- REHAB Basel, Clinic for Neurorehabilitation and Paraplegiology, Basel, Switzerland
- Department of Radiology, Division of Diagnostic and Interventional Neuroradiology, University Hospital Basel, Basel, Switzerland
- Department of Neurosurgery, University Hospital Basel, Basel, Switzerland
Correspondence Address:
Christian Saleh
REHAB Basel, Clinic for Neurorehabilitation and Paraplegiology, Basel, Switzerland
DOI:10.25259/SNI-189-2019
Copyright: © 2019 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Christian Saleh, Stefanie Wilmes, Kristine Ann Blackham, Dominik Cordier, Kerstin Hug, Margret Hund-Georgiadis. The cerebellar peduncle as localization of a recurrent atypical plexus papilloma: A case report. 24-Apr-2019;10:65
How to cite this URL: Christian Saleh, Stefanie Wilmes, Kristine Ann Blackham, Dominik Cordier, Kerstin Hug, Margret Hund-Georgiadis. The cerebellar peduncle as localization of a recurrent atypical plexus papilloma: A case report. 24-Apr-2019;10:65. Available from: https://surgicalneurologyint.com/surgicalint-articles/9304/
Abstract
Background:Choroid plexus papillomas (CPPs) are infrequently encountered brain tumors with the majority originating in the ventricular system. Rarely, CPP occurs outside of the ventricles.
Case Description:We report the case of a recurrent CPP that initially originated within the fourth ventricle, though years later it recurred in the left middle cerebellar peduncle.
Conclusion:Patients with cerebellar plexus papilloma need long-term follow-up comprising regular magnetic resonance imagings since, in patients with a history of CPP, any new mild symptomatology, even years after the initial presentation, may be an early sign of tumor recurrence.
Keywords: Brain tumors, choroid plexus papilloma, Nystagmus
INTRODUCTION
Choroid plexus papillomas (CPPs) are rather rare brain tumors originating from the epithelium of the CP. Some of the CP lies along the velum medullare inferius of the fourth ventricle and is responsible for the production of the cerebrospinal fluid (CSF). The tumor classification of the World Health Organization (WHO) differentiates between benign forms, CPP (Grade I), atypical forms (Grade II), and malignant forms, such as the CP carcinoma, CPC (Grade III).[
Here, we report a case of a recurrent CPP that originated from the fourth ventricle, while years later, it reoccurred in the left middle cerebellar peduncle (MCP). To the best of our knowledge, this is the first description of a CPP localized in the MCP.
CASE REPORT
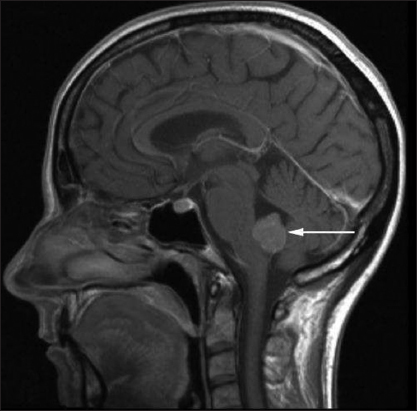
In 2010, a 52-year-old female patient complained of nausea present for more than 6 months, vomitus, intermittent vertigo, loss of appetite with B-symptoms, and weight loss of 10 kg. The initial diagnostic workup consisted of gastrointestinal imaging and a thorax X-ray, which were normal. However, a cranial magnetic resonance imaging (MRI) showed a tumor mass within the fourth ventricle (3 cm × 2 cm of size [Figures
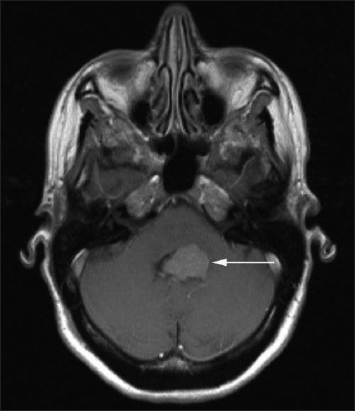
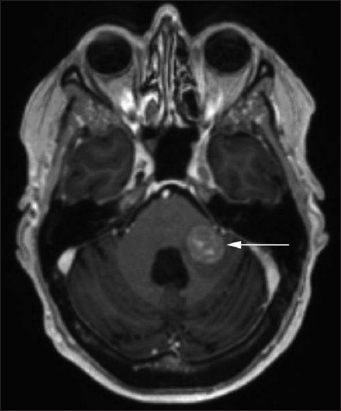
Seven years later (2017), the patient complained again of two episodes of paroxysmal vertigo. Ophthalmological examination revealed a bilateral upbeat nystagmus. In addition, the patient reported a mild headache and bilateral pulse-synchronous tinnitus. Neurological examination showed a mild left-sided limb ataxia. She continued to ambulate without a walking aid. In September 2017, a cranial MRI showed a localized, inhomogeneous contrast-enhancing tumor mass in the middle left cerebellar peduncle (circa 17 mm × 17 mm) [
DISCUSSION
Neuroanatomy
The cerebellum is structurally connected to the brainstem through six symmetrically positioned peduncles. The two inferior cerebellar peduncles project on the medulla oblongata, the MCPs adjoin it to the pons, while the superior CPs represent the link to the midbrain.[
Typical symptoms after MCP lesion
Typically, MCP lesions manifest with a vestibular syndrome (oculomotor symptoms, vertigo, and vomitus) or motor symptoms.
