- Department of Neurosurgery, Kanazawa University, Kanazawa, Japan.
Correspondence Address:
Kouichi Misaki, Department of Neurosurgery, Kanazawa University, Kanazawa, Japan.
DOI:10.25259/SNI_1100_2022
Copyright: © 2023 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Kenshu Nogami, Kouichi Misaki, Taishi Tsutsui, Iku Nambu, Tomoya Kamide, Mitsutoshi Nakada. The first coil embolization ratio is the key factor for retreatment for unruptured cerebral aneurysms. 10-Feb-2023;14:53
How to cite this URL: Kenshu Nogami, Kouichi Misaki, Taishi Tsutsui, Iku Nambu, Tomoya Kamide, Mitsutoshi Nakada. The first coil embolization ratio is the key factor for retreatment for unruptured cerebral aneurysms. 10-Feb-2023;14:53. Available from: https://surgicalneurologyint.com/?post_type=surgicalint_articles&p=12145
Abstract
Background: In coil embolization, a high volume embolization ratio prevents recanalization that may require retreatment. However, patients with a high volume embolization ratio may also require retreatment. Patients with inadequate framing with the first coil may experience recanalization of the aneurysm. We analyzed the relationship between embolization ratio of the first coil and recanalization requiring retreatment.
Methods: We reviewed data from 181 patients with unruptured cerebral aneurysms who underwent initial coil embolization between 2011 and 2021. We retrospectively analyzed the correlation between neck width, maximum aneurysm size, width, aneurysm volume, volume embolization ratio of the framing coil (first volume embolization ratio [1st VER]), and final volume embolization ratio (final VER) of cerebral aneurysms in patients and their retreatment.
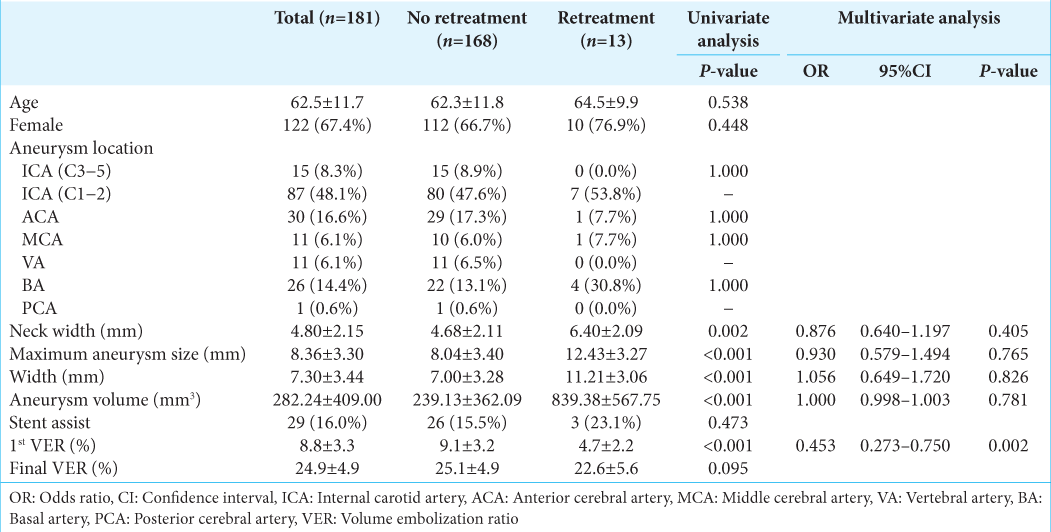
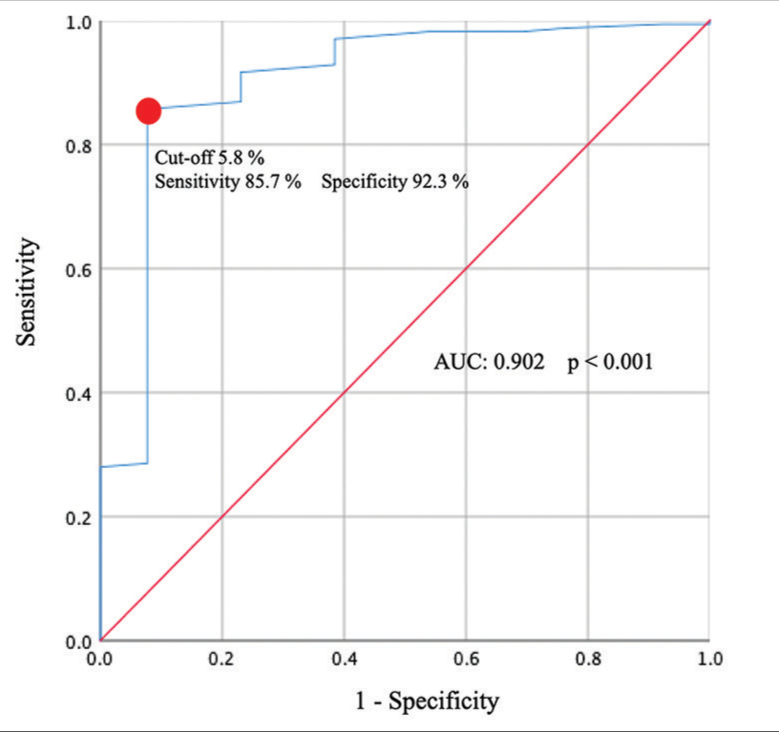
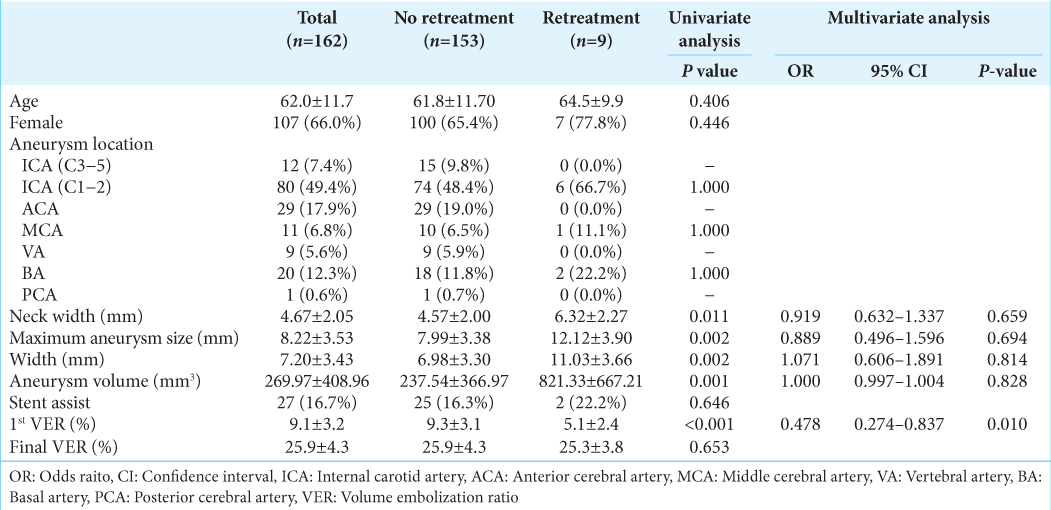
Results: Recanalization requiring retreatment was observed in 13 patients (7.2%). The factors related to recanalization were neck width, maximum aneurysm size, width, aneurysm volume, and 1st VER, but not the final VER. Multivariate analysis of the five factors showed a significant difference in the 1st VER (P = 0.002). The cutoff value for recanalization was a 1st VER of 5.8%. There were 162 cases with a VER ≥ 20% or higher, and the same analysis yielded similar results.
Conclusion: The 1st VER was significantly correlated with recanalization of cerebral aneurysms requiring retreatment. In coil embolization of unruptured cerebral aneurysms, it is important to achieve an embolization rate of at least 5.8% using a framing coil to prevent recanalization.
