- Department of NeuroScience/Neurosurgery, Winthrop University Hospital, Mineola, New York 11501, USA
Correspondence Address:
Nancy E. Epstein
Department of NeuroScience/Neurosurgery, Winthrop University Hospital, Mineola, New York 11501, USA
DOI:10.4103/2152-7806.166887
Copyright: © 2015 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Epstein NE. Timing and prognosis of surgery for spinal epidural abscess: A review. Surg Neurol Int 08-Oct-2015;6:
How to cite this URL: Epstein NE. Timing and prognosis of surgery for spinal epidural abscess: A review. Surg Neurol Int 08-Oct-2015;6:. Available from: http://surgicalneurologyint.com/surgicalint_articles/timing-and-prognosis-of-surgery-for-spinal-epidural-abscess/
Abstract
Background:The nonsurgical versus surgical management of spinal epidural abscesses (SEAs) remains controversial. Even with the best preoperative screening for multiple risk factors, high nonoperative failure rates are attended by considerable morbidity (e.g., irreversible paralysis) and mortality. Therefore, the focus remains on early surgery.
Methods:Most papers promote early recognition of the clinical triad (e.g., fever [50%], spinal pain [92–100%], and neurological deficits [47%]) for SEA. They also identify SEA-related risk factors for choosing nonsurgical versus surgical approaches; advanced age (>65 or 80), diabetes (15–30%), cancer, intravenous drug abuse (25%), smoking (23%), elevated white blood cell count (>12.5), high C-reactive protein >115, positive blood cultures, magnetic resonance imaging/computed tomographic documented cord compression, and significant neurological deficits (e.g., 19–45%).
Results:Surgical options include: decompressions, open versus minimally invasive biopsy/culture/irrigation, or fusions. Up to 75% of SEA involve the thoracolumbar spine, and 50% are located ventrally. Wound cultures are positive in up to 78.8% of cases and are often (60%) correlated with positive blood cultures. The most typical offending organism is methicillin resistant Staphylococcus aureus, followed by methicillin sensitive S. aureus. Unfortunately, the failure rates for nonoperative treatment of SEA remain high (e.g., 41–42.5%), contributing to significant morbidity (22% risk of permanent paralysis), and mortality (3–25%).
Conclusion:The vast majority of studies advocated early surgery to achieve better outcomes for treating SEA; this avoids high failure rates (41–42.5%) for nonoperative therapy, and limits morbidity/mortality rates.
Keywords: Medical management, neurological deficit, open surgery: Minimally invasive surgery, paraplegia, spinal epidural abscess, surgical decompression, timing
