- Departments of Neurosurgery, Hackensack University Medical Center, Hackensack, New Jersey, United States.
- Departments of Radiology, Hackensack University Medical Center, Hackensack, New Jersey, United States.
- Departments of Anesthesiology, Hackensack University Medical Center, Hackensack, New Jersey, United States.
- Departments of Operations, Hackensack University Medical Center, Hackensack, New Jersey, United States.
- Departments of Perioperative Services, Hackensack University Medical Center, Hackensack, New Jersey, United States.
Correspondence Address:
Hooman Azmi
Departments of Neurosurgery, Hackensack University Medical Center, Hackensack, New Jersey, United States.
DOI:10.25259/SNI-209-2019
Copyright: © 2019 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Hooman Azmi, Mary Gibbons, Michele C. DeVito, Mark Schlesinger, Jason Kreitner, Terri Freguletti, Joan Banovic, Donald Ferrell, Michael Horton, Sean Pierce, Patrick Roth. The interventional magnetic resonance imaging suite: Experience in the design, development, and implementation in a pre-existing radiology space and review of concepts. 19-Jun-2019;10:101
How to cite this URL: Hooman Azmi, Mary Gibbons, Michele C. DeVito, Mark Schlesinger, Jason Kreitner, Terri Freguletti, Joan Banovic, Donald Ferrell, Michael Horton, Sean Pierce, Patrick Roth. The interventional magnetic resonance imaging suite: Experience in the design, development, and implementation in a pre-existing radiology space and review of concepts. 19-Jun-2019;10:101. Available from: https://surgicalneurologyint.com/?post_type=surgicalint_articles&p=9383
Abstract
Background: Intraoperative magnetic resonance imaging (ioMRI) has led to significant advancements in neurosurgery with improved accuracy, assessment of the extent of resection, less invasive surgical alternatives, and real-time confirmation of targeting as well delivery of therapies. The costs associated with developing ioMRI units in the surgical suite have been obstacles to the expansion of their use. More recently, the development of hybrid interventional MRI (iMRI) units has become a viable alternative. The process of designing, developing, and implementing operations for these units requires the careful integration of environmental, technical, and safety elements of both surgical and MR practices. There is a paucity of published literature providing guidance for institutions looking to develop a hybrid iMRI unit, especially with a limited footprint in the radiology department.
Methods: The experience of designing, developing, and implementing an iMRI in a preexisting space for neurosurgical procedures at a single institution in light of available options and the literature is described.
Results: The development of the unit was accomplished through the engagement of a multidisciplinary team of stakeholders who utilized existing guidelines and recommendations and their own professional experience to address issues including physical layout, equipment selection, operations planning, infection control, and oversight/review, among others.
Conclusion: Successful creation of an iMRI program requires multidisciplinary collaboration in integrating surgical and MR practice. The authors’ aim is that the experience described in this article will serve as an example for facilities or neurosurgical departments looking to navigate the same process.
Keywords: Interventional magnetic resonance imaging design, Interventional magnetic resonance imaging, Intraoperative magnetic resonance imaging, Magnetic resonance imaging development
INTRODUCTION
Since the development of the first intraoperative magnetic resonance imaging (MRI) (ioMRI) units in the 1980s,[
