- Department of Neurological Surgery, University of California Irvine, Orange, California, USA
Correspondence Address:
Kiarash Golshani
Department of Neurological Surgery, University of California Irvine, Orange, California, USA
DOI:10.4103/sni.sni_190_17
Copyright: © 2018 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Benjamin Z. Ball, Panayiotis E. Pelargos, Catherine Christie, Kiarash Golshani. Vasospasm in the setting of traumatic bilateral carotid-cavernous fistulas and its effect on treatment. 16-Jan-2018;9:7
How to cite this URL: Benjamin Z. Ball, Panayiotis E. Pelargos, Catherine Christie, Kiarash Golshani. Vasospasm in the setting of traumatic bilateral carotid-cavernous fistulas and its effect on treatment. 16-Jan-2018;9:7. Available from: http://surgicalneurologyint.com/surgicalint-articles/vasospasm-in-the-setting-of-traumatic-bilateral-carotid%e2%80%91cavernous-fistulas-and-its-effect-on-treatment/
Abstract
Background:Direct, Type A, cavernous-carotid fistulas (CCFs) are predominantly caused by head trauma, especially when basilar skull fractures are present. Transarterial endovascular treatment of direct CCFs is the preferred method of treatment. Bilateral CCFs are estimated to be present in 1–2% of the cases. The treatment of bilateral CCFs is difficult often requiring a combination of endovascular and open surgical approaches.
Case Description:We present a case of traumatic bilateral CCFs presenting with vasospasm of the anterior circulation seen on the initial angiogram on day 1 and our treatment paradigm.
Conclusion:This case illustrates the challenges in managing bilateral CCFs as well as the changes in collateral circulation because of cerebral vasospasm which affected our treatment paradigm.
Keywords: Caroticocavernous fistula, carotid cavernous fistula, cerebral vasospasm, head trauma, traumatic subarachnoid hemorrhage
INTRODUCTION
Carotid cavernous fistula (CCF) is an abnormal communication between the internal carotid artery (ICA), external carotid artery (ECA), or any of their branches with the cavernous sinus. Type A CCFs are also considered as direct fistulas, whereas Type B, C, and D fistulas are indirect. CCFs can occur either spontaneously or because of trauma. Traumatic CCFs (tCCFs) make up about 75% of all CCFs and occur in 0.2% of the patients with craniocerebral trauma and approximately 4% of patients with basilar skull fracture.[
Endovascular embolization of direct CCFs is the preferred method of treatment.[
Cerebral vasospasm is a known sequelae of traumatic brain injury (TBI) both in the presence and absence of traumatic subarachnoid hemorrhage (tSAH).[
The treatment options for bilateral direct CCFs are equivalent to the treatment for unilateral CCFs. When complete occlusion of one of the CCFs in bilateral CCF is unachievable, ICA sacrifice is an option only if there is adequate collateral flow from the contralateral ICA after obliteration of the contralateral CCF. In the acute setting of bilateral tCCFs, sacrifice of one of the ICAs may be precluded by vasospasm which reduces collateral flow as can occur in TBI. Here, we present a case of bilateral tCCFs and our treatment paradigm in the setting of concurrent cerebral vasospasm.
CASE REPORT
A 44-year-old male presented as a critical trauma after his head and chest were pinned against a loading dock by a semi-truck traveling at 5 miles per hour. Upon arrival, the patient was in severe hypovolemic shock mainly from active large volume hemorrhage from the oropharynx. He required nasal and oropharyngeal packing, as well as aggressive fluid resuscitation with over two dozen units of blood products. In addition to numerous noncranial injuries, he suffered multiple skull fractures including bilateral fractures traversing the carotid canals.