INTRODUCTION
The nonsurgical versus surgical management of spinal epidural abscesses (SEAs) remain controversial. Even the best preoperative screening for multiple risk factors (clinical, laboratory, and radiographic) may result in high nonoperative failure rates, with significant morbidity/mortality. Most SEA patients, ranging in age from their mid 50s to mid 60s, present with the classical triad of fever (50%), spinal pain (47.1–100%), and weakness (19–55.8%).[
The timing of SEA surgery is typically categorized as early if performed at/within <24 h (mean 11.2 h) or late if performed after >24 h (mean 59 h).[
This review provides an overview of the presentation, clinical, radiographic, and surgical management of SEA [Tables
SPINAL EPIDURAL ABSCESS DUE TO EPIDURAL STEROID INJECTIONS
Cervical meningomyelitis following lumbar epidural steroid injection: Case study
Lee et al. noted that the greater number of lumbar epidural steroid injection (ESI) are correlated with higher complication rates.[
Spinal and paraspinal fungal infections (abscess, phlegmon, arachnoiditis, and osteomyelitis) associated with the contaminated methylprednisolone epidural steroid injection
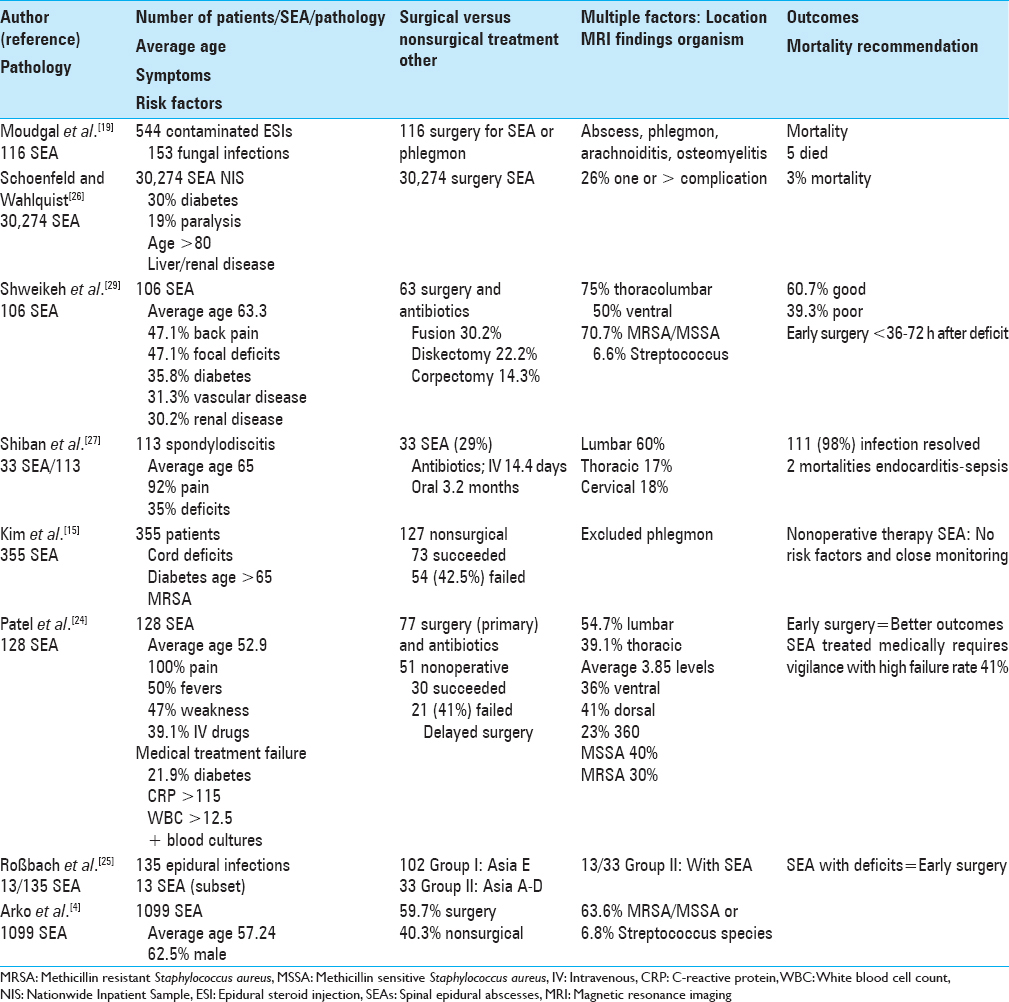
Moudgal et al. tracked patients who developed spinal or paraspinal fungal infections due to the injections of exserohilum-contaminated methylprednisolone (Michigan Department of Community Health, electronic medical records (EMRs), and MRI reports).[
TIMING OF SURGERY FOR SPINAL EPIDURAL ABSCESSES
Optimal timing of surgery for spinal epidural abscess within 24 h
Utilizing meta-analyses, Ghobrial et al. focused on the optimal timing of surgery for SEA or other spinal infections (e.g., osteomyelitis, osteodiscitis, and spondylodiscitis).[
Preoperative neurological status best determined outcome for bacterial spinal epidural abscess
In their literature review of 40 SEA articles, Shweikeh et al. emphasized the import of early recognition of bacterial SEA as the preoperative neurological status determined the best postoperative outcome.[
EPIDEMIOLOGY, DIAGNOSIS, AND MANAGEMENT OF OSTEOMYELITIS AND/OR SPINAL EPIDURAL ABSCESSES SUMMARIES OF STUDIES INVOLVING >100 CASES
Mortality, complications, and charges for spinal epidural abscesses in 30,274 patients from a Nationwide Inpatient Sample Study
Schoenfeld and Wahlquist looked at the Nationwide Inpatient Sample of 30,274 individuals (2006–2011) in order to predict the morbidity/mortality (in-hospital: Failure to rescue from a complication) and total charges for patients undergoing SEA surgery [