The acuity and complexity inherent in both a surgical environment and MRI space necessitate a careful, comprehensive approach to the hybridization of the two in an interventional MRI (iMRI) suite.[
Despite a wide number of recommendations and guidelines from accrediting and professional organizations on ioMRI safety and design, there is a relative paucity of published reports describing the experience of planning and developing an iMRI unit and the challenges faced in the process, in particular, when designing this unit to fit into a preexisting space with a limited footprint. To the authors’ knowledge, only three such reports, those of Childs and Bruch at Yale-New Haven Hospital, White et al. in the UK, and Cherkashin et al. in Russia, have been published to date.[
METHODS
The integration of operating room specifications and optimal magnet function and safety requirements is complex. At our institution, this process was informed by utilizing a number of key resources, including the American College of Radiology (ACR) guidance document for safe MR Practice, the Association of Perioperative Registered Nurses’ “Successful Management of Risk in the Hybrid OR,” the magnet manufacturer’s recommendations/specifications, and the Facilities Guidelines Institute’s Guidelines for Design and Construction of Hospitals.[
RESULTS
Suite design
Physical layout
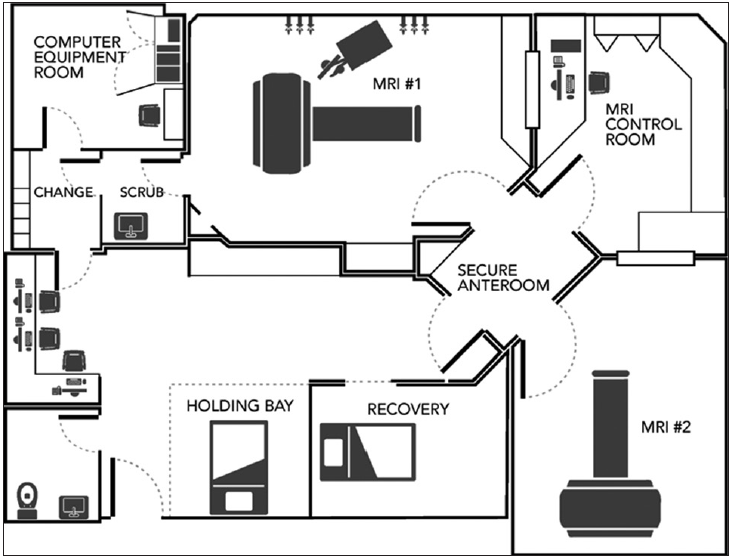
The limitations posed by designing the iMRI unit to conform to the pre-existing floor plan was our biggest challenge.. The existing MRI suite was designed with two imaging rooms with an adjoining control room and other necessary access areas. The redesign of the entire suite was both cost prohibitive and also would have rendered an inpatient magnet inoperable for a period of time, and this was simply not feasible. The best alternative was to reconfigure one of the rooms while staging the build and maintaining the other magnet functional. As such, the design had to incorporate all state requirements for an operating room build within our confined limitations including a secure antechamber, a scrub sink, a recovery area, and a holding area. Moreover, these requirements also had to be designed with strict adherence to MRI safety concerns. Multiple iterations of the design were reviewed, and in the end, what was submitted and ultimately approved is demonstrated in
In general, both ioMRI and iMRI suites can be configured with either mobile or stationary scanners. The former scanner type needing to be transported to and from the operating area, and the latter requiring the patient to moved into and out of the magnet. They each present their own set of challenges for costs, operating space, safety and surgical limitations, and expectations for the ratio of interventional versus diagnostic procedures. The advantages and disadvantages in these general approaches have been well documented in the literature.
These advantages are offset by certain detractors of this design. As ferromagnetic instruments are used for the surgery, they need to be removed when the magnet is entering the room. The transformation of the operating area to an MRI safe environment requires a high level of vigilance with multiple levels of safety checks prior to magnet entry. The more practical disadvantage, however, is the cost associated with this design. A standing 1.5T MRI scanner costs can range from 1 to 3 million US dollars. The cost of construction alone for the required ceiling rail system to move the multiton magnet into and out of the room is at least 1–2 times the cost of the scanner. This is in addition to the regular costs of an OR build-out.