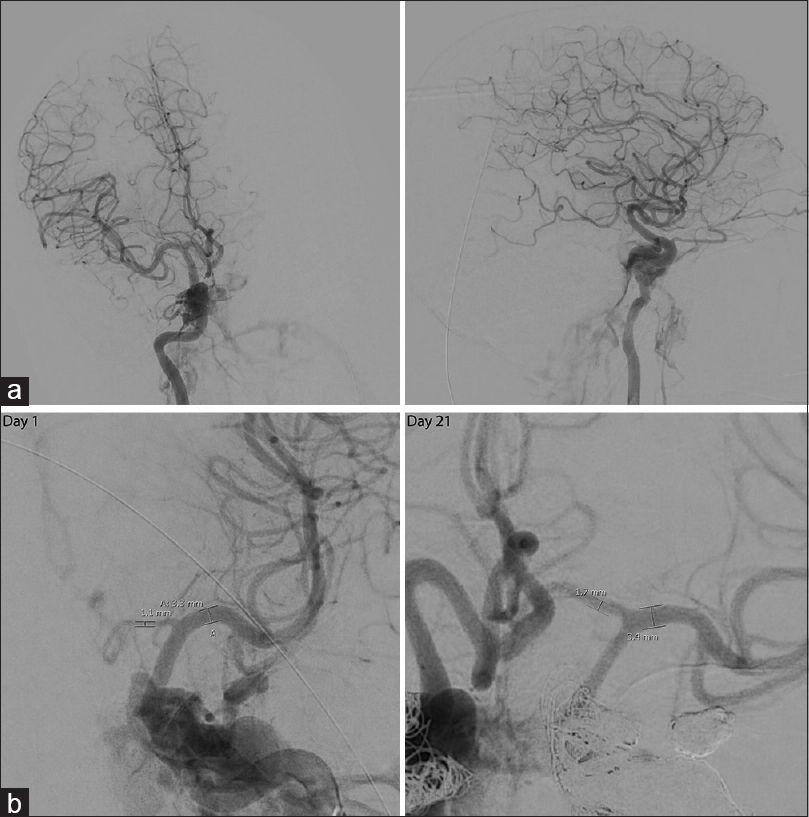
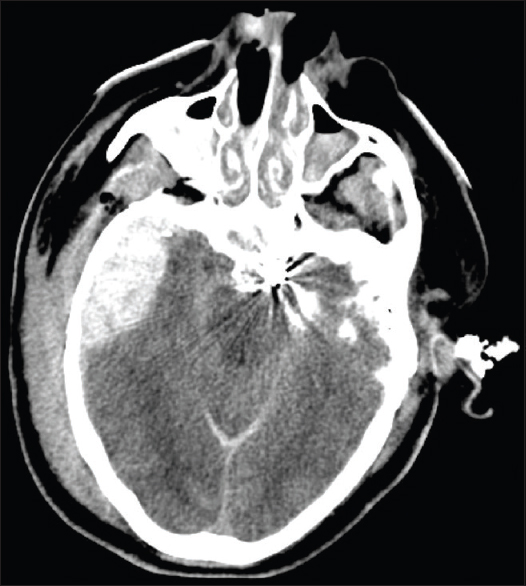
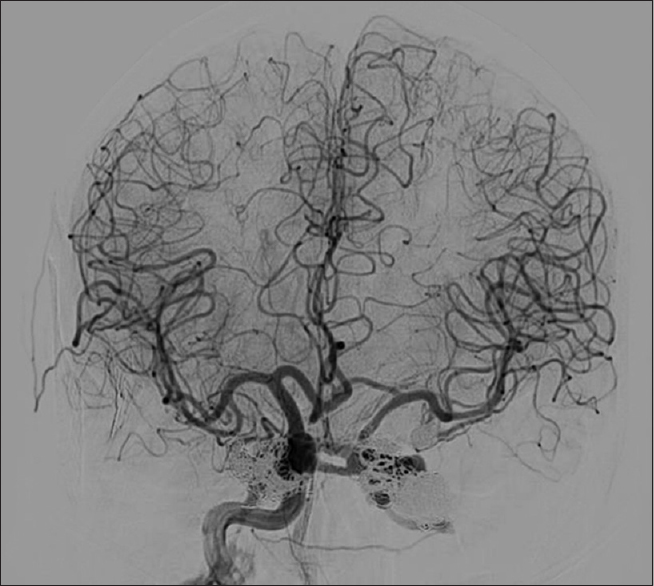
Computed tomography (CT) of the head revealed hemorrhage throughout the basal cisterns, trace hemorrhage within the fourth ventricle, and a thin retroclival hematoma. On CT angiography (CTA), there was suspicion of a focal tear in the cavernous right ICA and faint contrast blush at the left cavernous sinus suspicious for CCF. The angiogram revealed bilateral CCFs, left worse than right. There was no evidence of retrograde venous drainage into the right superior ophthalmic vein (SOV) or the right inferior petrosal sinus at this time [
Figure 1
(a) Anterior-posterior and lateral cerebrocervical angiograms of right ICA injection showing right ICA dissection and CCF near the petrous and cavernous portion of the right ICA without drainage into the superior ophthalmic vein or into the inferior petrosal sinus. (b) Angiogram of left ICA injection on day 1 of admission and angiogram of right ICA injection on day 21. There is vasospasm of the left A1 segment near its origin on day 1 which resolved by day 21
The patient was monitored closely in the ICU, and he needed medical management of his ICP. He had bilateral movement to central pain by day 4, and we had no indication that he was symptomatic from vasospasm. He was kept normotensive and normovolemic and was monitored closely.
