- Department of Neurosurgery, Geisel School of Medicine, Dartmouth College, Hanover, United States.
- Dartmouth-Hitchcock Medical Center, Section of Neurosurgery, 1 Medical Center Drive, Lebanon, New Hampshire, United States.
- Section of Neurology, 1 Medical Center Drive, Lebanon, New Hampshire, United States.
Correspondence Address:
Daniel R. Calnan
Dartmouth-Hitchcock Medical Center, Section of Neurosurgery, 1 Medical Center Drive, Lebanon, New Hampshire, United States.
DOI:10.25259/SNI-66-2019
Copyright: © 2019 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Erin N. D’Agostino, Daniel R. Calnan, Vyacheslav I. Makler, Imad Khan, John H. Kanter, David F. Bauer. Type I split cord malformation and tethered cord syndrome in an adult patient: A case report and literature review. 07-Jun-2019;10:90
How to cite this URL: Erin N. D’Agostino, Daniel R. Calnan, Vyacheslav I. Makler, Imad Khan, John H. Kanter, David F. Bauer. Type I split cord malformation and tethered cord syndrome in an adult patient: A case report and literature review. 07-Jun-2019;10:90. Available from: http://surgicalneurologyint.com/surgicalint-articles/9356/
Abstract
Background: In a split cord malformation (SCM), the spinal cord is divided longitudinally into two distinct hemicords that later rejoin. This can result in a tethered cord syndrome (TCS). Rarely, TCS secondary to SCM presents in adulthood. Here, we present an adult female with Type I SCM resulting in TCS and a review of literature.
Case Description: A 57-year-old female with a history of spina bifida occulta presented with a 2-year history of worsening back and left leg pain, difficulty with ambulation, and intermittent urinary incontinence; she had not responded to conservative therapy. Magnetic resonance imaging (MRI) revealed a tethered cord secondary to lumbar type I SCM. The patient underwent an L1–S1 laminectomy for resection of the bony septum with cord detethering. At 2-month follow-up, the patient had improvement in her motor symptoms and less pain. In literature, 25 cases of adult-onset surgically managed SCM with TCS were identified (between 1936 and 2018). Patients averaged 37 years of age at the time of diagnosis, and 56% were female.
Conclusion: TCS can present secondary to SCM in adulthood and is characterized predominantly by back and leg pain.
Keywords: Adult, Diastematomyelia, Split cord, Tethered cord
INTRODUCTION
With routine use of folic acid supplementation, spinal dysraphism is rare in Western countries.[
CASE REPORT
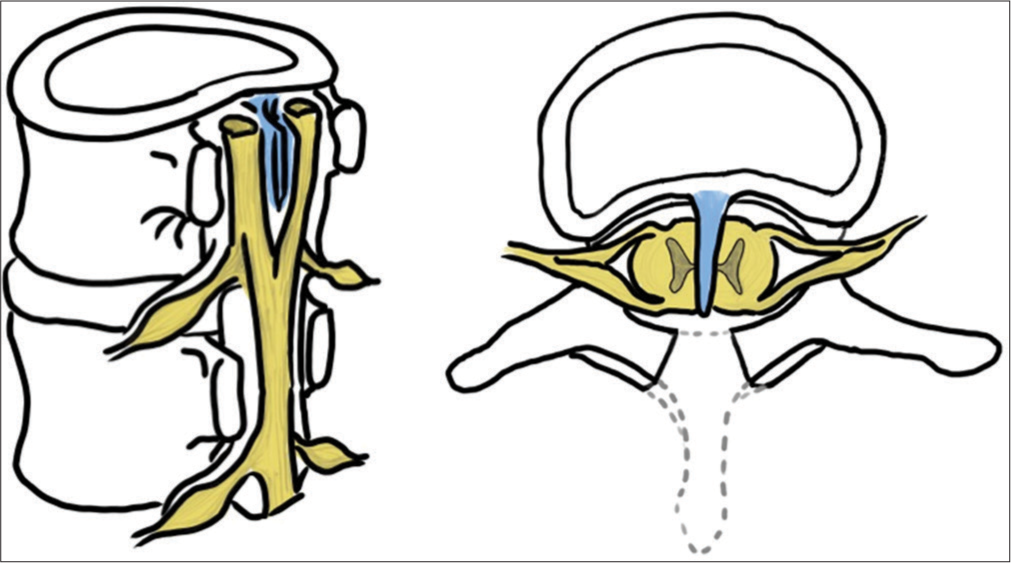
A 57-year-old female with a history of spina bifida occulta and a left-sided clubfoot presented at age 56 with a 2-year history of worsening back and left leg pain, difficulty with ambulation, and intermittent urinary incontinence. On neurologic examination, she had 4+/5 strength in her LLE (iliopsoas, knee extension, knee flexion, dorsiflexion, and plantar flexion) with decreased distal left lower extremity strength (everters absent/loss EHL and decreased sensation). Deep tendon reflexes were 2+ throughout, and she had Babinski sign present on the left. MRI revealed a TC with SCM starting in the upper lumbar spine, becoming two separate dural sacs with a bony septum, and reconstituting into one dural sac/grouping of nerve roots ending in the sacrum [
Figure 2
Split cord radiographic and operative images. (a) Preoperative photograph of an area of hypertrichosis over the lower lumbar spine. (b) Sagittal T2-weighted magnetic resonance imaging (MRI) showing the division of the spinal cord above the dural division and bony septum. (c) T2-weighted axial MRI showed the split cord within a single dural sac. (d) Split cord within divided thecal sac. (e) Intraoperative imaging showing the split cord with associated fat and ectopic ganglion cells from the intradural lesion.
