- Neurosurgery, Ascension Providence Hospital, Michigan State University College of Human Medicine, Southfield, Michigan, United States.
Correspondence Address:
Robert McCabe, Neurosurgery, Ascension Providence Hospital, Michigan State University College of Human Medicine, Southfield, Michigan, United States.
DOI:10.25259/SNI_172_2021
Copyright: © 2021 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Robert McCabe, Doris Tong, Connor Hanson, Dejan Slavnic, Teck Mun Soo. Using aneurysm clips for repair of cisterna chyli injury during posterior spinal fusion. 30-Aug-2021;12:428
How to cite this URL: Robert McCabe, Doris Tong, Connor Hanson, Dejan Slavnic, Teck Mun Soo. Using aneurysm clips for repair of cisterna chyli injury during posterior spinal fusion. 30-Aug-2021;12:428. Available from: https://surgicalneurologyint.com/surgicalint-articles/11081/
Abstract
Background: Injury to the cisterna chyli (CC) is a rare surgical complication with a lack of literature describing its repair. Aneurysm clips have been successfully used to repair durotomies. Its usage in lymphatic injury has never been described. We sought to demonstrate the use of aneurysm clips for the repair of lymphatic vessels.
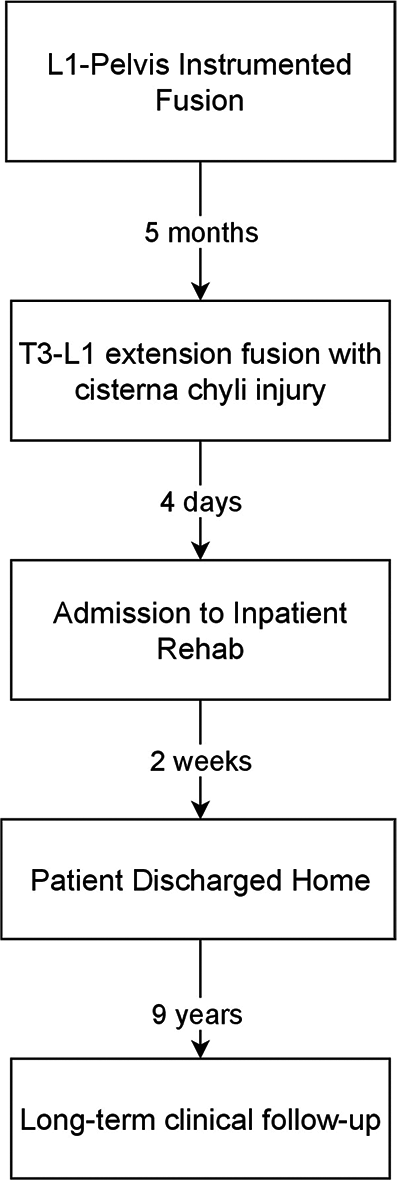
Case Description: A 60-year-old male retired physician with Parkinson’s disease underwent a lumbosacral instrumented fusion with pelvic fixation (L1-pelvis) in 2011. He returned 5 months postoperatively after a fall and was ambulatory with a cane upon admission. CT demonstrated worsening kyphosis with pedicular and superior endplate fracture at the fusion apex. MRI revealed spinal cord compression at the failed level. Extension thoracolumbar fusion was performed (T3-L1) with intraoperative violation of the anterior longitudinal ligament (ALL) during T12/L1 discectomy. CC laceration was suspected. The ALL was dissected from the CC and aorta, allowing visualization of the injury. Three curved aneurysm clips were applied to the lacerated CC, which was visually inspected to ensure a patent lumen. The disk space was filled with poly-methyl-methacrylate cement in place of an interbody cage, preventing migration of the clips. The patient underwent rehabilitation in an inpatient facility with improved ambulation. He has had regular clinic follow-up and was last seen in 2020 with no evidence of lymphedema noted.
Conclusion: CC injury is rare, and usage of aneurysm clips in its repair has never been described. We demonstrate the safe use of aneurysm clips to repair CC injury with long-term favorable clinical outcomes.