Keywords: Aneurysm, Coil embolization, First coil, Framing, Volume embolization ratio
INTRODUCTION
Cerebral endovascular treatment has made remarkable progress for the treatment of patients with intracranial aneurysms. Depending on the size, shape, and location of the aneurysm, a wide range of treatment options is now available. These options include simple coiling and adjunctive techniques using stent-assisted techniques, which have become effective alternatives to surgical clipping.[
Several studies have shown that recanalization is related to the volume of the aneurysm and the embolization ratio of the coil.[
MATERIALS AND METHODS
Database
In this study, we reviewed 181 patients with 230 unruptured cerebral aneurysms who underwent initial coil embolization at a single institution between 2011 and 2021. We excluded 27 patients with occlusion of the mother vessel, 15 patients who underwent flow-diverter implantation, six patients with thrombosed aneurysms, and one patient who was lost during follow-up. We retrospectively analyzed the correlation between neck width, maximum aneurysm size, width, aneurysm volume, 1st VER, and final VER of the cerebral aneurysm and recanalization requiring retreatment. Aneurysm volume was measured using a partially automated three-dimensional workstation (Allura 3D-RA workstation, Philips Medical Systems) from the rotational angiography data. The volume of the coils was defined as π (P/2)2 times the coil length, where p is the diameter of the primary coil. Final and 1st VERs were calculated according to the following equations:
1st VER = (the first coil volume)/(the aneurysm volume) × 100 Final VER = (total volume of all inserted coils)/(aneurysm volume) × 100
Endovascular treatment and follow-up
Patients with unruptured aneurysms were treated with two antiplatelet agents (aspirin 100 mg/day and clopidogrel 75 mg/day) starting 2 weeks before surgery. Coil embolization was performed under general anesthesia. Heparin was administered systemically intraoperatively. Postoperative patients who were treated with stent-assisted techniques continued to take the same two antiplatelet agents (aspirin 100 mg/day and clopidogrel 75 mg/day), whereas those who were not treated with stent-assisted techniques took only one agent (aspirin 100 mg/day or clopidogrel 75 mg/day). The diameter of the first coil was determined as the mean size of the maximum diameter and height of the aneurysm or equal to its height, and the longest possible coil in this selection was inserted as the first coil. The coils initially inserted into the aneurysm were the target coil (Stryker, Kalamazoo, Michigan, USA), Cashmere coil (Johnson and Johnson, Miami, Florida, USA), GDC 18 coil (Stryker), Presidio (Johnson and Johnson), and others. We obtained post-embolization follow-up gadolinium-enhanced time-of-flight images at 3, 6, and 12 months and yearly thereafter to detect any residual lesions. When these images indicated an increase in residual lesions, we performed cerebral angiography. The patient was re-treated if the Raymond scale score was III or higher on cerebral angiography.
Data analysis
All statistical analyses were performed using the International Business Machines Corporation and Statistical Package for the Social Sciences (SPSS) Statistics (version 25.0, SPSS Inc., Chicago, IL, USA). Chi-squared test was used for categorical variables, and non-parametric Mann–Whitney test was used for quantitative variables to determine the associations between patient characteristics and recanalization of cerebral aneurysms. Multivariate logistic regression analysis was used to determine the most significant factors for recanalization. Receiver operating characteristic (ROC) curve analysis of this factor was used to determine the cutoff value for recurrence. Differences were considered statistically significant at P < 0.05.
RESULTS
This study included 59 men and 122 women who underwent coil embolization for unruptured cerebral aneurysms [
The mean age of the patients was 62.5 ± 11.7 years. The lesion sites were internal carotid artery (ICA) (C3−5) in 15 patients (8.3%), ICA (C1−2) in 87 patients (48.1%), anterior cerebral artery in 30 patients (16.6%), middle cerebral artery in 11 patients (6.1%), vertebral artery in 11 patients (6.1%), basal artery in 26 patients (14.4%), and posterior cerebral artery in one patient (0.6%). The mean neck width of the aneurysms was 4.80 ± 2.15 mm, the mean maximum aneurysm size was 8.36 ± 3.30 mm, the mean width was 7.30 ± 3.44 mm, and the mean aneurysm volume was 282.24 ± 409.00 mm3. Twenty-nine patients were treated using stent-assisted techniques (16.0%). The mean 1st VER was 8.8 ± 3.3% and the mean final VER was 24.9 ± 4.9%. The mean follow-up period was 36.7 ± 30.3 months.