Medical and surgical management of 1099 spinal epidural ascesses: A systematic review
Arko et al. noted that SEA have typically (particularly prior to 1999) been managed with emergent surgery/decompression and antibiotic therapy [
Independent risk factors help predict failure of nonoperative management of 355 spinal epidural abscesses
Kim et al. stipulated that medical management may be successful in a subset of patients with SEA and that all do not require surgery [
Hematogenous vertebral osteomyelitis (not spinal epidural abscess) caused by Gram-negative bacteria in 313 cases
In a study involving three tertiary-care centers over a 7 year period, Park et al. found that Gram Negative bacteria (GNB) were responsible for 65 (20.8%) of the 313 hematogenously spread cases of vertebral osteomyelitis (HVO).[
Surgical treatment of spondylodiscitis in 135 patients with the neurological deficits: Predicting clinical outcomes
Roßbach et al. successfully utilized preoperative neurological deficits in 135 patients with CT/MRI documented SEA (13 patients) or spondylodiscitis (122 patients) to predict clinical outcomes [
High failure rates with nonoperative management of 128 spinal epidural abscesses; utility of four major risk factors
Patel et al. retrospectively evaluated how multiple risk factors impacted outcomes of SEA managed medically or with antibiotics combined with surgery [
Retrospective evaluation of surgically treated spondylodiscitis in 113 patients
Shiban et al. retrospectively analyzed 113 consecutive patients who had surgical decompression/instrumentation performed for spondylodiscitis (2006 and 2010) at one institution; SEA were noted in 33 patients (29%) [
Institutional series with bacterial spinal epidural abscesses in 106 patients
Shweikeh et al. evaluated bacterial SEA encountered at one institution over an 11-year period (2001–2012) utilizing the electronic medical record (EMR) while also reviewing the pertinent literature (e.g., PubMed 2000–2011) [
SUMMARIES OF SPINAL EPIDURAL ABSCESS CASES INVOLVING 40 TO <100 CASES
Surgery versus medical management of 82 spinal epidural abscesses in patients 50 years of age or older
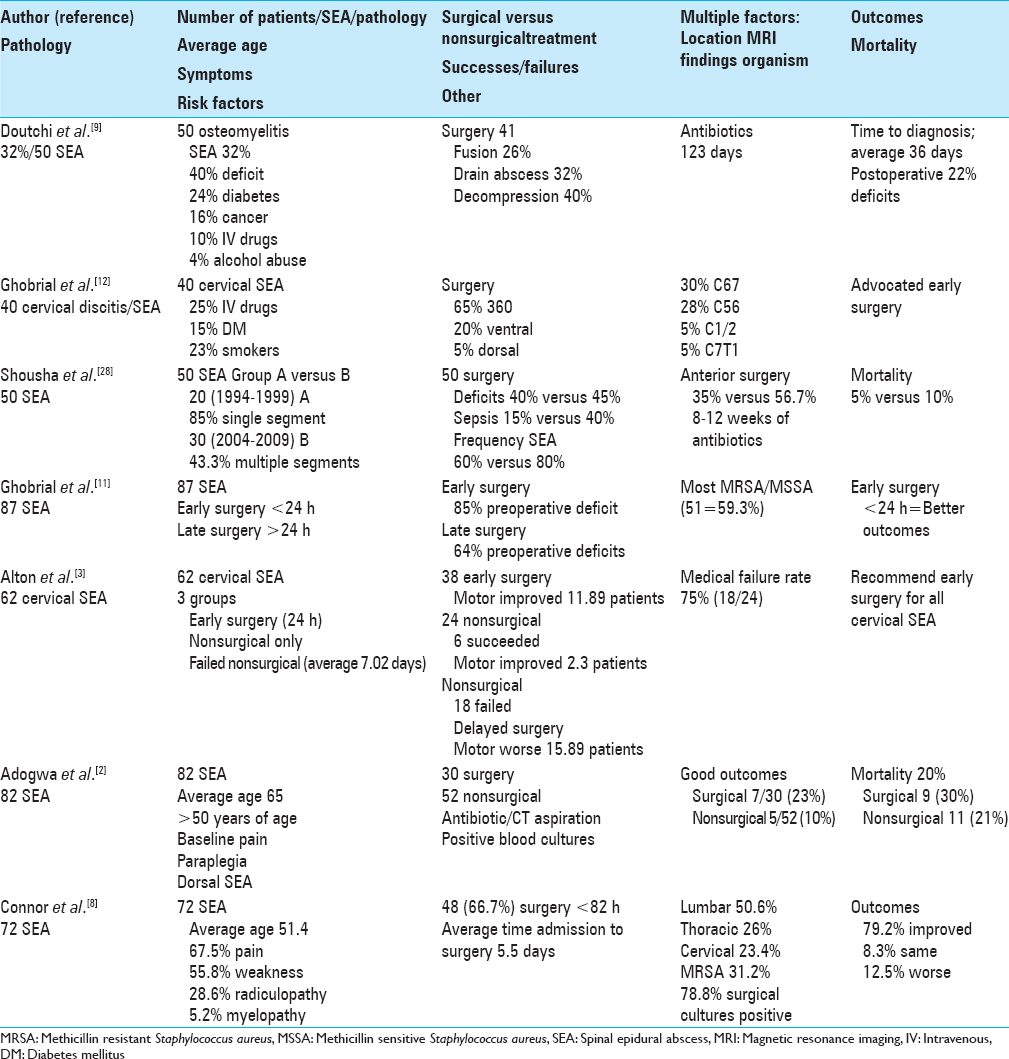
Adogwa et al. analyzed SEAs in 82 patients over the age of 50 treated at one institution (15 years) (1999 and 2013); 30 [37%] had surgery, while 52 [63%] were treated with antibiotics/CT-guided aspiration or antibiotics based on blood cultures alone.[
Operative versus nonoperative management of 72 spinal epidural abscess