Keywords: Cisterna chyli, Lacerations, Ligation
INTRODUCTION
Maintenance of lymphatic flow is crucial for wound healing, immunity, and nutrient uptake, all of which are essential for postoperative recovery.[
CASE DESCRIPTION
A 60-year-old male retired physician with a history of Parkinson’s disease underwent a lumbosacral instrumented fusion with pelvic fixation (L1-pelvis). He required admission to an extended-care facility due to his Parkinson’s disease with medication noncompliance. He was discharged with a walker and reported that his ambulation had improved since surgery. Subsequently, the patient had a fall, noting increased back pain and leg pain, and presented to us 5 months postoperatively.
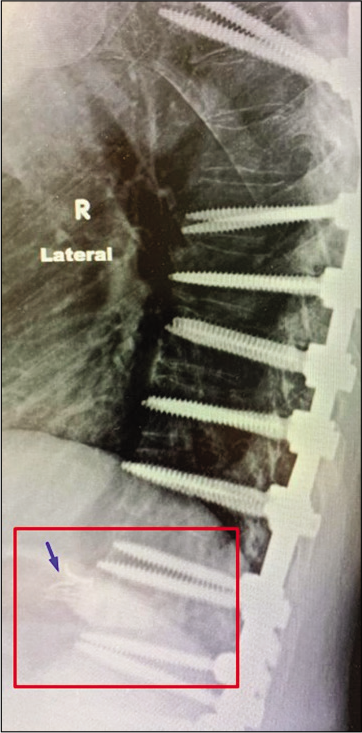
The patient was ambulatory with a cane on admission. However, he had severe back pain and bilateral lower extremity weakness, requiring assistance to stand up. CT thoracic and lumbar spine showed worsening of his kyphosis with pedicular fracture and superior endplate fracture at the apex of his fusion. MRI revealed spinal cord compression at the level of failure. Due to his adjacent segment failure with spinal cord compression, revision and extension of thoracolumbar fusion with instrumentation were performed (T3-L1). Intraoperatively, the anterior longitudinal ligament (ALL) was violated during the T12/L1 discectomy, with fluid noted to be leaking into the disk space.
Due to the proximity of the CC to the thoracolumbar junction, laceration was suspected. Direct visualization was the primary diagnosis method, with a milky fluid noted to be leaking from the CC. Lymphangiography is an imaging technique that may be used to assess the patency of the lymphatic vessels. However, this was not feasible as an intraoperative investigation.[
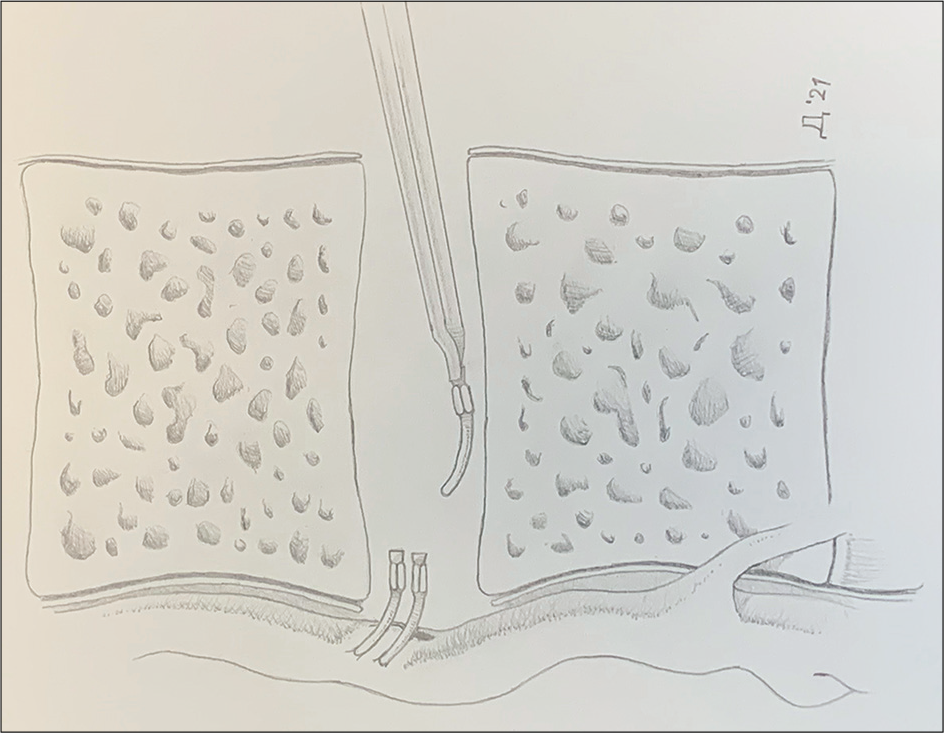
The ALL was dissected from the lacerated CC and descending aorta, allowing clear visualization of the injury. The CC was noted to be adherent to the ALL of the spine and immobile. Curved aneurysm clips [