Operative procedure
Under total intravenous anesthesia, with electromyograph (EMG) and somatosensory evoked potential (SEP) monitoring, the patient underwent an L1–S1 laminectomy. Under an operating microscope, the bony septum was removed followed by a durotomy caudal to the split in the thecal sac. No expansile duraplasty was performed. The split cord occurred well before the division in the thecal sac and so was under no tension or compression.
Examination of the cord revealed fat and ectopic ganglion cells and nerve roots extending to the filum. These were resected, and the filum was detached, detethering the cord. The durotomy was closed with a running suture and a Valsalva was performed to verify a watertight closure. Intraoperative monitoring revealed just transient but nonsignificant changes in EMG and SEP.
DISCUSSION
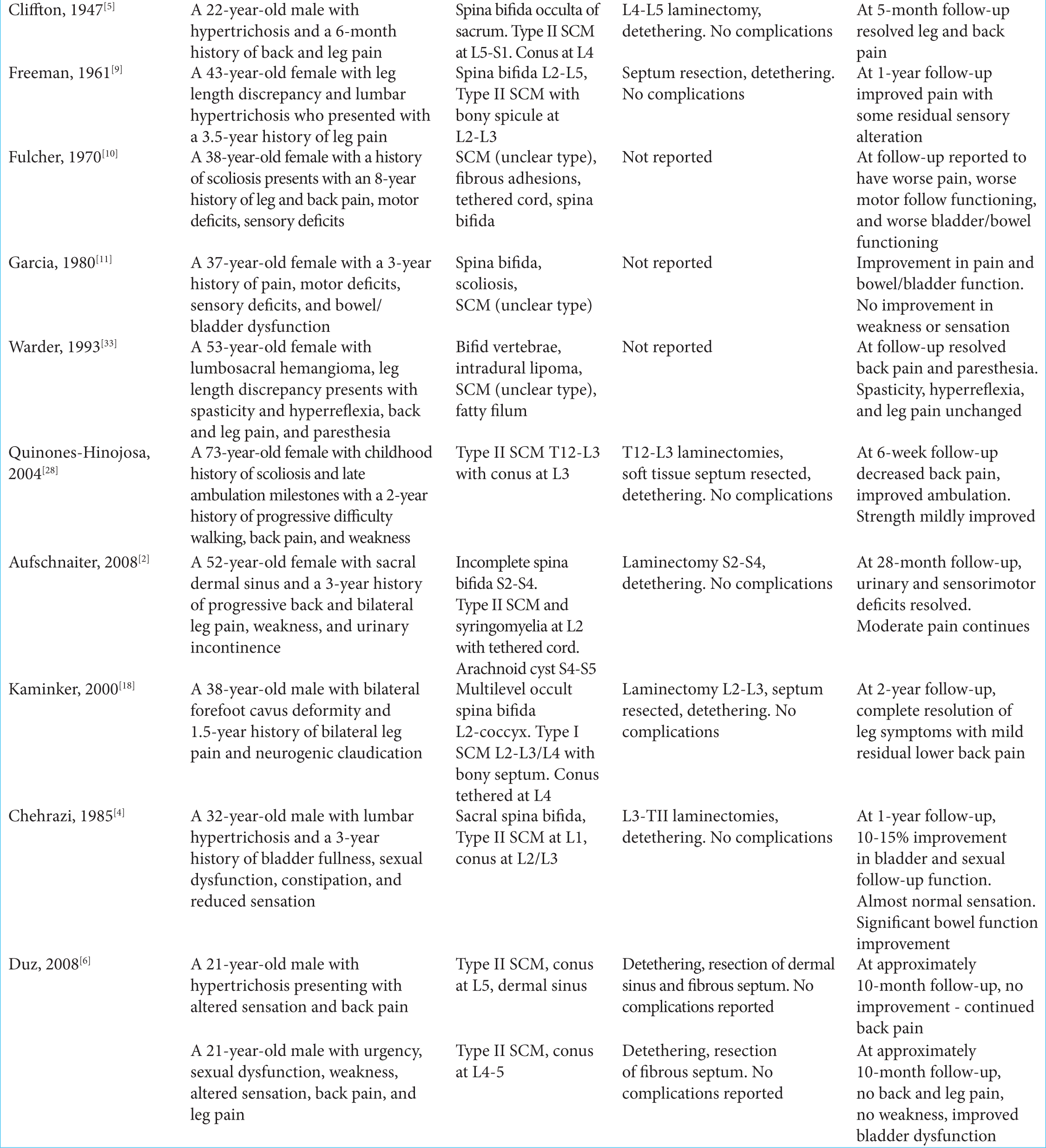
A total of 24 case reports from 20 studies between 1936 and 2018 described SCM and TC diagnosed and surgically treated in adulthood. We included patients over the age of 18 years of age with symptomatic TC/SCM and who underwent surgical detethering. Preoperative status, surgical procedures, and outcomes are summarized [
Infrequently, tethered cord syndrome (TCS) secondary to SCM presents in adulthood.
SCM is reported to account for 10%–38% of adult TC diagnoses.[
Adult TC surgical results
In the adult population, the general consensus is that surgery is appropriate if neurologic deficits exist or symptoms are progressive.[
Complications of adult detethering procedures
Adult patients who undergo detethering appear to have higher complication rates than pediatric patients but lower rates of retethering,[
CONCLUSION
TCS secondary to SCM rarely presents in adulthood. The adult presentation is primarily characterized by back and leg pain. Trauma on a macro/micro scale may result in increased traction on the cord, and progressive vascular compromise resulting in the symptom onset in adulthood. Surgical treatment is appropriate and effective when neurological symptoms are present.
References
1. Akay KM, Izci Y, Baysefer A, Timurkaynak E. Split cord malformation in adults. Neurosurg Rev. 2004. 27: 99-105
2. Aufschnaiter K, Fellner F, Wurm G. Surgery in adult onset tethered cord syndrome (ATCS): Review of literature on occasion of an exceptional case. Neurosurg Rev. 2008. 31: 371-83