Connor et al. retrospectively reviewed nonoperative versus operative treatment of SEA [
Poorer outcomes for managing 62 cervical spinal epidural abscesses without versus with early surgery
Alton et al. retrospectively evaluated the risk factors associated with the neurological outcomes for 62 patients with cervical SEA treated either with antibiotics alone (e.g. originally neurologically intact (Group 1) or with delayed deterioration (Group 3)) versus early surgery (Group 2) [
Epidemiology of vertebral osteomyelitis in 50 patients
Doutchi et al. evaluated the morbidity of vertebral osteomyelitis in 50 patients over a 5-year period [
Management of 50 patients with cervical spondylodiscitis/spinal epidural abscesses plus literature review
Shousha et al. evaluated the surgical treatment (debridement/reconstruction and antibiotics for 8–12 postoperative weeks) and outcomes for 50 consecutive patients with cervical spondylodiscitis/SEA managed at one institution [
Surgery for 40 cervical spinal epidural abscesses: Increased circumferential management
Ghobrial et al. retrospectively analyzed 40 consecutive patients from multiple studies (1997–2011) presenting with cervical SEA.[
SUMMARIES OF STUDIES INVOLVING 1 TO <40 CASES OF SPINAL EPIDURAL ABSCESSES
Treatment of primary pyogenic spinal epidural abscess: Import of timing of surgery in 23 patients
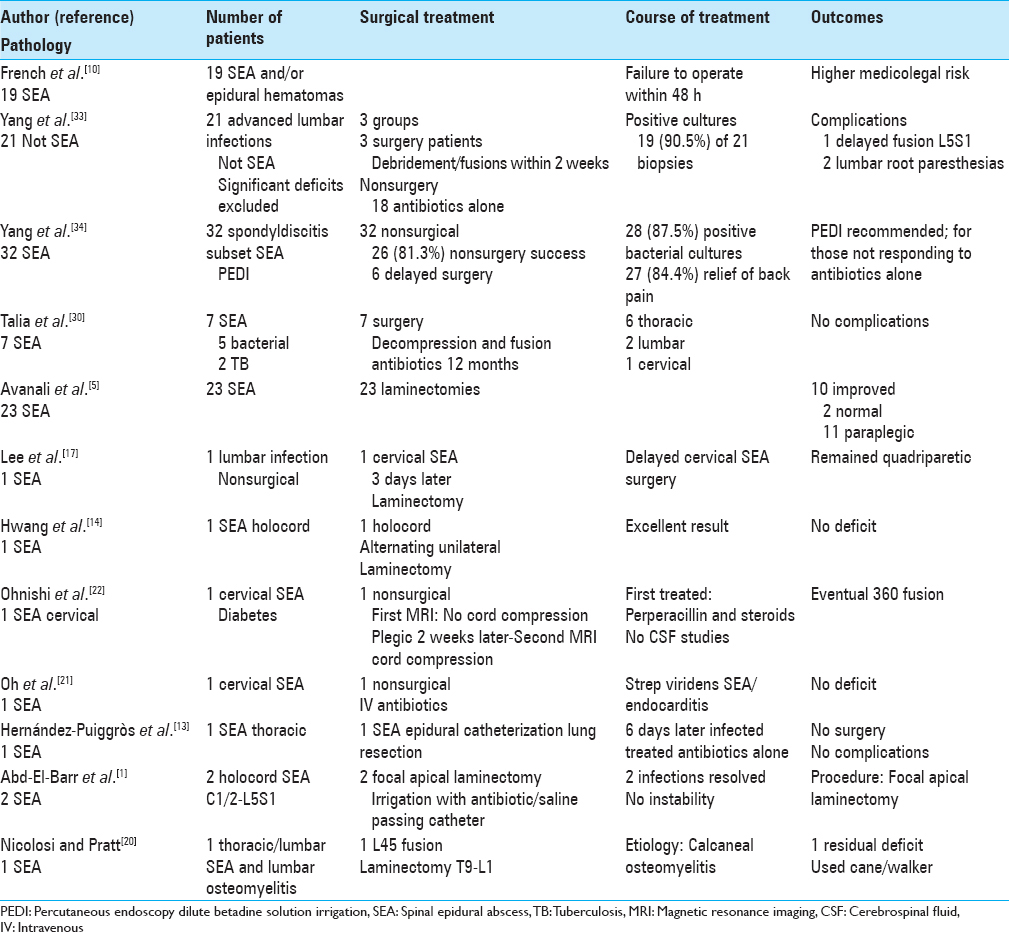
Avanali et al. noted that SEA, even when diagnosed and treated in a timely fashion, often leads to irretrievable neurological deficits [
Safety of instrumentation/fusion with surgical debridement of osteomyelitis for spinal epidural abscesses in 7 cases
Following radical debridement, Talia et al. evaluated the safety/efficacy of single-stage instrumentation/fusion (6 thoracic, 2 lumbar, and 1 cervical infections) in 9 patients (5 diabetics) with spinal infections (e.g., vertebral osteomyelitis or SEA).[
Cervical osteomyelitis/thoracic myelitis and meningitis in a diabetic patient
Ohnishi et al. reported on a 45-year-old male with untreated diabetes, who presented paraparetic after 2 weeks of fever, back pain, and what was diagnosed as flu/gastroenteritis [
Infectious spondylodiscitis, epidural phlegmon, and psoas abscess in a diabetic
In a 63-year-old diabetic male with back pain, hyperpyrexia, and right calcaneal osteomyelitis, Nicolosi and Pratt reported hematogenous spread of MRSA resulting in a right psoas muscle abscess, L4–L5 osteomyelitis, and an SEA extending from T9–S1 [
Cervical spinal epidural abscess due to bacterial endocarditis with streptococcus viridans: Single case study
