- Department of Neurological Surgery, Trauma and Emergency Hospital “Dr. Federico Abete”, Buenos Aires, Argentina
- LINT, Facultad de Medicina, Universidad Nacional de Tucumán, Tucumán, Argentina,
- Department of Clinical-Surgical, Diagnostic and Pediatric Sciences, University of Pavia, Italy,
- Neurosurgery Unit, Department of Surgical Sciences, Fondazione IRCCS Policlinico San Matteo, Pavia, Italy,
- Department of Neurological Surgery, Hospital San Fernando, Argentina, USA
- Laboratory of Microsurgical Neuroanatomy, Second Chair of Gross Anatomy, School of Medicine, University of Buenos Aires, Argentina, USA
- Department of Anesthesiology, Brigham and Women’s Hospital, Harvard Medical School, Boston, USA,
- Department of Brain and Behavioral Sciences, University of Pavia, Italy,
- Department of Neurological Surgery, Hospital Padilla, Tucumán, Argentina.
Correspondence Address:
Sabino Luzzi, Neurosurgery Unit, Department of Clinical-Surgical, Diagnostic and Pediatric Sciences, University of Pavia, Italy;
DOI:10.25259/SNI_1075_2022
Copyright: © 2023 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Derek O. Pipolo1,2, Sabino Luzzi3,4, Matias Baldoncini5,6, Andres Di Pietrantonio1, Walter Brennan1, Humberto Asmus1, Santiago J. Miyara7, Alice Giotta Lucifero3,8, Alvaro Campero2,9. Virtual preoperative planning and 3D tumoral reconstruction with Horos open-source software. 27-Jan-2023;14:32
How to cite this URL: Derek O. Pipolo1,2, Sabino Luzzi3,4, Matias Baldoncini5,6, Andres Di Pietrantonio1, Walter Brennan1, Humberto Asmus1, Santiago J. Miyara7, Alice Giotta Lucifero3,8, Alvaro Campero2,9. Virtual preoperative planning and 3D tumoral reconstruction with Horos open-source software. 27-Jan-2023;14:32. Available from: https://surgicalneurologyint.com/?post_type=surgicalint_articles&p=12124
Abstract
Background: Presurgical three-dimensional (3D) reconstructions allow spatial localization of cerebral lesions and their relationship with adjacent anatomical structures for optimal surgical resolution. The purpose of the present article is to present a method of virtual preoperative planning aiming to enhance 3D comprehension of neurosurgical pathologies using free DICOM image viewers.
Case Description: We describe the virtual presurgical planning of a 61-year-old female presenting a cerebral tumor. 3D reconstructions were created with the “Horos®” Digital Imaging and Communications in Medicine viewer, utilizing images obtained from contrast-enhanced brain magnetic resonance imaging and computed tomography. The tumor and adjacent relevant structures were identified and delimited. A sequential virtual simulation of the surgical stages for the approach was performed with the identification of local gyral and vascular patterns of the cerebral surface for posterior intraoperative recognition. Through virtual simulation, an optimal approach was gained. Accurate localization and complete removal of the lesion were achieved during the surgical procedure. Virtual presurgical planning with open-source software can be utilized for supratentorial pathologies in both urgent and elective cases. Virtual recognition of vascular and cerebral gyral patterns is helpful reference points for intraoperative localization of lesions lacking cortical expression, allowing less invasive corticotomies.
Conclusion: Digital manipulation of cerebral structures can increase anatomical comprehension of neurosurgical lesions to be treated. 3D interpretation of neurosurgical pathologies and adjacent anatomical structures is essential for developing an effective and safe surgical approach. The described technique is a feasible and accessible option for presurgical planning.