3. Barutcuoglu M, Selcuki M, Selcuki D, Umur S, Mete M, Gurgen SG. Cutting filum terminale is very important in split cord malformation cases to achieve total release. Childs Nerv Syst. 2015. 31: 425-32
4. Chehrazi B, Haldeman S. Adult onset of tethered spinal cord syndrome due to fibrous diastematomyelia: Case report. Neurosurgery. 1985. 16: 681-5
5. Cliffton EE, Rydell JR. Congenital dermal (pilonidal) sinus with dural connection; case report and discussion. J Neurosurg. 1947. 4: 276-82
6. Düz B, Gocmen S, Secer HI, Basal S, Gönül E. Tethered cord syndrome in adulthood. J Spinal Cord Med. 2008. 31: 272-8
7. Elmacı İ, Dağçinar A, Özgen S, Ekinci G, Pamir MN. Diastematomyelia and spinal teratoma in an adult: Case report. Neurosurg Focus. 2001. 10: 1-4
8. Erşahin Y, Mutluer S, Kocaman S, Demirtaş E. Split spinal cord malformations in children. J Neurosurg. 1998. 88: 57-65
9. Freeman L. Late symptoms from diastematomyelia. J Neurosurg. 1961. 18: 538-41
10. Fulcher OH, Twigg H. Diastematomyelia in the adult. Neurol Neurocir Psiquiatr. 1970. 11: 31-9
11. Garcia FA, Kranzler LI, Siqueira EB, Weinberg PE, Kranzler KJ. Diastematomyelia in an adult. Surg Neurol. 1980. 14: 93-4
12. Garg K, Singh PK, Kale SS, Sharma BS. Long segment bony spur in split cord malformation Type 1. Indian J Pediatr. 2017. 84: 246-8
13. Guilloton L, Allary M, Jacquin O, Billaud Y, Drouet A, Felten D. Split-cord malformation (diastematolmyelia) presenting in two adults: Case report and a review of the literature. Rev Neurol (Paris). 2004. 160: 1180-6
14. Hamby W, Buffalo N. Pilonidal cyst, spina bifida occulta and bifid spinal cord. Report of a case with review of the literature. Arch Pathol. 1936. 21: 831-8
15. Huang SL, He XJ, Wang KZ, Lan BS. Diastematomyelia: A 35-year experience. Spine (Phila Pa 1976). 2013. 38: E344-9
16. Hüttmann S, Krauss J, Collmann H, Sörensen N, Roosen K. Surgical management of tethered spinal cord in adults: Report of 54 cases. J Neurosurg. 2001. 95: 173-8
17. Iskandar BJ, McLaughlin C, Oakes WJ. Split cord malformations in myelomeningocele patients. Br J Neurosurg. 2000. 14: 200-3
18. Kaminker R, Fabry J, Midha R, Finkelstein JA. Split cord malformation with diastematomyelia presenting as neurogenic claudication in an adult: A case report. Spine (Phila Pa 1976). 2000. 25: 2269-71
19. Kennedy PR. New data on diastematomyelia. J Neurosurg. 1979. 51: 355-61
