- Department of Neurosurgery, David Geffen School of Medicine at UCLA, Los Angeles, USA
- Department of Neurosurgery, Kaiser Permanente, Los Angeles, California, USA
- Department of Plastic Surgery, Kaiser Permanente, Los Angeles, California, USA
Correspondence Address:
Daniel Diaz-Aguilar
Department of Neurosurgery, David Geffen School of Medicine at UCLA, Los Angeles, USA
DOI:10.4103/sni.sni_230_17
Copyright: © 2017 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Sergei Terterov, Daniel Diaz-Aguilar, Rudi Scharnweber, Alex Tucker, Tianyi Niu, Jos’lyn Woodard, Harsimran Brara, Melissa Poh, Catherine Merna, Stephanie Wang, Shayan Rahman. Surgical nuances of partial sacrectomy for chordoma. 20-Nov-2017;8:277
How to cite this URL: Sergei Terterov, Daniel Diaz-Aguilar, Rudi Scharnweber, Alex Tucker, Tianyi Niu, Jos’lyn Woodard, Harsimran Brara, Melissa Poh, Catherine Merna, Stephanie Wang, Shayan Rahman. Surgical nuances of partial sacrectomy for chordoma. 20-Nov-2017;8:277. Available from: http://surgicalneurologyint.com/surgicalint-articles/surgical-nuances-of-partial-sacrectomy-for-chordoma/
Abstract
Background:Sacral chordomas are rare, slow growing, locally aggressive tumors. Unfortunately, aggressive surgical resection is often associated with increased neurological morbidity.
Methods:This technical note focuses on the utilization of partial sacrectomy for the resection of complex spinal chordomas.
Results:The case presented documents the potential range of postoperative morbidity seen in patients undergoing partial sacrectomy for chordomas. Despite iatrogenic morbidity and tumor recurrence, with the cooperation of medical and surgical spine specialists, majority of patients can achieve good long-term outcomes.
Conclusions:Sacral chordomas are rare lesions and pose a therapeutic challenge for spinal surgeons and oncologists. En-bloc surgical resection (e.g., partial sacrectomy) is the treatment of choice for these lesions, and the cooperation between subspecialists can lead to good neurologic outcomes, particularly if gross total resection is achieved.
Keywords: En-bloc chordoma resection, partial sacrectomy, technical surgical nuances
INTRODUCTION
Sacral chordomas are rare, slow growing, and locally aggressive tumors that only have a limited response to radiation and chemotherapy. Although partial sacrectomy is associated with a better prognosis, there is also a concomitant increased neurological morbidity. Due to the complexity of treating these tumors, they are best managed at tertiary care centers by a multidisciplinary team.[
Surgical considerations