Keywords: Neuro-oncology, Simulation, Surgical planning, Three-dimensional reconstruction, Virtual reality
INTRODUCTION
Presurgical planning is an essential process that analyzes individual anatomical features for determining an ideal surgical approach and ensuring therapeutic success. The neurosurgical field is in constant synergy with the latest technological advances, allowing expansion of anatomical insight, pathophysiological bases of diseases, minimization of surgical complications and consequently reducing the morbidity and mortality of treated patients.[
Imaging modalities such as magnetic resonance imaging (MRI) and computed tomography (CT) are our first resources for the identification and examination of lesions of the central nervous system. The basic interpretation of an MRI or CT consists of the sequential review of multiple imaging slices in a two-dimensional (2D) format. The neurosurgeon’s workup does not end here, but in turn must extrapolate these images and construct a three-dimensional (3D) mental image for spatial orientation of the lesion at hand, considering relevant anatomical correlations and adjacent noble structures. In this way, the reality in vivo of the patient and the pathology is assimilated for optimal surgical resolution.[
Although the capability of mental 3D reconstruction is a necessary skill to be acquired by every neurosurgeon, it is not free from errors or individual variability.[
Virtual reality (VR) can be defined as a 3D environment generated by a computer, providing user interaction in real-time with images obtained from MRI and CT.[
To address this obstacle, we describe an alternative unknown to many: virtual preoperative planning and 3D reconstruction utilizing free Digital Imaging and Communications in Medicine (DICOM) image viewers. The objective of this article is to present a method of virtual presurgical planning and enhancement of 3D comprehension of neurosurgical pathologies utilizing free and accessible software.
MATERIALS AND METHODS
We describe the virtual presurgical planning of a 61-year-old female harboring a subcortical cerebral tumor. Images were acquired from a contrast-enhanced brain MRI utilizing T1 weighted volumetric sequences (Philips Ingenia Prodiva 1.5T CX) and a contrast-enhanced brain CT with 1 × 1 mm thickness (Toshiba Aquilion 64). For 3D reconstruction, the DICOM viewer “Horos®” (version 3.3.6;
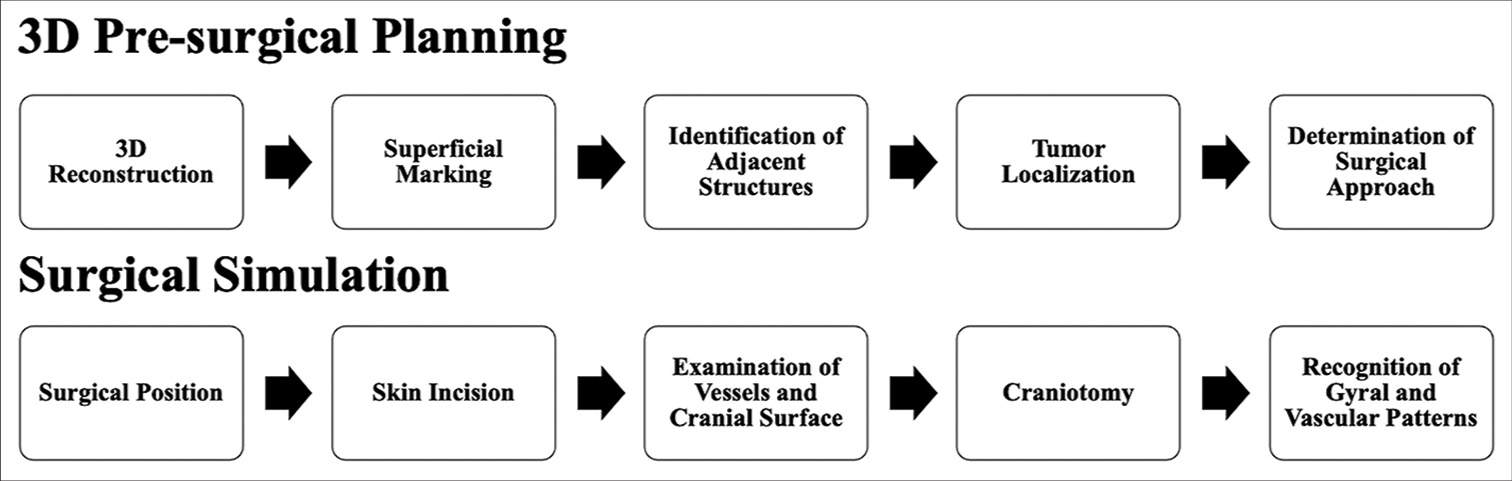
Reconstructions obtained by CT were utilized for visualization of extracranial structures; MRI was preferred for the recognition of intracranial structures and cerebral surfaces. Virtual planning was carried out in two stages: 3D presurgical planning and surgical simulation. Subsequent steps in each stage are described in
Virtual surgical simulation is carried out with head placement in surgical position, emulation of the skin incision, and examination of extracranial structures, such as scalp vessels and osseous landmarks. Simulating a craniotomy, we arrive at the cerebral surface with the identification of local gyral and vascular patterns for posterior intraoperative recognition.