The patient underwent extensive rehabilitation in an inpatient facility, and his ambulation improved. His balance was significantly compromised due to his Parkinson’s disease, requiring a cane for ambulation. He was regularly followed up in the clinic, and he was last seen in 2020. As a physician, the patient diligently monitored his symptoms and was aware of performing self-evaluations for lymphedema and abdominal ascites. As a result, a lymphangiogram and abdominal X-ray were not performed to evaluate the integrity of the CC. No evidence of lymphedema in the lower extremities has been noted in all subsequent visits [
LITERATURE REVIEW AND DISCUSSION
Untreated lymphatic injury can lead to persistent leaking of chyle and lymph. Long-chain fatty acids (LCFAs) are absorbed through lacteals in the small intestine, then transported to the venous system exclusively through the lymphatic vessels.[
Disruption in this pathway leads to deficiency in processing the LCFA, leading to malnutrition and the possible need for significant dietary restrictions or total parenteral nutrition (TPN), decreased immunity and persistent lymphedema.[
At present, there is no standard of care regarding the diagnosis and treatment of these injuries.[
Conservative management remains a common practice for the treatment of lymphatic leaks. Dietary modifications including TPN with close observation commonly result in the cessation of leaks. Persistent leaks require surgical intervention.[
Techniques for repair include direct surgical repair with absorbable or nonabsorbable sutures and utilization of muscle flaps. These techniques, along with thoracic duct ligation, have been successfully utilized in thoracic duct injuries during lower neck surgery.[
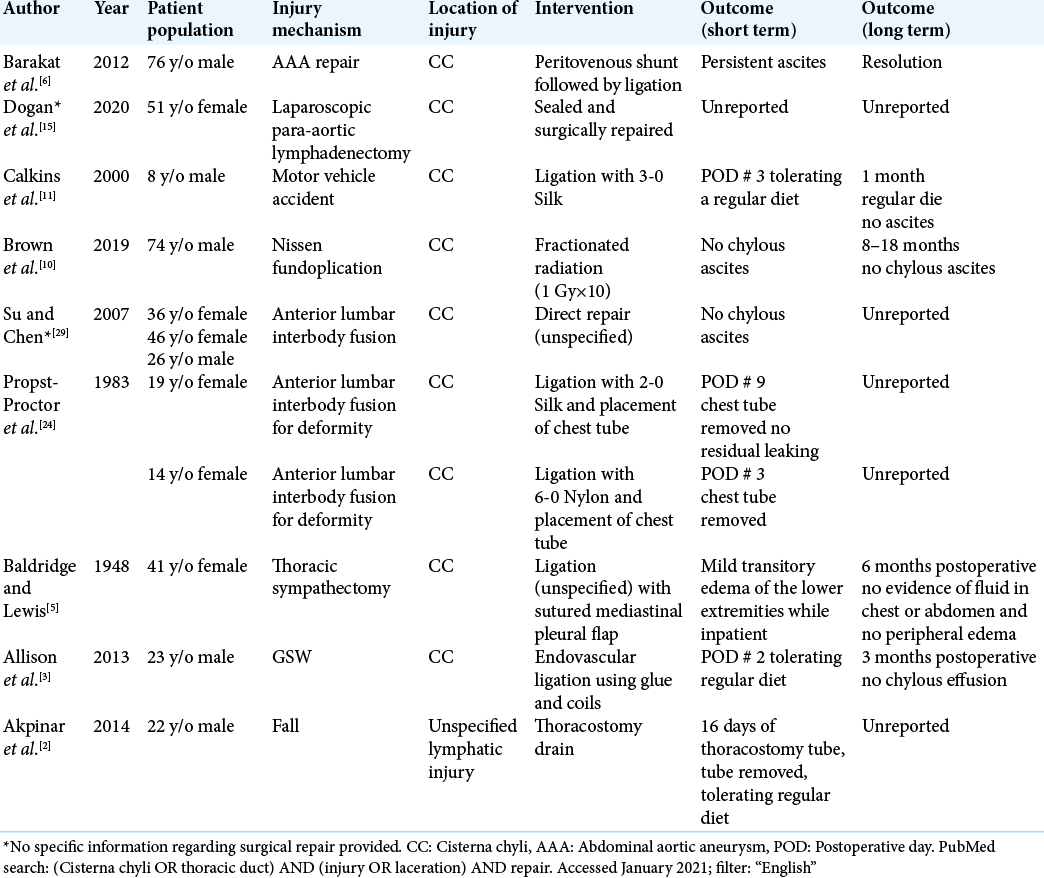
Our literature search resulted in nine articles regarding CC repair [
CONCLUSION
CC injury is rare. The use of aneurysm clips in its repair has never been described. We have demonstrated that aneurysm clips can safely repair CC injury with long-term favorable clinical outcomes.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Ahmed U, Davakis S, Syllaios A, Sdralis E, Lorenzi B, Mastoraki A. Prone position thoracoscopic management of neck chyle leak following major head and neck surgery. A case series. Ann Ital Chir. 2020. 91: 265-72
2. Akpinar V, Duran FY, Duman E, Özkalkanli MY, Duran Ö Horsanali B. Bilateral chylhotorax after Falling from Height. Case Rep Surg. 2014. 2014: e618708
3. Allison S, Rainey M, Aarabi S, Padia SA. Traumatic laceration of the cisterna chyli treated by lymphangiography and percutaneous embolization. Cardiovasc Intervent Radiol. 2014. 37: 267-70