This expense has limited the use of the mobile magnet to a few centers. The hybrid OR/MRI room has recently evolved to address this obstacle. Institutions interested in acquiring intraoperative MRI capabilities now have the option of building hybrid rooms with standing MRIs in the operating suite. The MRI room is adjacent to the OR, and patients are brought into the magnet room from the OR mid-surgery or at the end of the procedure when imaging is needed. When there is no need for intraoperative imaging, the MRI room can be used for diagnostic scans. While the discussion for these two options is relevant when designing a new building or complete OR suite, these choices were not available to us as we had to design our unit in a preexisting space within the radiology floorspace. The limitations enumerated at our institution allowed for a stationary second MRI scanner contiguous to the existing MRI suite. While this permitted cost savings in utilizing the existing control room, shielding, and the fringe field accountability, there were challenges in hybridizing the surgical and MRI environment within a space with such immutable parameters [
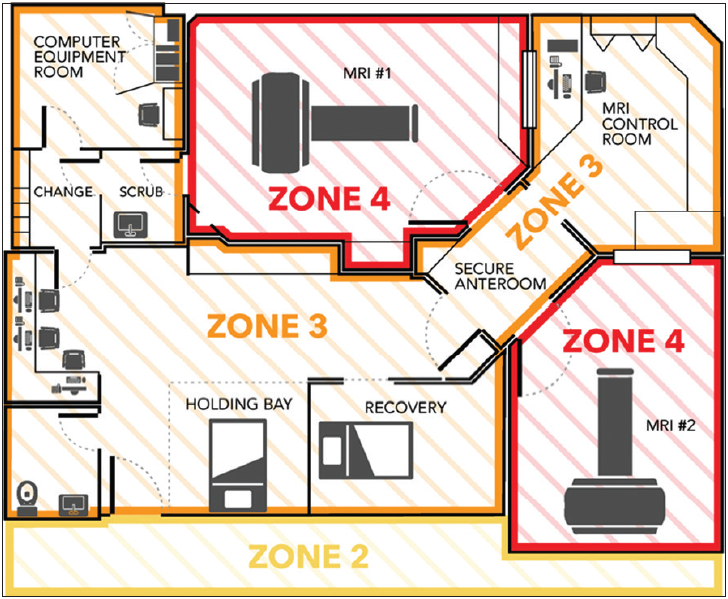
MRI zones
The ACR defines four critical zones relative to any MRI suite, which are outlined in
Table 2
MRI zones as defined by the ACR.[
Figure 2
Layout of the magnetic resonance imaging (MRI) suite with outlined MRI zones. Zone IV has two entrances, one from the secure anteroom in Zone III, adjacent to the control room, and another from the surgical scrub room. This direct access configuration minimizes the risk of the staff being contaminated when going through other areas.
Static field and radio-frequency (RF) screening
All MRIs must have RF screening provided by a Faraday cage for proper functioning.[
Air circulation and ventilation
Air curtain systems have been demonstrated to reduce air turbulence near the surgical field and potentially reduce contamination of the field by particulate matter. The iMRI room was designed with current recommendations and guides incorporating an air curtain system.[
Equipment decisions
Selection: Magnet and coil
The choice of scanner is arguably the most significant equipment decision in designing an interventional suite as it directly and indirectly will dictate the subsequent requirements of the design. At our institution, a 1.5T cylindrical bore diagnostic scanner (Magnetom AERA, Siemens, Erlangen, Germany) was chosen with a wide bore (70 cm). This decision was based on our prior experience with Siemens, bore size availability, familiarity of MR staff with the hardware and software, compatibility of the units with the existing IT setup, and the engineering of patient movements into and out of the scanner on the MRI bed.
The receiver coil used at our institution is a 4-channel flexible wrap-around coil that is iPAT-compatible (Flex Large 4, Siemens, Erlangen, Germany).
Selection: Surgical and anesthetic equipment
Our equipment selection was informed by the American Society for Testing and Materials (ASTM) designation for MRI equipment. ASTM categorizes MRI equipment safety. MRI safe designated equipment can be safely used within the 5-gauss line. MRI conditional equipment can be used within the 5-gauss line with certain parameters and MRI unsafe equipment should not be used in Zone IV.[
For our instruments, a standard neurosurgical MRI-safe instrument tray was created to include all instruments potentially needed in any procedure and was tested and approved by the MR Safety Officer (MRSO) before use in the iMRI. An MRI-safe titanium surgical drill (Aria Drill systems, Stryker, Kalamazoo, MI) was also purchased to be available for these procedures [
Selection: Lighting
Lighting is obviously an important element in optimizing the surgical staff’s visualization of both the surgical site and surgical equipment. In the iMRI suite, light-emitting diode sources were selected as they have been shown to reduce noise or image artifact.[
Operation planning
Access control
Access control is a critical issue in any MRI suite; however, it becomes even more important in an iMRI unit in which the presence of surgical personnel entering and exiting the suite adds to the overall volume of foot traffic and air turbulence. Entrance from Zone II into the secure anteroom (Zone III) is through a locked door which requires a security ID swipe, issued only to MRI staff, for opening. The only staff who are permitted unsupervised access into Zones III and IV are level 2 trained personnel. Level 2 certification requires completion of advanced MRI safety training. All Level 1 personnel, including anesthesiology and surgical staff, must be accompanied by Level 2 personnel and must defer to Level 2 while in Zone III or IV.