RESULTS
Clinical case
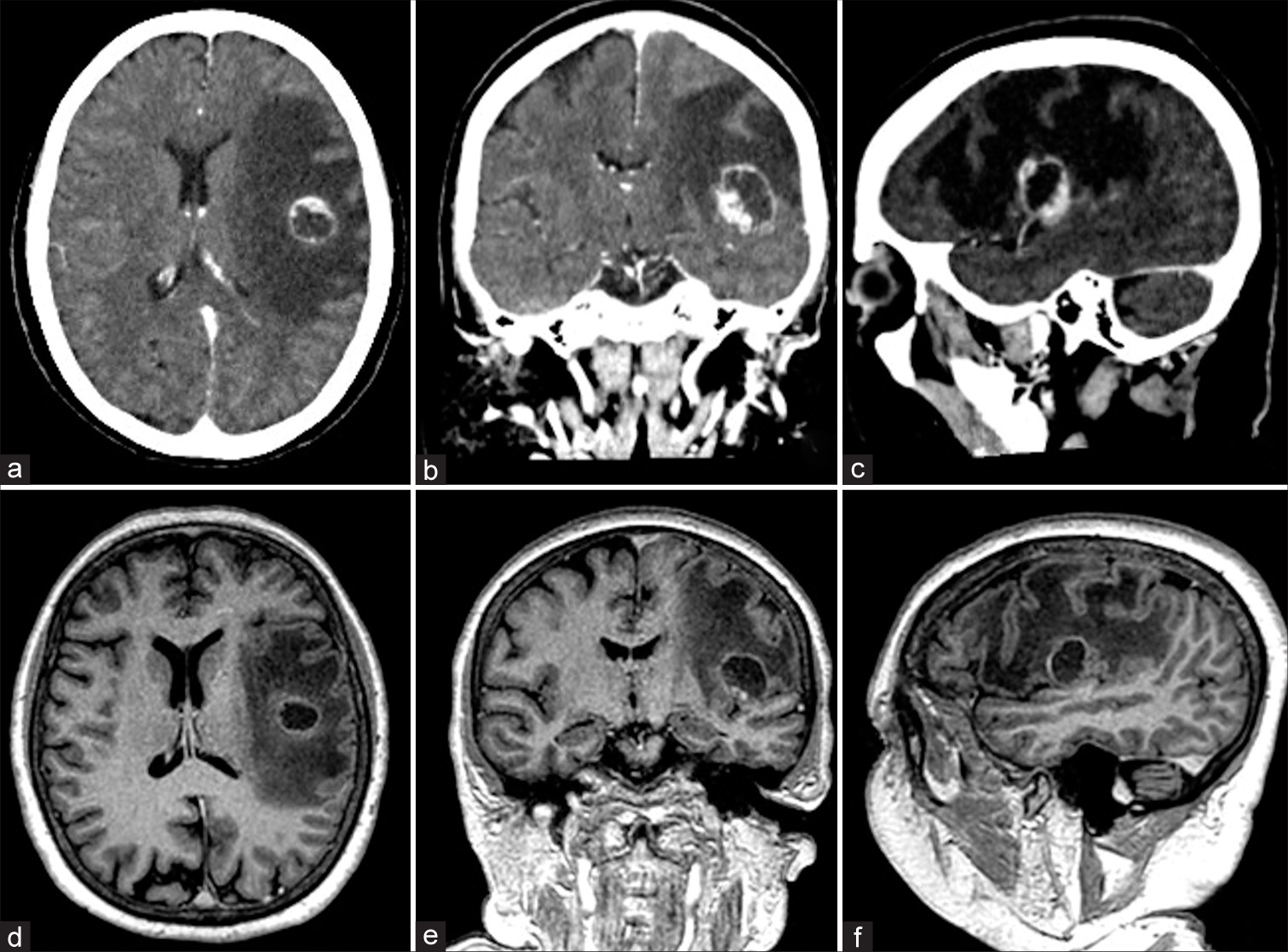
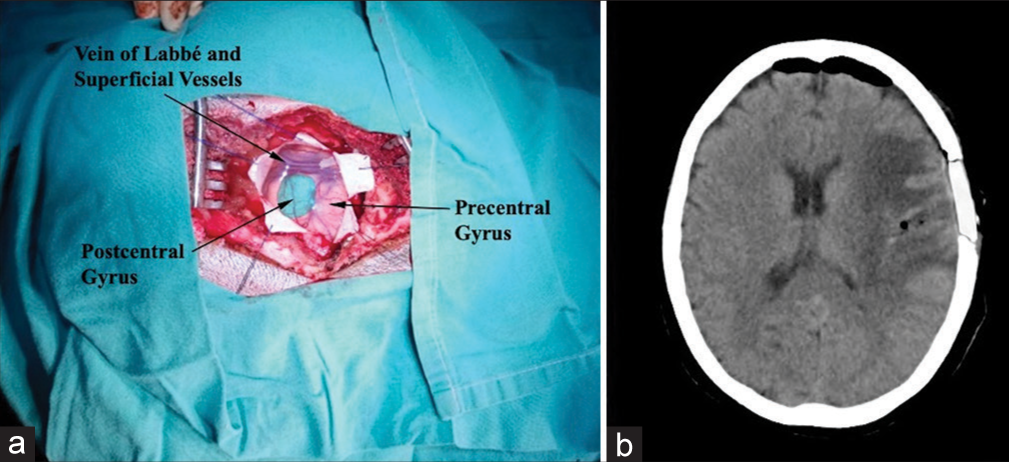
A 61-year-old female with a history of a pulmonary adenocarcinoma treated with chemo and radiotherapy showed up with a 7-day history of a speech disorder and right upper limb weakness. On physical examination, she presented motor aphasia, moderate right facio-brachio hemiparesis, right hemi-hypoesthesia, and a Karnofsky