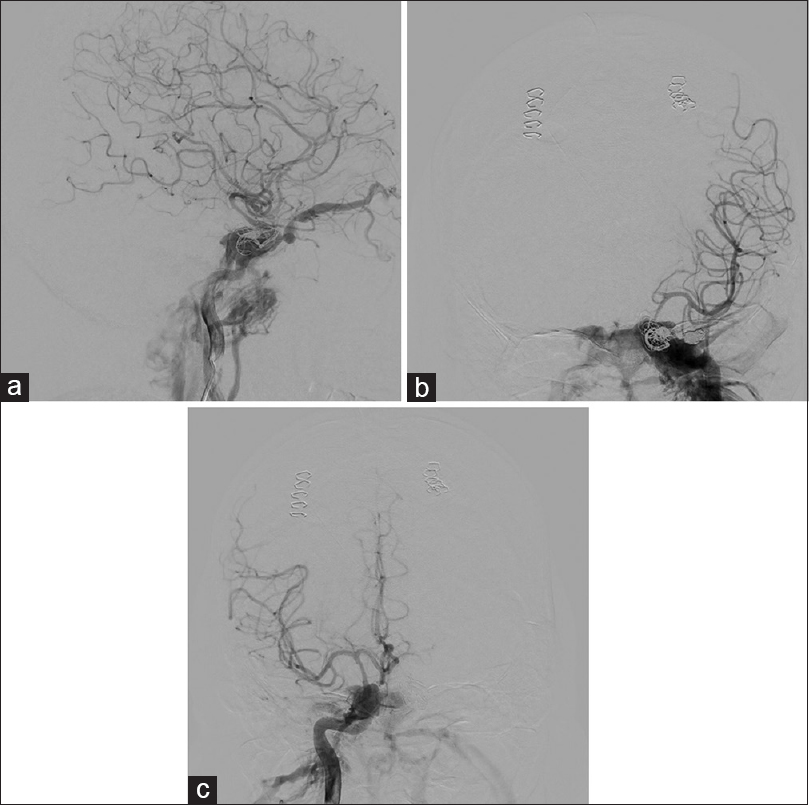
On day 12, after his ICP was well-controlled, he was taken back to the angiography suite where transvenous embolization of the left SOV was performed via left internal jugular vein access to protect the left eye. An attempt was made to embolize the right SOV as well via the right internal jugular vein. However, despite multiple attempts, the microcatheter could not be navigated anteriorly into the right SOV. Although postprocedural arteriograms revealed successful embolization of the left SOV, there was retrograde filling of the right SOV which was not previously observed [
Figure 3
(a) Lateral cerebral angiogram of right ICA injection showing early venous drainage through the right CCF with retrograde filling of the right SOV. Note evidence of vasospasm along the right communicating portion of the ICA and the right M1. (b) Anterior-posterior cerebral angiogram of left ICA injection showing no flow into the left ACA due to severe vasospasm of the left A1 segment and mild to moderate vasospasm of the left ICA and M1 segment of the left MCA. (c) Anterior-posterior cerebral angiogram of right ICA injection showing mild vasospasm of the right ICA and M1 segment of the right MCA. There is no evidence of retrograde cortical venous drainage
The patient was returned to the angiography suite on day 13 for attempted transvenous embolization of the left cavernous sinus and CCF. Multiple attempts to protect the cavernous left ICA using a Scepter C balloon were unsuccessful as the catheter and balloon could not be navigated beyond the fistula site. Because the left ICA could not be protected, transvenous embolization of the CCF was aborted. Sacrifice of the left ICA was not a viable option at the time due to poor collateral supply from the right anterior circulation to the left anterior circulation.
