl). Dedicated MRI of the pituitary with gadolinium contrast revealed a 4 mm hypointense sellar lesion that was suspicious for recurrent pituitary microadenoma [
The patient was scheduled for elective surgery, but it was canceled as she was found to be pregnant on the day of the surgery, which corresponded to her 12th week of pregnancy (G4P3). After being re-evaluated by endocrinology and high-risk obstetrics, surgical resection was recommended during pregnancy in order to minimize potential life-threating maternal and fetal complications as a result of hypercortisolism. In addition, it was recommended that surgery be performed during the second trimester of pregnancy. After a thorough discussion of the risks and benefits to both her and the baby, she elected to proceed.
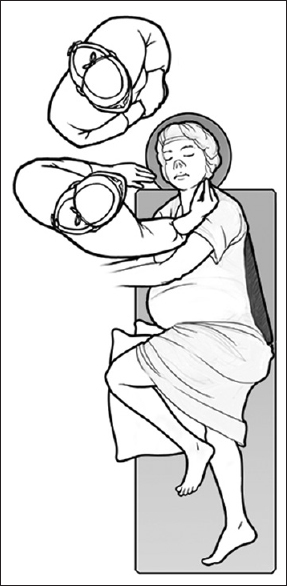
The patient underwent an image-guided endoscopic endonasal transsphenoidal approach during the 18th week of pregnancy. The patient was positioned in semilateral supine position [
DISCUSSION
Physiology of the hypothalamic-pituitary-adrenal axis in pregnancy
The HPA axis function is upregulated during pregnancy. The placenta produces corticotropin-releasing hormone (CRH), which is structurally identical to hypothalamic CRH.[
Endocrinological diagnostic considerations
Clinical diagnosis of Cushing's syndrome in pregnancy is difficult due to an overlap in clinical features such as weight gain, fatigue, emotional change, abdominal striae, hypertension, and hyperglycemia.[
Imaging of the pituitary gland in pregnancy
Detection of a pituitary adenoma on MRI can help confirm the diagnosis of Cushing's disease. However, imaging the pituitary gland during pregnancy requires awareness of special considerations. MRI is generally considered safe during pregnancy, although some authors have recommended avoiding MRI during the first trimester due to potential unknown adverse effects during organogenesis.[
Neurosurgical and anesthetic considerations
Given the high rate of maternal and fetal complications from untreated Cushing's syndrome, treatment is usually justified during pregnancy. The goal of treatment is to reduce UFC to the upper part of normal observed in pregnancy.[
Anesthetic considerations include ensuring adequate uterine blood flow, avoidance of anesthetic agents with possible teratogenic effects, and avoidance of hypoxia and acidosis.[
Medical therapy
Although surgery is the first line treatment option for pregnant patients with Cushing's disease, medical therapy should be considered if surgery is not feasible. Despite being classified as a Class C drug by the FDA, meytrapone is the most commonly used medical therapy and is generally well-tolerated.[
CONCLUSION
We present a case of successful surgical treatment of recurrent Cushing's disease during pregnancy. Biochemical remission was achieved in this case during pregnancy and the outcome was a healthy full-term neonate with no maternal complications during pregnancy or labor. Transsphenoidal surgery for Cushing's disease can be performed safely during the second trimester of pregnancy but requires extra vigilance and close communication between sub-specialty team members.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Abd-Elsayed AA, Diaz-Gomez J, Barnett GH, Kurz A, Inton-Santos M, Barsoum S. A case series discussing the anaesthetic management of pregnant patients with brain tumours. F1000Res. 2013. 2: 92-