Oh et al. presented a patient with a cervical SEA due to streptococcus viridans endocarditis that was successfully treated with IV antibiotics for 8 weeks without sequelae [
Successful antibiotic treatment alone of thoracic spinal epidural abscess after epidural catheterization; single case study
Hernández-Puiggròs et al. presented the case of a thoracic epidural abscess forming following epidural catheter placement for a patient undergoing lobar lung resection. Six days after surgery the patient complained of “pulsatile” back pain accompanied by purulence at the catheter site [
DRAINAGE OF SPINAL EPIDURAL ABSCESSES WITH MINIMALLY INVASIVE SURGERY
Minimally invasive surgery endoscopy to treat 32 cases of lumbar infectious spondylitis
In 32 patients with lumbar single-level infectious spondylodiscitis, postoperative infectious spondylodiscitis, and SEA, Yang et al. evaluated the efficacy of percutaneous endoscopic epidural debridement using “dilute betadine solution irrigation (percutaneous endoscopy dilute betadine solution irrigation [PEDI])” (2005 to July 2010) [
Percutaneous endoscopic lavage/drainage for 21 patients with lumbar infectious spondylodiscitis
Yang et al. retrospectively assessed the safety/efficacy of percutaneous endoscopic lavage and drainage (PELD) for 21 patients with infectious spondylitis to help determine whether MIS (PELD) would suffice and avoid open surgery [
Minimally invasive spine surgery in spinal infections
Verdú-López et al. noted that new MIS techniques might address spine infections.[
Holocord spinal epidural abscess treated with alternating unilateral approach (skip laminectomy): Single case study
In a 51 year old with bilateral lower extremity weakness attributed to a C4–S1 holocord SEA, Hwang et al. utilized a unilateral level-skipping laminectomy technique that effectively preserved stability [
Holocord spinal epidural abscesses treated with “apical laminectomies:” 2 case studies
Abd-El-Barr et al. (JNS 2015) reported 2 cases in which apical laminectomies (e.g., an alternative to skip laminectomies) provided irrigation/drainage of extensive C1–C2 to L5–S1 SEA [
REVIEW ARTICLES [TABLE 4]
Treatment algorithm for timing of surgery versus nonsurgical management for spinal epidural abscesses
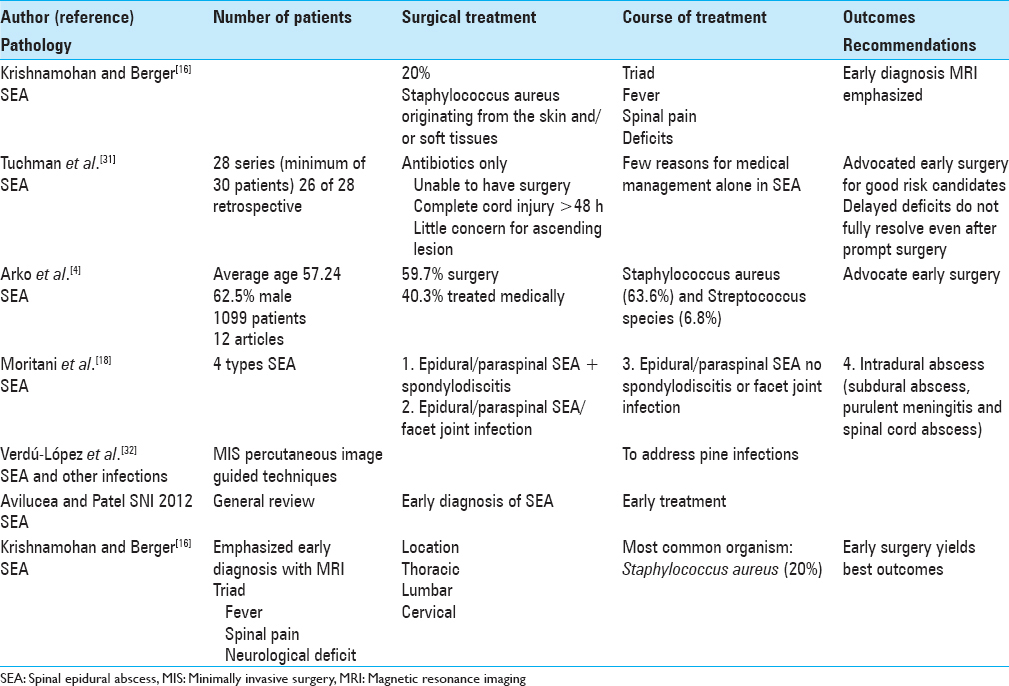
Tuchman et al. emphasized that although the typical treatment for SEA includes emergency surgery and protracted antibiotic therapy, some series with very carefully selected patients have successfully utilized antibiotic treatment alone [
Epidural infection: Is it an abscess?