Safety training
A detailed description of the cross-training of both MR and surgical personnel is beyond the scope of this article, and the essential elements required have been previously outlined by Larson et al.[
All staff with access to the iMRI suite must undergo an MRI safety class with the MRSO. Ongoing competency is ensured by an annual refresher course and examination. The head of MRI technology serves as the procedure safety officer and is present for every surgical procedure, while the head clinical radiology nurse serves as the circulating nurse for each procedure.
All surgeons undergo MRI safety education supervised by the MRSO in addition to being proctored for their initial surgeries by the primary neurosurgeon.
Staff responsibilities and workflow
A patient’s journey for an interventional procedure begins in the Radiology Day Accommodation Room, which is in Zone I. Before transport of the patient to Zone IV, the patient is seen by the surgical and anesthesia teams and on completion of safety checklists.
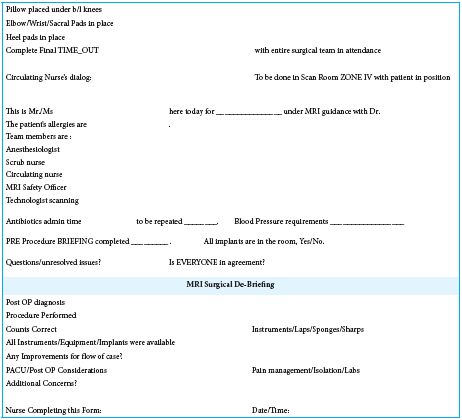
Safety checklists have long been proven to be a highly effective means of providing this monitoring for both MRI and surgical procedures and have become a standard of care.[
Infection control
Perhaps, one of the most challenging and yet critical issues related to the hybridization of the OR and MRI is that of infection prevention and control of the MRI environment. There are no formal recommendations from any professional or accrediting organization regarding cleaning protocol for an intraoperative MRI suite. The ACR guidance document on MR safe practice addresses the fact that the design of the suite should facilitate easy cleaning (e.g., surfaces that can be scrubbed, seamless flooring). It further states that cleaning staff that enters Zones III and IV must either be Level 2-trained or supervised by Level 2 staff in those areas, in addition to being thoroughly screened before entry into them.[
Emergencies
Emergencies in the iMRI can be broadly categorized into those that are primarily related to the patient and those that are primarily related to the magnet. Before initial use of the iMRI, a protocol for a cardiopulmonary emergency code had already been established. The protocol conforms to that recommended by the American Society of Anesthesiologists Task Force on Anesthetic Care for MRI.[
The most significant magnet-related emergency that can occur in an MRI is a quenching, in which the magnetic coils rapidly lose their absolute zero temperature leading to the elimination of the magnetic field. Helium escapes from the magnet’s cryogen bath when this occurs, and a venting system is required to direct the helium out of the suite to avoid atmospheric oxygen depletion and increased pressurization in Zone IV.[
Oversight/review
A critical aspect of the ongoing success of an iMRI is the continued review of practice and protocol by all stakeholders.[
The current checklist and briefing tool have been developed and expanded over the course of the past 5 years by incorporating the lessons learned at our institution, utilizing and modifying the currently available tools, and the consensus developed as a group to anticipate potential safety issues, in a best attempt to avoid these lapses. Fortunately, there has been no compromise of the safety of patients during these interventional procedures, and nearly 100 of which have been performed to date, suggesting that the safety protocol has served well. Nevertheless, the protocol continues to be assessed and reevaluated to ensure any gaps if remaining are addressed.