2. Berwaerts J, Verhelst J, Mahler C, Abs R. Cushing's syndrome in pregnancy treated by ketoconazole: Case report and review of the literature. Gynecol Endocrinol. 1999. 13: 175-82
3. Boronat M, Marrero D, López-Plasencia Y, Barber M, Schamann Y, Nóvoa FJ. Successful outcome of pregnancy in a patient with Cushing's disease under treatment with ketoconazole during the first trimester of gestation. Gynecol Endocrinol. 2011. 27: 675-7
4. Boscaro M, Arnaldi G. Approach to the patient with possible Cushing's syndrome. J Clin Endocrinol Metab. 2009. 94: 3121-31
5. Bronstein MD, Salgado LR, de Castro Musolino NR. Medical management of pituitary adenomas: The special case of management of the pregnant woman. Pituitary. 2002. 5: 99-107
6. Buescher MA, McClamrock HD, Adashi EY. Cushing syndrome in pregnancy. Obstet Gynecol. 1992. 79: 130-7
7. Carr BR, Parker CR, Madden JD, MacDonald PC, Porter JC. Maternal plasma adrenocorticotropin and cortisol relationships throughout human pregnancy. Am J Obstet Gynecol. 1981. 139: 416-22
8. Casson IF, Davis JC, Jeffreys RV, Silas JH, Williams J, Belchetz PE. Successful management of Cushing's disease during pregnancy by transsphenoidal adenectomy. Clin Endocrinol (Oxf). 1987. 27: 423-8
9. Close CF, Mann MC, Watts JF, Taylor KG. ACTH-independent Cushing's syndrome in pregnancy with spontaneous resolution after delivery: Control of the hypercortisolism with metyrapone. Clin Endocrinol (Oxf). 1993. 39: 375-9
10. Cohen-Kerem R, Railton C, Oren D, Lishner M, Koren G. Pregnancy outcome following non-obstetric surgical intervention. Am J Surg. 2005. 190: 467-73
11. Connell JM, Cordiner J, Davies DL, Fraser R, Frier BM, McPherson SG. Pregnancy complicated by Cushing's syndrome: Potential hazard of metyrapone therapy. Case report. Br J Obstet Gynaecol. 1985. 92: 1192-5
12. Cousins L, Rigg L, Hollingsworth D, Meis P, Halberg F, Brink G. Qualitative and quantitative assessment of the circadian rhythm of cortisol in pregnancy. Am J Obstet Gynecol. 1983. 145: 411-6
13. Coyne TJ, Atkinson RL, Prins JB. Adrenocorticotropic hormone-secreting pituitary tumor associated with pregnancy: Case report. Neurosurgery. 1992. 31: 953-5
14. Day J, Batjer HH, Loftus M.editors. Operative positioning and fetal monitoring considerations. Neurosurgical Aspects of Pregnancy. Illinois: American Association of Neurological Surgeons; 1995. p. 183-90
15. Demey-Ponsart E, Foidart JM, Sulon J, Sodoyez JC. Serum CBG, free and total cortisol and circadian patterns of adrenal function in normal pregnancy. J Steroid Biochem. 1982. 16: 165-9
16. Elster AD, Sanders TG, Vines FS, Chen MY. Size and shape of the pituitary gland during pregnancy and post partum: Measurement with MR imaging. Radiology. 1991. 181: 531-5
17. Goland RS, Wardlaw SL, Blum M, Tropper PJ, Stark RI. Biologically active corticotropin-releasing hormone in maternal and fetal plasma during pregnancy. Am J Obstet Gynecol. 1988. 159: 884-90
18. Gonzalez JG, Elizondo G, Saldivar D, Nanez H, Todd LE, Villarreal JZ. Pituitary gland growth during normal pregnancy: An in vivo study using magnetic resonance imaging. Am J Med. 1988. 85: 217-20
19. Gopal RA, Acharya SV, Bandgar TR, Menon PS, Shah NS. Cushing disease with pregnancy. Gynecol Endocrinol. 2012. 28: 533-5