Avilucea and Patel assessed the literature/PubMed regarding organisms identified, and the diagnosis/management of SEA [
Vertebral osteomyelitis and spinal epidural abscess: Evidence-based review
Boody et al. noted that spinal infections are typically associated with significant morbidity and mortality rates.[
A review of spinal epidural abscesses: Early diagnosis with magnetic resonance imaging studies
Krishnamohan and Berger emphasized that for SEA, early diagnosis with MRI studies, and surgical management yielded the best outcomes [
Pyogenic and nonpyogenic spinal infections diagnosed with diffusion-weighted magnetic resonance imaging: Abscesses and pus collections
Although Moritani et al. noted that conventional MRI is typically utilized to diagnose SEA, in fact, diffusion-weighted imaging (DWI) provides earlier and more accurate detection of abscess and pus collections within the spine along with an excellent means of following the efficacy of treatment.[
MEDICOLEGAL CASES INVOLVING SPINAL EPIDURAL HEMATOMA AND SPINAL EPIDURAL ABSCESS FAVORING EARLY SURGERY
French et al. discussed the fact that spinal epidural hematoma and SEA, although infrequently encountered constitute neurosurgical emergencies.[
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Abd-El-Barr MM, Bi WL, Bahluyen B, Rodriguez ST, Groff MW, Chi JH. Extensive spinal epidural abscess treated with “apical laminectomies” and irrigation of the epidural space: Report of 2 cases. Spine. 2015. 22: 318-23
2. Adogwa O, Karikari IO, Carr KR, Krucoff M, Ajay D, Fatemi P. Spontaneous spinal epidural abscess in patients 50 years of age and older: A 15-year institutional perspective and review of the literature: Clinical article. J Neurosurg Spine. 2014. 20: 344-9
3. Alton TB, Patel AR, Bransford RJ, Bellabarba C, Lee MJ, Chapman JR. Is there a difference in neurologic outcome in medical versus early operative management of cervical epidural abscesses?. Spine J. 2015. 15: 10-7
4. Arko L, Quach E, Nguyen V, Chang D, Sukul V, Kim BS. Medical and surgical management of spinal epidural abscess: A systematic review. Neurosurg Focus. 2014. 37: E4-
5. Avanali R, Ranjan M, Ramachandran S, Devi BI, Narayanan V. Primary pyogenic spinal epidural abscess: How late is too late and how bad is too bad?- A study on surgical outcome after delayed presentation. Br J Neurosurg. 2015. p. 1-6
6. Avilucea FR, Patel AA. Epidural infection: Is it really an abscess?. Surg Neurol Int. 2012. 3: S370-6
7. Boody BS, Jenkins TJ, Maslak J, Hsu WK, Patel AA. Vertebral osteomyelitis and spinal epidural abscess: An evidence-based review. J Spinal Disord Tech. 2015. 28: E316-27
8. Connor DE, Chittiboina P, Caldito G, Nanda A. Comparison of operative and nonoperative management of spinal epidural abscess: A retrospective review of clinical and laboratory predictors of neurological outcome. J Neurosurg Spine. 2013. 19: 119-27
9. Doutchi M, Seng P, Menard A, Meddeb L, Adetchessi T, Fuentes S. Changing trends in the epidemiology of vertebral osteomyelitis in Marseille, France. New Microbes New Infect. 2015. 7: 1-7
10. French KL, Daniels EW, Ahn UM, Ahn NU. Medicolegal cases for spinal epidural hematoma and spinal epidural abscess. Orthopedics. 2013. 36: 48-53
11. Ghobrial GM, Beygi S, Viereck MJ, Maulucci CM, Sharan A, Heller J. Timing in the surgical evacuation of spinal epidural abscesses. Neurosurg Focus. 2014. 37: E1-
12. Ghobrial GM, Viereck MJ, Margiotta PJ, Beygi S, Maulucci CM, Heller JE. Surgical management in 40 consecutive patients with cervical spinal epidural abscesses: Shifting towards circumferential treatment. Spine (Phila Pa 1976). 2015. 40: E949-53
13. Hernández-Puiggròs P, Montes A, Solano A, Aguilar JL, Conesa G, Horcajada JP. Conservative treatment of an epidural abscess after thoracic epidural catheterization. A A Case Rep. 2014. 3: 162-5
14. Hwang R, Yung BH, Sedney C, Miele VJ. Treatment of holocord spinal epidural abscess via alternating side unilateral approach for bilateral laminectomy. W V Med J. 2015. 111: 14, 16-8
15. Kim SD, Melikian R, Ju KL, Zurakowski D, Wood KB, Bono CM. Independent predictors of failure of nonoperative management of spinal epidural abscesses. Spine J. 2014. 14: 1673-9
16. Krishnamohan P, Berger JR. Spinal epidural abscess. Curr Infect Dis Rep. 2014. 16: 436-
17. Lee Y, Kim JS, Kim JY. Cervical meningomyelitis after lumbar epidural steroid injection. Ann Rehabil Med. 2015. 39: 504-7
18. Moritani T, Kim J, Capizzano AA, Kirby P, Kademian J, Sato Y. Pyogenic and non-pyogenic spinal infections: Emphasis on diffusion-weighted imaging for the detection of abscesses and pus collections. Br J Radiol. 2014. 87: 20140011-
19. Moudgal V, Singal B, Kauffman CA, Brodkey JA, Malani AN, Olmsted RN. Spinal and paraspinal fungal infections associated with contaminated methylprednisolone injections. Open Forum Infect Dis. 2014. 1: ofu022-
20. Nicolosi N, Pratt C. Infectious spondylodiscitis, epidural phlegmon, and psoas abscess complicating diabetic foot infection: A case report. J Foot Ankle Surg. 2014. p.