4. Andreou Z, Trinidade A, Shakeel M, Argiris K, Panesar J, Kothari P. Severe hypo-osmotic hyponatraemia due to chylous leak following radical neck dissection. J Coll Physicians Surg Pak. 2013. 23: 221-3
5. Baldridge RR, Lewis RV. Traumatic chylothorax. Ann Surg. 1948. 128: 1056-78
6. Barakat HM, Shahin Y, McCollum P. Chylous ascites complicating elective abdominal aortic aneurysm repair: Case report and review of treatment options. Vasc Endovascular Surg. 2012. 46: 682-5
7. Baumeister RG, Seifert J, Wiebecke B. Behavior of resorbable and nonresorbable suture material in lymph vessel suture. Handchir Mikrochir Plast Chir. 1982. 14: 87-91
8. Beier AD, Barrett RJ, Soo TM. Aneurysm clips for durotomy repair: Technical note. Oper Neurosurg (Hagerstown). 2010. 66: ons-E124-5
9. Börm W, Hübner F, Haffke T, Richter HP, Kast E, Rath SA. Approach-related complications of transthoracic spinal reconstruction procedures. Zentralbl Neurochir. 2004. 65: 1-6
10. Brown S, Abana CO, Hammad H, Brown A, Mhlanga J, Binder C. Low-dose radiation therapy is an effective treatment for refractory postoperative chylous ascites: A case report. Pract Radiat Oncol. 2019. 9: 153-7
11. Calkins CM, Moore EE, Huerd S, Patten R. Isolated rupture of the cisterna chyli after blunt trauma. J Pediatr Surg. 2000. 35: 638-40
12. Campisi CC, Boccardo F, Piazza C, Campisi C. Evolution of chylous fistula management after neck dissection. Curr Opin Otolaryngol Head Neck Surg. 2013. 21: 150-6
13. Chang F, Cheng D, Qian M, Lu W, Li H, Tang H. Thoracic duct chylous fistula following severe electric injury combined with sulfuric acid burns: A case report. Am J Case Rep. 2016. 17: 730-3
14. Doerr CH, Miller DL, Ryu JH. Chylothorax. Semin Respir Crit Care Med. 2001. 22: 617-26
15. Dogan NU, Dogan S, Erol M, Uzun BT. Cisterna chyli: An important landmark in laparoscopic paraaortic lymphadenectomy. Gynecol Oncol. 2020. 156: 511
16. Hsu YJ, Chen PR, Lin YS, Fang HY, Chen CK. Chylothorax following endovascular aortic repair with subclavian revascularization-a case report. J Cardiothorac Surg. 2014. 9: 165
17. Ito H, Usui A, Uchida W, Mutsuga M. Usefulness of lymphography and computed tomography for detecting the site of chyle leakage. Indian J Thorac Cardiovasc Surg. 2019. 35: 104-7