The right SOV was embolized via direct SOV cut-down access on day 19. Cerebral angiogram showed resolution of the vasospasm in the left A1 segment and improved collateral flow from the left anterior circulation to the right across the anterior communicating artery (Acomm). The amount of arteriovenous shunting on the right ICA was significantly reduced. On the left, anterograde flow into the anterior circulation was significantly reduced likely because of the improved collateral supply. There was still evidence of retrograde corticovenous drainage into the left superficial middle cerebral vein.
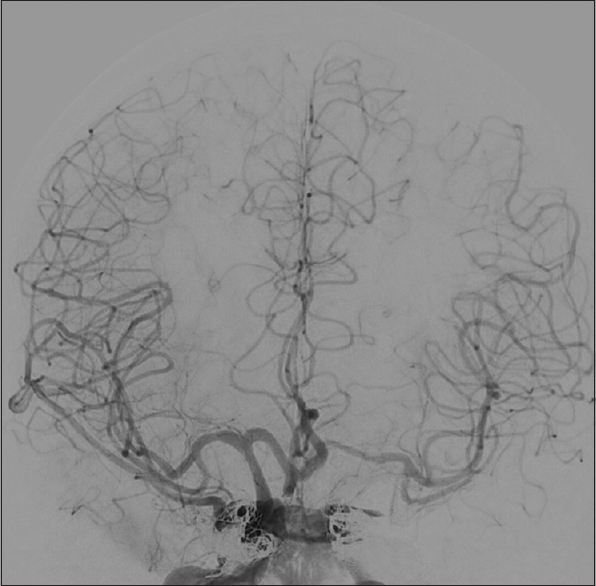
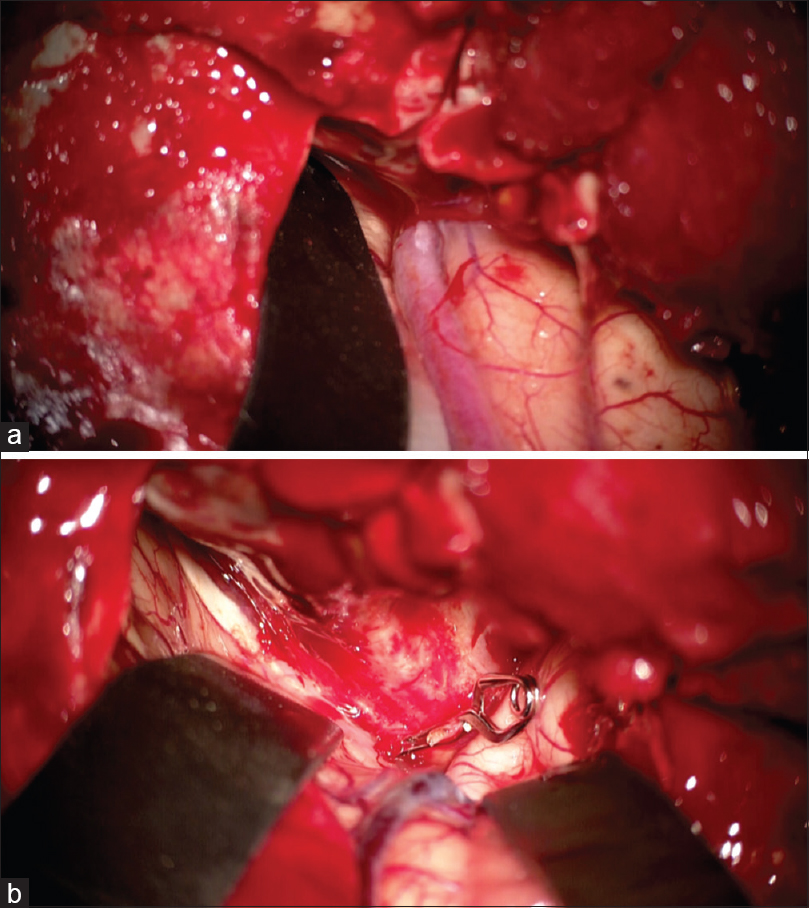
Given the new evidence of collateral flow from the left ICA to the right anterior circulation via the Acomm, at this point the plan became to sacrifice the left ICA because it was the main contributor to the CCF. A balloon occlusion test of the left ICA showed that the venous phase of the left anterior circulation delayed by 1.75 s compared to the right side. Therefore, it was determined that sacrifice of the left ICA was feasible. Because the left ICA distal to the fistula could not be easily accessed, the left cavernous sinus was aggressively embolized through a transvenous route using coils. After transcavernous embolization, there was significant reduction of the arteriovenous flow but a channel remained patent to the distal left anterior circulation. There was concern that coils from the tranvenous embolization could be a source for emboli to the left MCA circulation. Therefore, the proximal left ICA was occluded using amplatzer vascular plugs. Postprocedural angiogram revealed adequate perfusion of the left anterior circulation from the right ICA. Although there was no longer any evidence of retrograde corticovenous drainage on the left, the right superficial middle cerebral vein filled retrograde from the right ICA injection [
Figure 4
Anterior-posterior cerebral angiogram of right ICA injection after left ICA and left cavernous sinus occlusion showing antegrade flow into both anterior circulation. The venous phase on the left was delayed by 1 second compared to the right side. There continues to be evidence of retrograde cortical venous drainage from the right cavernous sinus into the right superficial middle cerebral vein