At the time of tumor recurrence, our patient presented with an acute vestibular syndrome characterized by an upbeat nystagmus. Distinct eye findings appear to be common with MCP lesions. Kim and Kim[
Complementary observations were reported in other clinical settings: Hall et al.[
CONCLUSION
Recurrence of CPP is rare, especially in extraventricular regions. However, in patients with a history of CPP, even years after the initial presentation, one should keep in mind that a mild symptomatology, such as vomitus or vertigo (even without eye symptoms), may already be early signs pointing to tumor recurrence. Therefore, patients with cerebellar plexus papilloma need long-term follow-up with sequential MRIs.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form, the patient has given her consent for her images and other clinical information to be reported in the journal. The patient understands that her name and initials will not be published and due efforts will be made to conceal her identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Hall DA, Fraint A, Dafer R. Acute stroke in middle cerebellar peduncle in a patient with FXTAS. Front Genet. 2018. 9: 187-
2. Jang SH, Kwon HG. Injury of the cortico-ponto-cerebellar tract in a patient with mild traumatic brain injury:A case report. Medicine (Baltimore). 2017. 96: e8749-
3. Kim JS, Yoon B, Choi KD, Oh SY, Park SH, Kim BK. Upbeat nystagmus:Clinicoanatomical correlations in 15 patients. J Clin Neurol. 2006. 2: 58-65
4. Kim SH, Kim JS. Eye movement abnormalities in middle cerebellar peduncle strokes. Acta Neurol Belg. 2019. 119: 37-45
5. Louis DN, Ohgaki H, Wiestler OD, Cavenee WK.editors. Choroid plexus tumours. WHO Classification of Tumours of the Central Nervous System. Lyon, France: IARC Press; 2016. p. 123-9
6. Mohanty S, Rout SS, Sarangi GS, Devi K. Choroid plexus papilloma arising from the temporal horn with a bilateral hypersecretory hydrocephalus:A case report and review of literature. World J Oncol. 2016. 7: 51-6
7. Morales H, Tomsick T. Middle cerebellar peduncles:Magnetic resonance imaging and pathophysiologic correlate. World J Radiol. 2015. 7: 438-47
8. Ozdogan S, Gergin YE, Gergin S, Senol O, Tiryaki M, Tatarli N. Choroid plexus carcinoma in adults:An extremely rare case. Pan Afr Med J. 2015. 20: 302-
9. Pierrot-Deseilligny C, Milea D. Vertical nystagmus:Clinical facts and hypotheses. Brain. 2005. 128: 1237-46
10. Safaee M, Oh MC, Bloch O, Sun MZ, Kaur G, Auguste KI. Choroid plexus papillomas:Advances in molecular biology and understanding of tumorigenesis. Neuro Oncol. 2013. 15: 255-67
11. Sethi D, Arora R, Garg K, Tanwar P. Choroid plexus papilloma. Asian J Neurosurg. 2017. 12: 139-41
12. Sharma CM, Kumawat BL, Garg A, Sharma V. Concurrent infarction in bilateral middle cerebellar peduncles:Rare presentation of stroke. Indian J Health Sci Biomed Res. 2017. 10: 340-2
13. Shi Y, Li X, Chen X, Xu Y, Bo G, Zhou H. Imaging findings of extraventricular choroid plexus papillomas:A study of 10 cases. Oncol Lett. 2017. 13: 1479-85
14. Thomas C, Sill M, Ruland V, Witten A, Hartung S, Kordes U. Methylation profiling of choroid plexus tumors reveals 3 clinically distinct subgroups. Neuro Oncol. 2016. 18: 790-6