Recanalization was observed in 13 patients (7.18%), and retreatment was performed an average of 17.8 ± 14.2 months after initial treatment in all cases. The factors found to be associated with retreatment were neck width (no retreatment: 4.68 ± 2.11 mm vs. retreatment: 6.40 ± 2.09 mm, P = 0.002), maximum aneurysm size (8.04 ± 3.40 mm vs. 12.43 ± 3.27 mm, P < 0.001), width (7.00 ± 3.28 mmvs. 11.21 ± 3.06 mm, P < 0.001), aneurysm volume (239.13 ± 362.09 mm3 vs. 839.38 ± 567.75 mm3, P < 0.001), and 1st VER (9.1 ± 3.2% vs. 4.7 ± 2.2%, P < 0.001). There was no significant difference in final VER (25.1 ± 4.9% vs. 22.6 ± 5.6%, P = 0.095). Multivariate analysis of the five factors showed a significant difference in 1st VER (Odds ratio [OR] 0.453, 95% confidence interval [CI], 0.273–0.750; P = 0.002) [
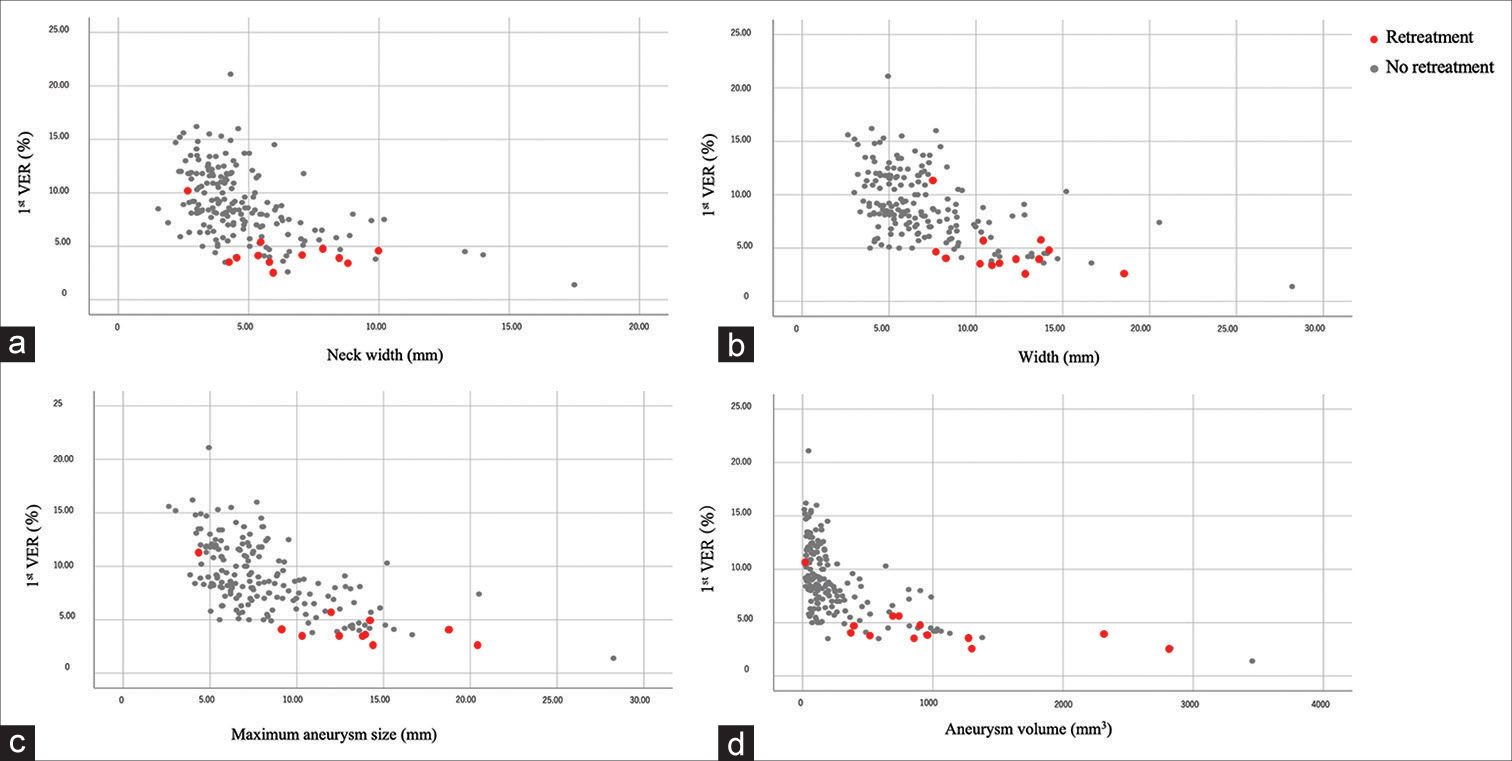
Scatter plots were constructed for the 1st VER and the other recurrence factors. As aneurysm size increased, the 1st VER tended to decrease [
When the same analysis was performed in patients with a final VER of 20% or higher, significant differences were found in neck width (4.57 ± 2.00 mm vs. 6.32 ± 2.27 mm, P = 0.011), maximum aneurysm size (7.99 ± 3.38 mm vs. 12.12 ± 3.90 mm, P = 0.002), width (6.98 ± 3.30 mm vs. 11.03 ± 3.66 mm, P = 0.002), aneurysm volume (237.54 ± 366.97 mm3 vs. 821.33 ± 667.21 mm3, P = 0.001), and 1st VER (9.3 ± 3.1% vs. 5.1 ± 2.4%, P < 0.001) as recanalization factors [
DISCUSSION
In unruptured cerebral aneurysms, this is the study to show that the 1st VER has a stronger correlation with retreatment after coil embolization than the final VER. The treatment options for cerebral aneurysms include open cranial microsurgery and endovascular treatment. The advantages of endovascular treatment are that it is less invasive than craniotomy, requires less hospitalization, and allows patients to recover faster. However, coil embolization has the disadvantage that the aneurysm may recanalization, increasing the risk of aneurysm growth and bleeding.[
In this study, retreatment was performed in 13 of the 181 treated patients with cerebral aneurysms. Statistically, retreatment was associated with 1st VER in addition to factors related to aneurysm shape. Neki et al. analyzed unruptured and ruptured cerebral aneurysms and reported a significant correlation between maximum aneurysm size, ruptured aneurysm, 1st VER, and final VER as factors for recanalization.[
Neki et al. reported that the 1st VER was 15.0–17.5%,[
Our study showed that the larger the aneurysm, the lower the 1st VER tended to be, and most of the unruptured cerebral aneurysms that required retreatment were large. Compared to other reports, the 1st VER tended to be lower for the larger aneurysm in our report because coils with larger volumes did not exist at that time. The results suggest that the challenge is to obtain a high 1st VER for large aneurysms. To achieve this, it is important to select a coil with a large primary diameter and long coil. Coils with primary coil diameters of 0.014 inches and 0.017 inches are twice and 3 times as large as coils with a diameter of 0.010 inches, respectively.