A remaining concern at this point was the retrograde cortical venous drainage to the right superficial middle cerebral vein. The site of the fistula could not be accessed from the right inferior petrosal sinus, and although coils were deployed in the cavernous sinus during the cut-down access of the left SOV, it did not cure the fistula. Viable treatment options for treating the residual fistula directly included accessing the cavernous sinus directly via needle puncture through the orbit or surgically accessing of the right superficial middle cerebral vein. Because treatment of the fistula site could result in coils herniating into the right carotid artery or stenosis of the carotid artery from external pressure caused by the coil mass, we elected not to attempt to treat the fistula site directly. Instead, we performed a right craniotomy and ligated the arterialized right superficial middle cerebral vein at the fistula site [Figure
Follow-up cerebral angiogram at 6 months revealed interval healing of the residual right CCF. There continued to be adequate collateral flow into the left anterior circulation from the right via the Acomm. However, there was residual filling of the left CCF via retrograde flow from the right anterior circulation across the Acomm [
DISCUSSION
The preceding case highlights two unique features which alter the management of CCFs – bilateral CCFs and concurrent cerebral vasospasm. Reports indicate that up to 90% of patients with CCFs can be treated successfully with a combination of transarterial embolization, transvenous embolization, or ICA sacrifice.[
Cerebral vasospasm is a known sequelae of TBI both in the presence and absence of tSAH. Vasospasm has been detected angiographically in up to 41% of patients with tSAH and by ultrasonography in up to 68%.[
In the present case, cerebral vasospasm was found to occur within hours of the trauma and continued for at least 13 days post-trauma. It was difficult initially to distinguish cerebral vasospasm from hypoplastic A1s which are typical variants of the circle of Willis that limit collateral flow. However, because narrowing was also seen in the ICA and MCA, vasospasm was easier to distinguish. The presence of vasospasm in these arteries severely limited our ability to definitively treat the patient's more symptomatic CCF with early ICA sacrifice. Therefore, the patient's CCFs had to be managed more conservatively until the cerebral vasospasm ceased. Because we had no indication that he was symptomatic from vasospasm, we did not monitor TCDs. In addition, we did not have very effective tools to manage vasospasm in this setting. Angioplasty and/or intra-arterial infusion of verapamil were not good options because we did not have access to the distal intracranial vessels due to the injury. We felt that proximal infusion will mostly be shunted to the venous system and using pressors to treat the vasospasm may increase venous hypertension resulting in venous infarction of the retina or the brain. We did attempt nimodipine but the patient became hypotensive even with a 2-mg dose so we aborted this effort.