20. Kim YD, Sung JH, Hong JT, Lee SW. Split cord malformation combined with tethered cord syndrome in an adult. J Korean Neurosurg Soc. 2013. 54: 363-5
21. Klekamp J. Tethered cord syndrome in adults. J Neurosurg Spine. 2011. 15: 258-70
22. Lewandrowski KU, Rachlin JR, Glazer PA. Diastematomyelia presenting as progressive weakness in an adult after spinal fusion for adolescent idiopathic scoliosis. Spine J. 2004. 4: 116-9
23. Linn RM, Ford LT. Adult diastematomyelia. Spine. 1994. 19: 852-4
24. Maiti TK, Bhat DI, Devi BI, Sampath S, Mahadevan A, Shankar SK. Teratoma in split cord malformation: An unusual association: A report of two cases with a review of the literature. Pediatr Neurosurg. 2010. 46: 238-41
25. Maroun FB, Jacob JC, Mangan MA, Hardjasudarma M. Adult diastematomyelia: A complex dysrhaphic state. Surg Neurol. 1982. 18: 289-94
26. Pallatroni HF, Ball PA, Duhaime AC. Split cord malformation as a cause of tethered cord syndrome in a 78-year-old female. Pediatr Neurosurg. 2004. 40: 80-3
27. Pang D, Wilberger JE Jr. Tethered cord syndrome in adults. J Neurosurg. 1982. 57: 32-47
28. Quinones-Hinojosa A, Gadkary CA, Mummaneni PV, Rosenberg WS. Split spinal cord malformation in an elderly patient: Case report. Surg Neurol. 2004. 61: 201-3
29. Russell NA, Benoit BG, Joaquin AJ. Diastematomyelia in adults. A review. Pediatr Neurosurg. 1990. 16: 252-7
30. Sheehan JP, Sheehan JM, Lopes MB, Jane JA. Thoracic diastematomyelia with concurrent intradural epidermoid spinal cord tumor and cervical syrinx in an adult. Case report. J Neurosurg. 2002. 97: 231-4
31. van Leeuwen R, Notermans NC, Vandertop WP. Surgery in adults with tethered cord syndrome: Outcome study with independent clinical review. J Neurosurg. 2001. 94: 205-9
32. Viswanathan VK, Minnema AJ, Farhadi HF. Surgical management of adult Type 1 split cord malformation. Report of two cases with literature review. J Clin Neurosci. 2018. 52: 119-21
33. Warder DE, Oakes WJ. Tethered cord syndrome and the conus in a normal position. Neurosurgery. 1993. 33: 374-8
34. Wolf AL, Tubman DE, Seljeskog EL. Diastematomyelia of the cervical spinal cord with tethering in an adult. Neurosurgery. 1987. 21: 94-8
35. Yamada S, Won DJ, Pezeshkpour G, Yamada BS, Yamada SM, Siddiqi J. Pathophysiology of tethered cord syndrome and similar complex disorders. Neurosurg Focus. 2007. 23: E6-