21. Oh JS, Shim JJ, Lee KS, Doh JW. Cervical epidural abscess: Rare complication of bacterial endocarditis with Streptococcus viridans: A case report. Korean J Spine. 2015. 12: 22-5
22. Ohnishi Y, Iwatsuki K, Ishida S, Yoshimine T. Cervical osteomyelitis with thoracic myelitis and meningitis in a diabetic patient. Clin Med Insights Case Rep. 2015. 8: 37-40
23. Park KH, Cho OH, Jung M, Suk KS, Lee JH, Park JS. Clinical characteristics and outcomes of hematogenous vertebral osteomyelitis caused by gram-negative bacteria. J Infect. 2014. 69: 42-50
24. Patel AR, Alton TB, Bransford RJ, Lee MJ, Bellabarba CB, Chapman JR. Spinal epidural abscesses: Risk factors, medical versus surgical management, a retrospective review of 128 cases. Spine J. 2014. 14: 326-30
25. Roßbach BP, Niethammer TR, Paulus AC, Melcher C, Birkenmaier C, Jansson V. Surgical treatment of patients with spondylodiscitis and neurological deficits caused by spinal epidural abscess (SEA) is a predictor of clinical outcome. J Spinal Disord Tech. 2014. 27: 395-400
26. Schoenfeld AJ, Wahlquist TC. Mortality, complication risk, and total charges after the treatment of epidural abscess. Spine J. 2015. 15: 249-55
27. Shiban E, Janssen I, Wostrack M, Krieg SM, Ringel F, Meyer B. A retrospective study of 113 consecutive cases of surgically treated spondylodiscitis patients.A single-center experience. Acta Neurochir (Wien). 2014. 156: 1189-96
28. Shousha M, Heyde C, Boehm H. Cervical spondylodiscitis: Change in clinical picture and operative management during the last two decades.A series of 50 patients and review of literature. Eur Spine J. 2015. 24: 571-6
29. Shweikeh F, Saeed K, Bukavina L, Zyck S, Drazin D, Steinmetz MP. An institutional series and contemporary review of bacterial spinal epidural abscess: Current status and future directions. Neurosurg Focus. 2014. 37: E9-
30. Talia AJ, Wong ML, Lau HC, Kaye AH. Safety of instrumentation and fusion at the time of surgical debridement for spinal infection. J Clin Neurosci. 2015. 22: 1111-6
31. Tuchman A, Pham M, Hsieh PC. The indications and timing for operative management of spinal epidural abscess: Literature review and treatment algorithm. Neurosurg Focus. 2014. 37: E8-
32. Verdú-López F, Vanaclocha-Vanaclocha V, Gozalbes-Esterelles L, Sánchez-Pardo M. Minimally invasive spine surgery in spinal infections. J Neurosurg Sci. 2014. 58: 45-56
33. Yang SC, Chen WJ, Chen HS, Kao YH, Yu SW, Tu YK. Extended indications of percutaneous endoscopic lavage and drainage for the treatment of lumbar infectious spondylitis. Eur Spine J. 2014. 23: 846-53
34. Yang SC, Fu TS, Chen HS, Kao YH, Yu SW, Tu YK. Minimally invasive endoscopic treatment for lumbar infectious spondylitis: A retrospective study in a tertiary referral center. BMC Musculoskelet Disord. 2014. 15: 105-






Joseph Grossman
Posted October 22, 2015, 4:02 pm
I don’t know if you are continuing to collect data for any ongoing study, but in the event that you are, I thought I’d share my story with you.
I am the father of two teenaged children. In April of 2014, I was 46 years old and in the best physical condition of my life. I was training for my second degree black belt in Karate. I had no risk factors whatsoever for SEA. I did not have diabetes, renal problems, vascular disease or cancer. I did not have any history of smoking, alcohol abuse or intravenous drug use. I had no history of any back pain. I had an open reduction and internal fixation for a fractured right radius one year earlier and have a plate and screws installed, but within a couple of months this had healed uneventfully and I recovered fully.