20. Gormley MJ, Hadden DR, Kennedy TL, Montgomery DA, Murnaghan GA, Sheridan B. Cushing's syndrome in pregnancy – Treatment with metyrapone. Clin Endocrinol (Oxf). 1982. 16: 283-93
21. Guilhaume B, Sanson ML, Billaud L, Bertagna X, Laudat MH, Luton JP. Cushing's syndrome and pregnancy: Aetiologies and prognosis in twenty-two patients. Eur J Med. 1992. 1: 83-9
22. Hána V, Dokoupilová M, Marek J, Plavka R. Recurrent ACTH-independent Cushing's syndrome in multiple pregnancies and its treatment with metyrapone. Clin Endocrinol (Oxf). 2001. 54: 277-81
23. Hunt AB, McConahey WM. Pregnancy associated with diseases of the adrenal glands. Am J Obstet Gynecol. 1953. 66: 970-87
24. Leiba S, Weinstein R, Shindel B, Lapidot M, Stern E, Levavi H. The protracted effect of o, p’-DDD in Cushing's disease and its impact on adrenal morphogenesis of young human embryo. Ann Endocrinol (Paris). 1989. 50: 49-53
25. Lindsay JR, Jonklaas J, Oldfield EH, Nieman LK. Cushing's syndrome during pregnancy: Personal experience and review of the literature. J Clin Endocrinol Metab. 2005. 90: 3077-83
26. Lindsay JR, Nieman LK. Adrenal disorders in pregnancy. Endocrinol Metab Clin North Am. 2006. 35: 1-20, v
27. Lindsay JR, Nieman LK. The hypothalamic-pituitary-adrenal axis in pregnancy: Challenges in disease detection and treatment. Endocr Rev. 2005. 26: 775-99
28. Magiakou MA, Mastorakos G, Webster E, Chrousos GP. The hypothalamic-pituitary-adrenal axis and the female reproductive system. Ann N Y Acad Sci. 1997. 816: 42-56
29. McCarroll F, Lindsay JR, Swearingen B, Biller BM.editors. Cushing's syndrome in pregnancy. Cushing's Disease. New York: Springer; 2011. p.
30. McLean M, Bisits A, Davies J, Woods R, Lowry P, Smith R. A placental clock controlling the length of human pregnancy. Nat Med. 1995. 1: 460-3
31. Mellor A, Harvey RD, Pobereskin LH, Sneyd JR. Cushing's disease treated by trans-sphenoidal selective adenomectomy in mid-pregnancy. Br J Anaesth. 1998. 80: 850-2
32. Molitch ME. Pituitary tumors and pregnancy. Growth Horm IGF Res. 2003. 13: S38-44
33. Moudgal VV, Sobel JD. Antifungal drugs in pregnancy: A review. Expert Opin Drug Saf. 2003. 2: 475-83
34. Murphy BE. Conversion of cortisol to cortisone by the human uterus and its reversal in pregnancy. J Clin Endocrinol Metab. 1977. 44: 1214-7
35. Newell-Price J, Trainer P, Perry L, Wass J, Grossman A, Besser M. A single sleeping midnight cortisol has 100% sensitivity for the diagnosis of Cushing's syndrome. Clin Endocrinol (Oxf). 1995. 43: 545-50
36. Nolten WE, Rueckert PA. Elevated free cortisol index in pregnancy: Possible regulatory mechanisms. Am J Obstet Gynecol. 1981. 139: 492-8
37. Odagiri E, Ishiwatari N, Abe Y, Jibiki K, Adachi T, Demura R. Hypercortisolism and the resistance to dexamethasone suppression during gestation. Endocrinol Jpn. 1988. 35: 685-90
38. Okamoto E, Takagi T, Makino T, Sata H, Iwata I, Nishino E. Immunoreactive corticotropin-releasing hormone, adrenocorticotropin and cortisol in human plasma during pregnancy and delivery and postpartum. Horm Metab Res. 1989. 21: 566-72
39. Oldfield EH, Doppman JL, Nieman LK, Chrousos GP, Miller DL, Katz DA. Petrosal sinus sampling with and without corticotropin-releasing hormone for the differential diagnosis of Cushing's syndrome. N Engl J Med. 1991. 325: 897-905