DISCUSSION
In their 2000 review, intraoperative magnetic resonance imaging and interventional magnetic resonance imaging: recommendations for A Safe Environment, Kettenbach et al. advised that there should be “a gradual progression in establishing an iMRI suite. The right team members, well thought-out architectural planning, and proper policy and training are an essential start. Human vigilance and enforcement of policy must be ongoing. For any new procedure, a dry run should be executed to identify pitfalls. A small core staff should be involved in the first cases… in expanding the services, the experienced core staff should train new staff members.”[
Integrating all of these elements, from architectural planning to staff cross-training, to achieve a fully functional ioMRI unit obviously requires considerable capital outlay and resource expenditure. This funding is often a significant barrier to a facility’s development of an ioMRI program. A seminal study by Hall et al.[
While such an analysis has not yet been performed at our institution, the realized benefits of having an iMRI, both for patients and the hospital, have been multifold. There has been a significant expansion of services in the neurosurgical department, including the ability to perform deep brain stimulation procedures with MRI guidance, laser ablation for tumors and medication-resistant epilepsy, minimally invasive brain biopsy for deep-seated tumors, and MR-guided direct drug delivery. Access to a separate iMRI has allowed for increased patient throughput in the conventional MRI suite which is consistently in high demand at our institution. In addition, other surgical specialties may begin utilizing the iMRI unit for other procedures such as prostate and breast biopsies, helping to maximize utilization and further achieve a return on investment.
CONCLUSION
As MRI guidance technology continues to play a more significant role in surgical procedures, the need to hybridize the MR and surgical environment will increasingly become an issue for health-care facilities. Institutions will need to assess their surgical needs, along with their financial and physical limitations, to determine what scanning configuration (i.e., use of a mobile scanner brought into the OR or a stationary scanner in a dedicated unit) best suits them. Careful consideration of the differences among these configurations in light of an institution’s needs and limitations can inform their design decision. Ongoing multidisciplinary collaboration in the development and subsequent implementation of that design, in addition to reliance on available guidelines, recommendations, and other facilities’ experiences, can result in a highly efficient and safe hybridized MR/OR environment.
Financial support and sponsorship
Nil.
Conflicts of interest
The corresponding author has a consulting agreement with Medtronic Inc and Abbott Scientific. All of the other authors certify that they have no conflicts of interest.
Acknowledgment
The authors would like to thank Helen Boehm Johnson, MD, and Denise Graham for their invaluable assistance in writing and editing this manuscript.
References
1. 2. 3. Chabardes S, Isnard S, Castrioto A, Oddoux M, Fraix V, Carlucci L. Surgical implantation of STN-DBS leads using intraoperative MRI guidance: Technique, accuracy, and clinical benefit at 1-year follow-up. Acta Neurochir (Wien). 2015. 157: 729-737 4. Cherkashi NM, Berezina NA, Serov AV. Safety management for MRI-guided interventions. Investig Magn Reson Imaging. 2016. 20: 152-7 5. Childs S, Bruch P. Successful management of risk in the hybrid OR. AORN J. 2015. 101: 223-34 6. Drane DL, Loring DW, Voets NL, Price M, Ojemann JG, Willie JT. Better object recognition and naming outcome with MRI-guided stereotactic laser amygdalohippocampotomy for temporal lobe epilepsy. Epilepsia. 2015. 56: 101-13 7. Kanal E, Barkovich AJ, Bell C, Borgstede JP, Bradley WG. ACR guidance document on MR safe practices: 2013. J Magn Reson Imaging. 2013. 37: 501-30 8. . Available from: https://www.fgiguidelines.org/guidelines/2014-fgi-guidelines/ [Last accessed on 2017 May 23]. 