Goal for treatment of sacral chordomas: En-bloc oncologic resection The surgical goal for sacral chordomas is en-bloc resection with adequate oncologic tumor margins. There is no role for intralesional debulking, as partial removal with capsule violation invariably leads to tumor cell spillage, and greatly increases the risk of local recurrence.
Anterior approach for partial sacrectomy
A plane must be established between the ventral sacrum and critical retroperitoneal structures (rectum, iliac vessels, and nerve plexi). If such a plane cannot be developed separating the rectum, it should be resected and a colostomy should be performed.
Posterior approach for partial sacrectomy
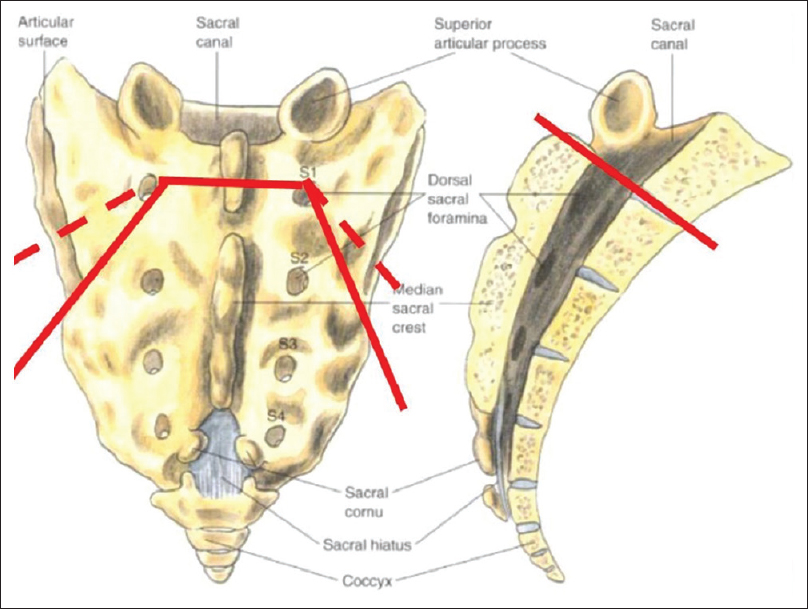
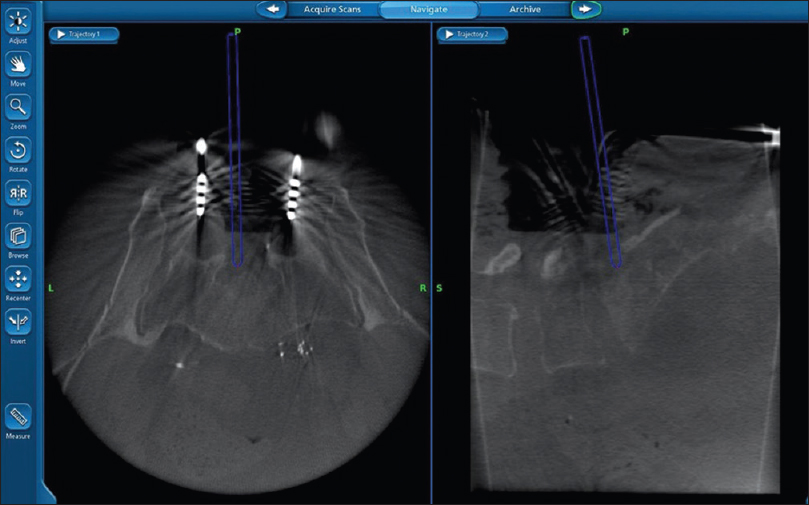
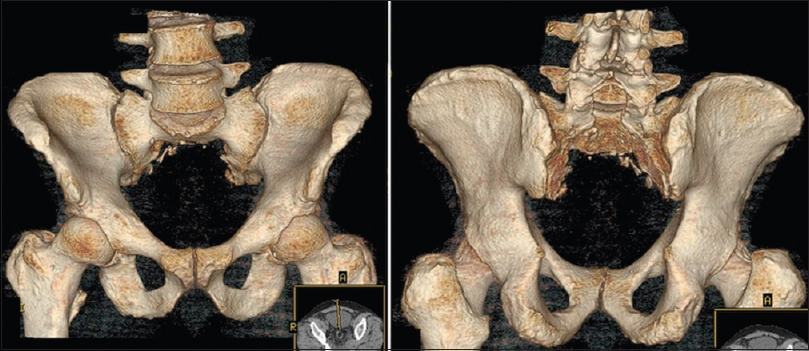
The posterior approach we recommend first involves a midline lumbosacral incision. Dissection is continued until the sacral ala, coccyx, sacropelvic ligaments, and mesorectum are all clearly visualized and the sacropelvic ligaments are then divided. Next, an inferior L5 laminectomy and superior S1 laminotomy are performed, with the traversing S1 nerve roots are identified and preserved. Sacral osteotomies are then guided by fluoroscopy and then performed in an inverted U shape while preserving the sacral nerves in the pelvis [Figures
Sacropelvic stability and reconstruction considerations
In cases of total sacrectomy, biomechanical stability relies on the integrity of iliac instrumentation. For most sacral defects, reconstruction relies on transpelvis vertical rectus abdominisabdominus myocutaneous flap (VRAM) or gluteal flaps. Perineal defects can be reconstructed with similar flaps, as used for sacral defects.
Case Illustration
History
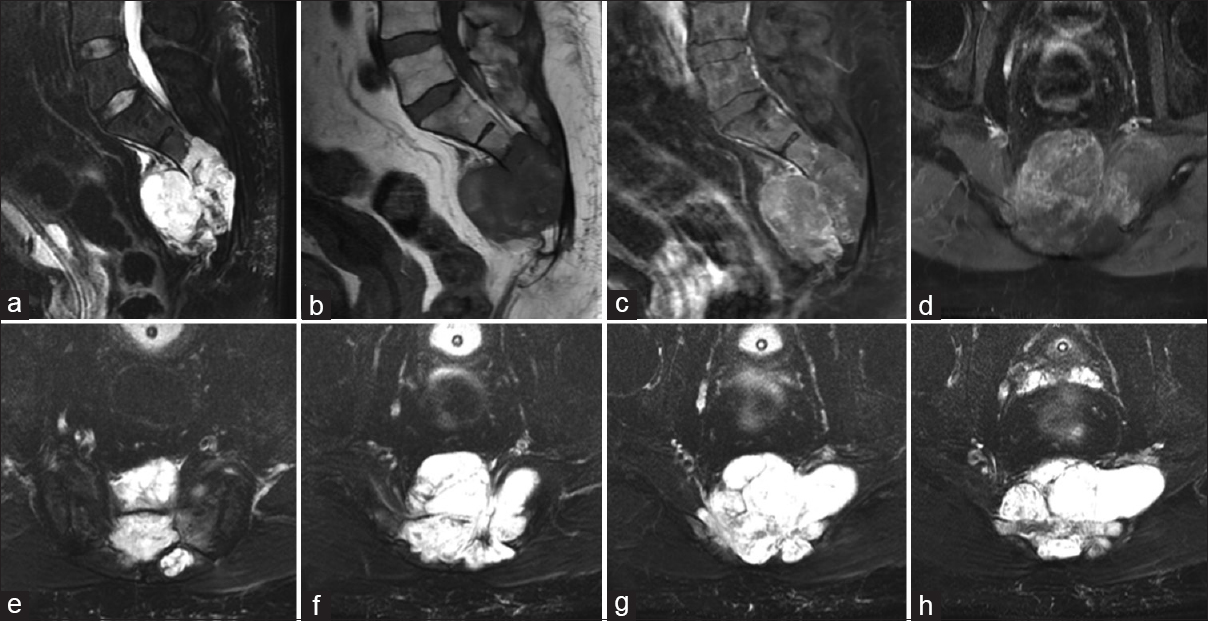
A 73-year-old male had a sacral chordoma identified on magnetic resonance (MR) [
Figure 3
Sagittal magnetic resonance images of T2 (a), T1 precontrast (b), T1 postcontrast and T2 STIR sequences (c and d), demonstrating a T2 hyperintense, weakly enhancing sacral mass with a small amount of extension into the pelvis. Serial axial T2 sequences demonstrating the lateral extent of the mass in the sacrum (e-h)
Postoperatively, the patient had no motor or sensory deficits in the lower extremities. He developed permanent perineal numbness, urinary retention, and fecal incontinence.[
DISCUSSION
Due to the indolent and aggressive nature of sacral chordomas, aggressive surgical treatment is often warranted and offers the greatest chance of cure.[