40. Pickard J, Jochen AL, Sadur CN, Hofeldt FD. Cushing's syndrome in pregnancy. Obstet Gynecol Surv. 1990. 45: 87-93
41. Pinette MG, Pan YQ, Oppenheim D, Pinette SG, Blackstone J. Bilateral inferior petrosal sinus corticotropin sampling with corticotropin-releasing hormone stimulation in a pregnant patient with Cushing's syndrome. Am J Obstet Gynecol. 1994. 171: 563-4
42. Ross RJ, Chew SL, Perry L, Erskine K, Medbak S, Afshar F. Diagnosis and selective cure of Cushing's disease during pregnancy by transsphenoidal surgery. Eur J Endocrinol. 1995. 132: 722-6
43. Sam S, Molitch ME. Timing and special concerns regarding endocrine surgery during pregnancy. Endocrinol Metab Clin North Am. 2003. 32: 337-54
44. Sammour RN, Saiegh L, Matter I, Gonen R, Shechner C, Cohen M. Adrenalectomy for adrenocortical adenoma causing Cushing's syndrome in pregnancy: A case report and review of literature. Eur J Obstet Gynecol Reprod Biol. 2012. 165: 1-7
45. Sasaki A, Liotta AS, Luckey MM, Margioris AN, Suda T, Krieger DT. Immunoreactive corticotropin-releasing factor is present in human maternal plasma during the third trimester of pregnancy. J Clin Endocrinol Metab. 1984. 59: 812-4
46. Sasaki A, Shinkawa O, Yoshinaga K. Immunoreactive corticotropin-releasing hormone in amniotic fluid. Am J Obstet Gynecol. 1990. 162: 194-8
47. Sasaki A, Tempst P, Liotta AS, Margioris AN, Hood LE, Kent SB. Isolation and characterization of a corticotropin-releasing hormone-like peptide from human placenta. J Clin Endocrinol Metab. 1988. 67: 768-73
48. Seckl JR, Cleasby M, Nyirenda MJ. Glucocorticoids, 11beta-hydroxysteroid dehydrogenase, and fetal programming. Kidney Int. 2000. 57: 1412-7
49. Sheeler LR. Cushing's syndrome and pregnancy. Endocrinol Metab Clin North Am. 1994. 23: 619-27
50. Suda T, Iwashita M, Ushiyama T, Tozawa F, Sumitomo T, Nakagami Y. Responses to corticotropin-releasing hormone and its bound and free forms in pregnant and nonpregnant women. J Clin Endocrinol Metab. 1989. 69: 38-42
51. Tabarin A, Laurent F, Catargi B, Olivier-Puel F, Lescene R, Berge J. Comparative evaluation of conventional and dynamic magnetic resonance imaging of the pituitary gland for the diagnosis of Cushing's disease. Clin Endocrinol (Oxf). 1998. 49: 293-300
52. Terhune KP, Jagasia S, Blevins LS, Phay JE. Diagnostic and therapeutic dilemmas of hypercortisolemia during pregnancy: A case report. Am Surg. 2009. 75: 232-4
53. Tuncali B, Aksun M, Katircioglu K, Akkol I, Savaci S. Intraoperative fetal heart rate monitoring during emergency neurosurgery in a parturient. J Anesth. 2006. 20: 40-3
54. Verdugo C, Alegría J, Grant C, Briano E, González MI, Meza H. Cushing's disease treatment with transsphenoidal surgery during pregnancy. Rev Med Chil. 2004. 132: 75-80
55. Vilar L, Freitas Mda C, Lima LH, Lyra R, Kater CE. Cushing's syndrome in pregnancy: An overview. Arq Bras Endocrinol Metabol. 2007. 51: 1293-302
56. Wadhwa PD, Porto M, Garite TJ, Chicz-DeMet A, Sandman CA. Maternal corticotropin-releasing hormone levels in the early third trimester predict length of gestation in human pregnancy. Am J Obstet Gynecol. 1998. 179: 1079-85
57. Wallace C, Toth EL, Lewanczuk RZ, Siminoski K. Pregnancy-induced Cushing's syndrome in multiple pregnancies. J Clin Endocrinol Metab. 1996. 81: 15-21