This case illustrates these challenges, especially because the patient did not appear to have adequate collateral flow to sacrifice the severely injured left ICA. Vasospasm added to the complexity of this case not only because management of vasospasm was difficult in the setting of bilateral direct CCFs but also because the vasospasm affected the collateral flow and limited the treatment options for the left CCF. We propose that, if preservation of the injured carotid is not an option, patients such as this can initially be managed by protecting the brain and retina from venous hypertension with focal embolization until vasospasm has resolved. At this time, collateral circulation can be reassessed and sacrifice of the injured cavernous ICA may become a good option to definitive treatment of the direct CCF. Another significant point to consider is that, if one of the injured carotid arteries is treated via sacrifice, the contralateral side must be treated with much care because any compromise of flow in this vessel can result in devastating bilateral anterior circulation infarction.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form the patient has given her consent for her images and other clinical information to be reported in the journal. The patient understands that name and initial will not be published and due efforts will be made to conceal identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
None.
Conflicts of interest
There are no conflicts of interest.
References
1. Aminmansour B, Ghorbani A, Sharifi D, Shemshaki H, Ahmadi A. Cerebral vasospasm following traumatic subarachnoid hemorrhage. J Res Med Sci. 2009. 14: 343-8
2. Armin SS, Colohan AR, Zhang JH. Vasospasm in traumatic brain injury. Acta Neurochir Suppl. 2008. 104: 421-5
3. Armonda RA, Bell RS, Vo AH, Ling G, DeGraba TJ, Crandall B. Wartime traumatic cerebral vasospasm: Recent review of combat casualties. Neurosurgery. 2006. 59: 1215-25
4. Barrow DL, Spector RH, Braun IF, Landman JA, Tindall SC, Tindall GT. Classification and treatment of spontaneous carotid-cavernous sinus fistulas. J Neurosurg. 1985. 62: 248-56
5. Bavinzski G, Killer M, Gruber A, Richling B. Treatment of post-traumatic carotico-cavernous fistulae using electrolytically detachable coils: Technical aspects and preliminary experience. Neuroradiology. 1997. 39: 81-5
6. Chan KH, Dearden NM, Miller JD. The significance of posttraumatic increase in cerebral blood flow velocity: A transcranial Doppler ultrasound study. Neurosurgery. 1992. 30: 697-700
7. Compton JS, Teddy PJ. Cerebral arterial vasospasm following severe head injury: A transcranial Doppler study. Br J Neurosurg. 1987. 1: 435-9
8. Grolimund P, Weber M, Seiler RW, Reulen HJ. Time course of cerebral vasospasm after severe head injury. Lancet. 1988. 1: 1173-
9. Gupta AK, Purkayastha S, Krishnamoorthy T, Bodhey NK, Kapilamoorthy TR, Kesavadas C. Endovascular treatment of direct carotid cavernous fistulae: A pictorial review. Neuroradiology. 2006. 48: 831-9
10. Halbach VV, Higashida RT, Barnwell SL, Dowd CF, Hieshima GB. Transarterial platinum coil embolization of carotid-cavernous fistulas. AJNR Am J Neuroradiol. 1991. 12: 429-33
11. Helmke K, Kruger O, Laas R. The direct carotid cavernous fistula: A clinical, pathoanatomical, and physical study. Acta Neurochir. 1994. 127: 1-5