During operative planning, the surgical team must decide their style of approach. An anterior approach allows the surgical team to visualize and directly protect the visceral organs. A posterior approach allows for increased visualization/exploration of the neural elements. The combined approach offers the benefits of each, but is associated with increased operative and recovery times.[
When dealing with chordomas, the benefits of preserving neural function and the integrity of visceral organs must also be balanced against those of disease recurrence. Preservation of both S3 roots is often associated with a near 100% preservation of urinary and bowel dysfunction.[
CONCLUSION
Sacral chordomas are rare tumors, and en-bloc surgical resection with partial sacrectomy is the primary treatment of choice. In certain cases, partial sacrectomy to achieve gross total resection may be safe and effective for removal of a sacral chordoma, resulting in acceptable neurologic outcomes.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Asavamongkolkul A, Waikakul S. Wide resection of sacral chordoma via a posterior approach. Int Orthop. 2012. 36: 607-12
2. Cheng EY, Ozerdemoglu RA, Transfeldt EE, Thompson RC. Lumbosacral chordoma. Prognostic factors and treatment. Spine. 1999. 24: 1639-45
3. Garofalo F, di Summa PG, Christoforidis D, Pracht M, Laudato P, Cherix S. Multidisciplinary approach of lumbo-sacral chordoma: From oncological treatment to reconstructive surgery. J Surg Oncol. 2015. 112: 544-54
4. Gottfried ON, Schmidt MH, Stevens EA. Embolization of sacral tumors. Neurosurg Focus. 2003. 15: E4-
5. Guo Y, Palmer JL, Shen L, Kaur G, Willey J, Zhang T. Bowel and bladder continence, wound healing, and functional outcomes in patients who underwent sacrectomy. J Neurosurg Spine. 2005. 3: 106-10
6. Hosalkar HS, Jones KJ, King JJ, Lackman RD. Serial Arterial Embolization for Large Sacral Giant-Cell Tumors. Spine. 2007. 32: 1107-15
7. Puri A, Agarwal MG, Shah M, Srinivas CH, Shukla PJ, Shrikhande SV. Decision making in primary sacral tumors. Spine J. 2009. 9: 396-403
8. Ramamurthy R, Bose JC, Muthusamy V, Natarajan M, Kunjithapatham D. Staged sacrectomy--an adaptive approach. J Neurosurg Spine. 2009. 11: 285-94
9. Randall RL, Bruckner J, Lloyd C, Pohlman TH, Conrad EU. Sacral resection and reconstruction for tumors and tumor-like conditions. Orthopedics. 2005. 28: 307-13
10. Sahakitrungruang C, Chantra K. One-staged subtotal sacrectomy for primary sacral tumor. Ann Surg Oncol. 2009. 16: 2594-
11. Todd LT, Yaszemski MJ, Currier BL, Fuchs B, Kim CW, Sim FH. Bowel and bladder function after major sacral resection. Clin Orthop Relat Res. 2002. 397: 36-9