After working out one day in April of 2014, I suddenly developed numbness in my calves which, within a couple of hours, had turned into lower back pain. Over the next 10 days, the pain became excruciating and was refractory to muscle relaxants, analgesics and even strong narcotics such as Percocets. I was unable to even transfer without assistance. I did not have any fever and had no neurological deficits, although I did have profuse sweating every time I slept. The first hospital I attended concluded that absent any risk factors or neurological symptoms, a MRI was not indicated and they sent me away. 10 days after the onset of symptoms, I had an MRI without contrast which showed abnormal epidural soft tissues extending from L1 through S1 as well as extensive abnormal and infiltration of the paraspinal muscles and fascial planes with fluid-like collections in the paraspinal muscles. There was a questionable abdominal mass, as well as diffuse decreased T1 signal of the lumbar spine. I then had an MRI with contrast which showed a large SEA. Cultures were positive for MSSA. I was admitted to hospital and developed septicemia. I underwent a laminectomy from T12-S1. a mediastinal exploration via a cervical incision due to a questionable mass pushing on my trachea. I remained in hospital for 13 days.
I had a PICC line inserted into my arm for eight weeks following my discharge from TWH. It infused cloxacillin at two hour intervals six times a day. After the PICC line was removed in late July, I continued to take heavy doses of oral cloxacillin but then developed a form of reactive arthritis which resulted in widespread swollen and painful joints. This fortunately cleared up after approximately six weeks. The oral cloxacillin was eventually discontinued in late August, approximately 14 weeks post-surgery.
I am very fortunate that I have no neurological issues. Other than some periodic stiffness in the lumbar spine which does not result in any interference with my ADLs, I have achieved essentially a full recovery. I returned to my Karate training and was able to get my second degree black belt.
To this day, neither I, my treating neurosurgeon nor my infectious disease specialist has any idea how I contracted the infection. I had no open cuts and there was no obvious portal of entry.
I would be glad to answer any questions you may have.
Regards,
JMG
PHILIP
Posted November 25, 2018, 6:12 pm
MY CASE IS IDENTICAL MINE WAS T12-S2 IM 44 YEAROLD GREAT SHAPE I WAS WORKING 14-16 HOURS A DAY THEN ONE DAY I STARTED HAVING SOME PAIN THOUGHT IT WAS SIATIC NERVE ACTING UP WENT TO ER DID MRI NOTHING TREATED ME FOR SIATIC NERVE GAVE ME SHOT MUSCLE RELAXER AND PAIN PILLS THAT NIGHT WOKE UP COULDNT WALK AND SEVERE PAIN CALLED AMBULANCE WENT BACK TO ER DID ANOTHER MRI THEN THEY CAUGHT ABCESS AND DID SURGERY IMEDIATELY THE ABCESS INFECTION WAS STREPOCCOUS HAVE NO IDEA HOW IT GOT THERE WENT HOME WITH IV PIC ANTIBIOTICS ANYWAY GLAD TO HEAR YOU FULLY RECOVERED MY QUESTION HOW LONG DID IT TAKE U TO U WAS ABLE TO WALK ON YOUR OWN I NEVER LOST SENSATION IN MY LOWER EXTREMITIES AND THE DOCTORS SEEMED TO AMAZED BEEN IN THERAPY FOR BOUT 12 DAYS NOW I CAN DO ALL MY ADLS INDEPENDENTLY AND WALKING WITH WALKER 200 FEET AT A TIME BUT STILL REAL WEEK FROM KNEES DOWN ALL THERAPIST SEEM TO BE THINKING IM DOING GREAT BUT WOULD LIKE TO KNOW HOW LONG IT TOOK YOU TO WALK ON YOUR OWN CAUS WHEN I READ YOUR STORY I MEAN IT BLOWED MY MINE I MEAN IDENTICAL ALMOST THANK YOU FOR YOUR TIME FOR READING AND HOPEFULLY YOU WILL YOU WILL ANSWER ME GIVE ME SOME INSIGHT AND HOPE THANKS AGAIN
JMG
Posted November 26, 2018, 8:16 pm
Philip: Thanks for your comment. I wrote that post three years ago and had almost forgotten about it. As best as I can recall, by the time I left the hospital almost three weeks after my laminectomy, I was able to walk, slowly and with a cane. Over the next month, my wife forced me to walk with her with my cane at least one a day, short distances at first and gradually longer. She refused to let me get discouraged. I then started an intensive physiotherapy program. I was able to finally get rid of the cane and walk unassisted approximately 2 months after the surgery. If you’re in good physical shape, are willing to work really hard at recovering and, most importantly, had no loss of sensation at the time you had your surgery, there is no reason you shouldn’t be able to make a full or near-full recovery. I’m now 4.5 years after surgery and have continued to train in Karate and actually just got my 3rd degree black belt this year. The 4 tests were gruelling but I did it, at the age of 51. The key is to keep yourself in very good physical shape and don’t stop doing the exercises you’re shown in physio. Even now, years later, I regularly do stretching exercises on a huge blue ball so that my back muscles don’t stiffen up. You can do this. Just keep working hard and don’t give up!