As Ishida et al. reported, we also analyzed the percentage of framing coil volume (FCP) in total volume.[
This study has several limitations. The first limitation is the small sample size. Second, VER was an indicator of coil volume relative to the whole aneurysm volume and did not represent compartmentation or partial tight packing. Third, we did not include the rigidity of the inserted coils in our analysis. Fourth, there are many variations in the treatment itself due to improvements of devices including coils and embolization techniques over the past 10 years, but these were not analyzed in our study. Fifth, we included only unruptured cases and did not evaluate ruptured cases. Finally, this study was conducted retrospectively. Therefore, selection bias is inevitable. Further prospective studies with larger sample sizes are required.
CONCLUSION
The results of this study showed that in unruptured cerebral aneurysms 1st VER is an important factor in recanalization requiring retreatment. We found that a 1st VER of at least 5.8% is required to prevent recanalization.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Disclaimer
The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Journal or its management. The information contained in this article should not be considered to be medical advice; patients should consult their own physicians for advice as to their specific medical needs.
References
1. Fujimura S, Takao H, Suzuki T, Dahmani C, Ishibashi T, Mamori H. Hemodynamics and coil distribution with changing coil stiffness and length in intracranial aneurysms. J Neurointerv Surg. 2018. 10: 797-801
2. Gonda DD, Khalessi AA, McCutcheon BA, Marcus LP, Noorbakhsh A, Chen CC. Long-term follow-up of unruptured intracranial aneurysms repaired in California. J Neurosurg. 2014. 120: 1349-57
3. Ishida W, Sato M, Amano T, Matsumaru Y. The significant impact of framing coils on long-term outcomes in endovascular coiling for intracranial aneurysms: How to select an appropriate framing coil. J Neurosurg. 2016. 125: 705-12
4. Jiang Z, Chen Y, Zeng C, Feng J, Wan Y, Zhang X. Neurosurgical clipping versus endovascular coiling for patients with intracranial aneurysms: A systematic review and meta-analysis. World Neurosurg. 2020. 138: e191-222
5. Kawanabe Y, Sadato A, Taki W, Hashimoto N. Endovascular occlusion of intracranial aneurysms with Guglielmi detachable coils: Correlation between coil packing density and coil compaction. Acta Neurochir (Wien). 2001. 143: 451-5
6. Misaki K, Uchiyama N, Nambu I, Aida Y, Kamide T, Mohri M. Optimizing the volume of the initial framing coil to facilitate tight packing of intracranial aneurysms. World Neurosurg. 2016. 90: 397-402
7. Neki H, Kohyama S, Otsuka T, Yonezawa A, Ishihara S, Yamane F. Optimal first coil selection to avoid aneurysmal recanalization in endovascular intracranial aneurysmal coiling. J Neurointerv Surg. 2018. 10: 50-4
8. Takao H, Ishibashi T, Saguchi T, Arakawa H, Ebara M, Irie K. Validation and initial application of a semiautomatic aneurysm measurement software: A tool for assessing volumetric packing attenuation. AJNR Am J Neuroradiol. 2014. 35: 721-6
9. Uchiyama N, Kida S, Nomura M, Hasegawa M, Yamashima T, Yamashita J. Significance of volume embolization ratio as a predictor of recanalization on endovascular treatment of cerebral aneurysms with guglielmi detachable coils. Interv Neuroradiol. 2000. 6: 59-63