9. Hall WA, Kowalik K, Liu H, Truwit CL, Kucharezyk J. Costs and benefits of intraoperative MR-guided brain tumor resection. Acta Neurochir Suppl. 2003. 85: 137-42 10. Hawasli AH, Bagade S, Shimony JS, Miller-Thomas M, Leuthardt EC. Magnetic resonance imaging-guided focused laser interstitial thermal therapy for intracranial lesions: Single-institution series. Neurosurgery. 2013. 73: 1007-17 11. . Available from: https://www.cms.gov/Medicare/Provider-Enrollment-and-Certification/SurveyCertificationGenInfo/Downloads/Survey-and-Cert-Letter-15-12-Attachment-1.pdf [Last accessed on 2017 Nov 05]. 12. Kettenbach J, Kacher DF, Kanan AR, Rostenberg B, Fairhurst J, Stadler A. Intraoperative and interventional MRI: Recommendations for a safe environment. Minim Invasive Ther Allied Technol. 2006. 15: 53-64 13. Kettenbach J, Kacher DF, Koskinen SK, Silverman SG, Nabavi A, Gering D. Interventional and intraoperative magnetic resonance imaging. Annu Rev Biomed Eng. 2000. 2: 661-90 14. Koenigshofer D.editorsASHRAE HVAC Design Manual for Hospitals and Clinics. Atlanta, GA: W. Stephen Comstoch; 2003. p. 15. Larson PS, Starr PA, Bates G, Tansey L, Richardson RM, Martin AJ. An optimized system for interventional magnetic resonance imaging-guided stereotactic surgery: Preliminary evaluation of targeting accuracy. Neurosurgery. 2012. 70: 95-103 16. Larson PS, Vadivelu S, Azmi-Ghadimi H, Nichols A, Fauerbach L, Johnson HB. Neurosurgical laser ablation and MR thermometry: Risks of multisite workflow pattern. J Healthc Risk Manag. 2017. 36: 7-18 17. Larson PS, Willie JT, Vadivelu S, Azmi-Ghadimi H, Nichols A, Fauerbach LL. MRI-guided stereotactic neurosurgical procedures in a diagnostic MRI suite: Background and safe practice recommendations. J Healthc Risk Manag. 2017. 37: 31-9 18. Medvid R, Ruiz A, Komotar RJ, Jagid JR, Ivan ME, Quencer RM. Current applications of MRI-guided laser interstitial thermal therapy in the treatment of brain neoplasms and epilepsy: A radiologic and neurosurgical overview. AJNR Am J Neuroradiol. 2015. 36: 1998-2006 19. Memarzadeh F. Comparison of operating room ventilation systems in the protection of the surgical suite. ASHRAE Trans. 2002. 108: 4549-62 20. Mislow JM, Golby AJ, Black PM. Origins of intraoperative MRI. Neurosurg Clin N Am. 2009. 20: 137-46 21. Mohyeldin A, Elder JB. Stereotactic biopsy platforms with intraoperative imaging guidance. Neurosurg Clin N Am. 2017. 28: 465-75 22. . Available from: https://www.cfm.va.gov/til/dGuide/dgMRI.pdf [Last accessed on 2017 Sep 27]. 23. Ostrem JL, Galifianakis NB, Markun LC, Grace JK, Martin AJ, Starr PA. Clinical outcomes of PD patients having bilateral STN DBS using high-field interventional MR-imaging for lead placement. Clin Neurol Neurosurg. 2013. 115: 708-12 24. Rennert RC, Carroll KT, Ali MA, Hamelin T, Chang L, Lemkuil BP. Safety of stereotactic laser ablations performed as treatment for glioblastomas in a conventional magnetic resonance imaging suite. Neurosurg Focus. 2016. 41: E7- 25. Ridgeway J, Plein S, Greenwood J, Ridgeway J.editors. The MRI environment. Cardiovascular MR Manual. New York: Springer; 2011. p. 11-5 26. Scheer JK, Hamelin T, Chang L, Lemkuil B, Carter BS, Chen CC. Real-time magnetic resonance imaging-guided biopsy using smartFrame® stereotaxis in the setting of a conventional diagnostic magnetic resonance imaging suite. Oper Neurosurg (Hagerstown). 2017. 13: 329-37 27. Tatsui CE, Nascimento CN, Suki D, Amini B, Li J, Ghia AJ. Image guidance based on MRI for spinal interstitial laser thermotherapy: Technical aspects and accuracy. J Neurosurg Spine. 2017. 26: 605-12 28. White MJ, Thornton JS, Hawkes DJ, Hill DL, Kitchen N, Mancini L. Design, operation, and safety of single-room interventional MRI suites: Practical experience from two centers. J Magn Reson Imaging. 2015. 41: 34-43 29. Willie JT, Laxpati NG, Drane DL, Gowda A, Appin C, Hao C. Real-time magnetic resonance-guided stereotactic laser amygdalohippocampotomy for mesial temporal lobe epilepsy. Neurosurgery. 2014. 74: 569-84 30. . Available from: http://www.who.int/patientsafety/safesurgery/checklist/en. [Last accessed on 2017 Aug 16].