12. Kramer DR, Winer JL, Pease BA, Amar AP, Mack WJ. Cerebral vasospasm in traumatic brain injury. Neurol Res Int. 2013. 2013: 415813-
13. Lewis AI, Tomsick TA, Tew JM. Management of 100 consecutive direct carotid-cavernous fistulas: Results of treatment with detachable balloons. Neurosurgery. 1995. 36: 239-
14. Liang W, Xiaofeng Y, Weiguo L, Wusi Q, Gang S, Xuesheng Z. Traumatic carotid cavernous fistula accompanying basilar skull fracture: A study on the incidence of traumatic carotid cavernous fistula in the patients with basilar skull fracture and the prognostic analysis about traumatic carotid cavernous fistula. J Trauma. 2007. 63: 1014-20
15. Lin CJ, Luo CB, Chang FC, Teng MM, Wang KL, Chu SH. Combined transarterial, transvenous, and direct puncture of the cavernous sinus to cure a traumatic carotid cavernous fistula. J Clin Neurosci. 2009. 16: 1663-5
16. Luo CB, Teng MM, Chang FC, Chang CY. Transarterial balloon-assisted n-butyl-2-cyanoacrylate embolization of direct carotid cavernous fistulas. AJNR Am J Neuroradiol. 2006. 27: 1535-40
17. Luo CB, Teng MM, Yen DH, Chang FC, Lirng JF, Chang CY. Endovascular embolization of recurrent traumatic carotid-cavernous fistulas managed previously with detachable balloons. J Trauma. 2004. 56: 1214-20
18. Macpherson P, Graham DI. Correlation between angiographic findings and the ischaemia of head injury. J Neurol Neurosurg Psychiatry. 1978. 41: 122-7
19. Oertel M, Boscardin WJ, Obrist WD, Glenn TC, McArthur DL, Gravori T. Posttraumatic vasospasm: The epidemiology, severity, and time course of an underestimated phenomenon: A prospective study performed in 299 patients. J Neurosurg. 2005. 103: 812-24
20. Parkinson D. Carotid cavernous fistula: Direct repair with preservation of the carotid artery. Technical note. J Neurosurg. 1973. 38: 99-106
21. Perrein A, Petry L, Reis A, Baumann A, Mertes P, Audibert G. Cerebral vasospasm after traumatic brain injury: An update. Minerva Anestesiol. 2015. 81: 1219-28
22. Suwanwela C, Suwanwela N. Intracranial arterial narrowing and spasm in acute head injury. J Neurosurg. 1972. 36: 314-23
23. Taneda M, Kataoka K, Akai F, Asai T, Sakata I. Traumatic subarachnoid hemorrhage as a predictable indicator of delayed ischemic symptoms. J Neurosurg. 1996. 84: 762-8
24. Uysal E, Kizilkilic O, Ulusay M, Basak M. Endovascular trapping of direct carotid-cavernous fistula. J Clin Neurosci. 2010. 17: 392-4
25. Wang W, Li MH, Li YD, Gu BX, Lu HT. Reconstruction of the Internal Carotid Artery After Treatment of Complex Traumatic Direct Carotid-Cavernous Fistulas With the Willis Covered Stent: A Retrospective Study With Long-Term Follow-up. Neurosurgery. 2016. 79: 794-805
26. Weber M, Grolimund P, Seiler RW. Evaluation of posttraumatic cerebral blood flow velocities by transcranial Doppler ultrasonography. Neurosurgery. 1990. 27: 106-12
27. Zenteno M, Santos-Franco J, Rodriguez-Parra V, Balderrama J, Aburto-Murrieta Y, Vega-Montesinos S. Management of direct carotid-cavernous sinus fistulas with the use of ethylene-vinyl alcohol (Onyx) only: Preliminary results. J Neurosurg. 2010. 112: 595-602
28. Zubkov AY, Lewis AI, Raila FA, Zhang J, Parent AD. Risk factors for the development of post-traumatic cerebral vasospasm. Surg Neurol. 2000. 53: 126-30
29. Zubkov AY, Pilkington AS, Parent AD, Zhang J. Morphological presentation of posttraumatic vasospasm. Acta Neurochir Suppl. 2000. 76: 223-6