18. Johnson OW, Chick JF, Chauhan NR, Fairchild AH, Fan CM, Stecker MS. The thoracic duct: Clinical importance, anatomic variation, imaging, and embolization. Eur Radiol. 2016. 26: 2482-93
19. Khelif K, Maassarani F, Dassonville M, Laet MH. Thoracoscopic thoracic duct sealing with LigaSure in two children with refractory postoperative chylothorax. J Laparoendosc Adv Surg Tech A. 2007. 17: 137-9
20. Lucente FE, Diktaban T, Lawson W, Biller HF. Chyle fistula management. Otolaryngol Head Neck Surg. 1981. 89: 575-8
21. Marsac J, Frija G, Bismuth V. Chylothorax and the pathology of the lymphatic pleura. Rev Fr Mal Respir. 1982. 10: 227-41
22. Parmeggiani D, Gualtieri G, Terracciano G, Gambardella C, Parisi S, Brusciano L. Prolonged iatrogenic thoracic duct chylous fistula in neck surgery: Conservative management or surgery? A literature review. Scand J Surg. 2021. p. 1457496920987076
23. Paul S, Altorki NK, Port JL, Stiles BM, Lee PC. Surgical management of chylothorax. Thorac Cardiovasc Surg. 2009. 57: 226-8
24. Propst-Proctor SL, Rinsky LA, Bleck EE. The cisterna chyli in orthopaedic surgery. Spine (Phila Pa 1976). 1983. 8: 787-92
25. Rodi T, Nguyen BT, Fritsche E, Rajan G, Scaglioni MF. Direct repair of iatrogenic thoracic duct injury through lymphovenous anastomosis (LVA): A case report. J Surg Oncol. 2020. 121: 224-7
26. Sachs PB, Zelch MG, Rice TW, Geisinger MA, Risius B, Lammert GK. Diagnosis and localization of laceration of the thoracic duct: Usefulness of lymphangiography and CT. AJR Am J Roentgenol. 1991. 157: 703-5
27. Scorza LB, Goldstein BJ, Mahraj RP. Modern management of chylous leak following head and neck surgery: A discussion of percutaneous lymphangiography-guided cannulation and embolization of the thoracic duct. Otolaryngol Clin North Am. 2008. 41: 1231-40
28. Sriram K, Meguid RA, Meguid MM. Nutritional support in adults with chyle leaks. Nutrition. 2016. 32: 281-6
29. Su IC, Chen CM. Spontaneous healing of retroperitoneal chylous leakage following anterior lumbar spinal surgery: A case report and literature review. Eur Spine J. 2007. 16: 332-7
30. Toliyat M, Singh K, Sibley RC, Chamarthy M, Kalva SP, Pillai AK. Interventional radiology in the management of thoracic duct injuries: Anatomy, techniques and results. Clin Imaging. 2017. 42: 183-92