index of 80. The patient underwent brain CT showing a 2 × 2 cm round subcortical lesion located at the lower extremity of the left pre- and post-central gyrus, presenting annular enhancement after intravenous contrast administration, and accompanied by perilesional edema [
Tumor localization and identification of adjacent structures
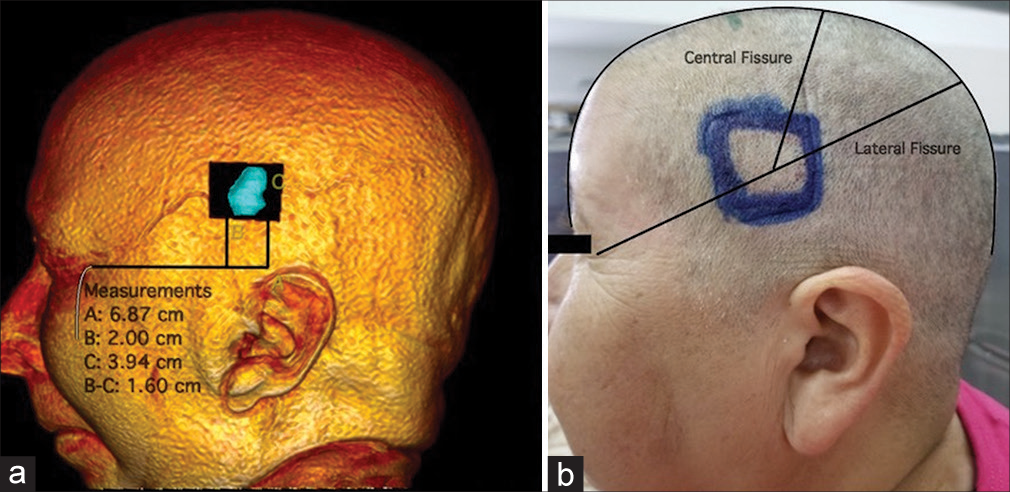
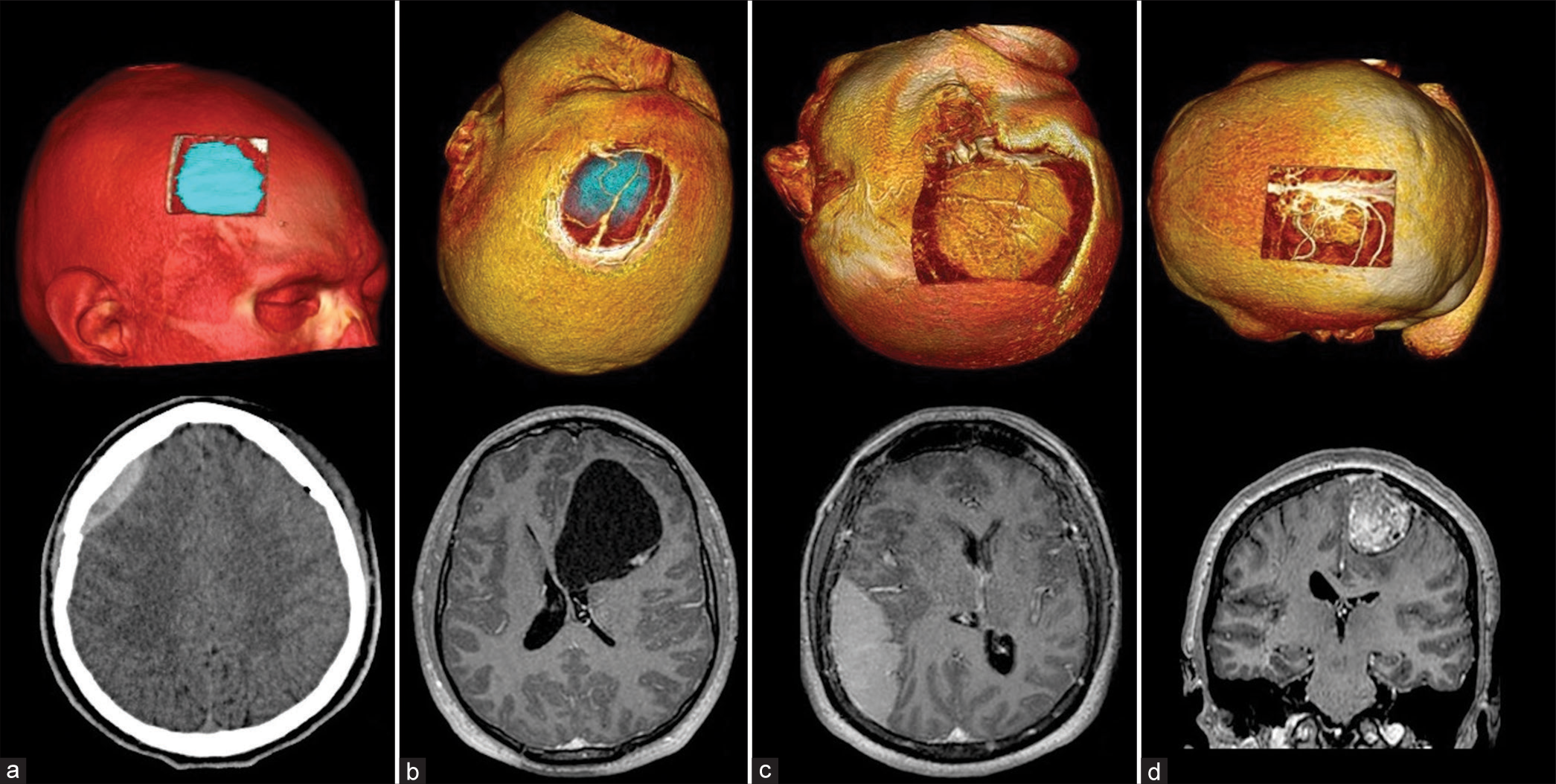
We begin 3D reconstruction and digital tumor localization by marking the tumor’s approximate dimensions projected onto the cranial surface [
Video 1
Furthermore, 3D cranial reconstruction and visualization of the lesion from the surface are carried out, allowing 3D spatial orientation of the lesion within the skull.
The following step consists of measuring and locating the tumor for subsequent marking over the patient [
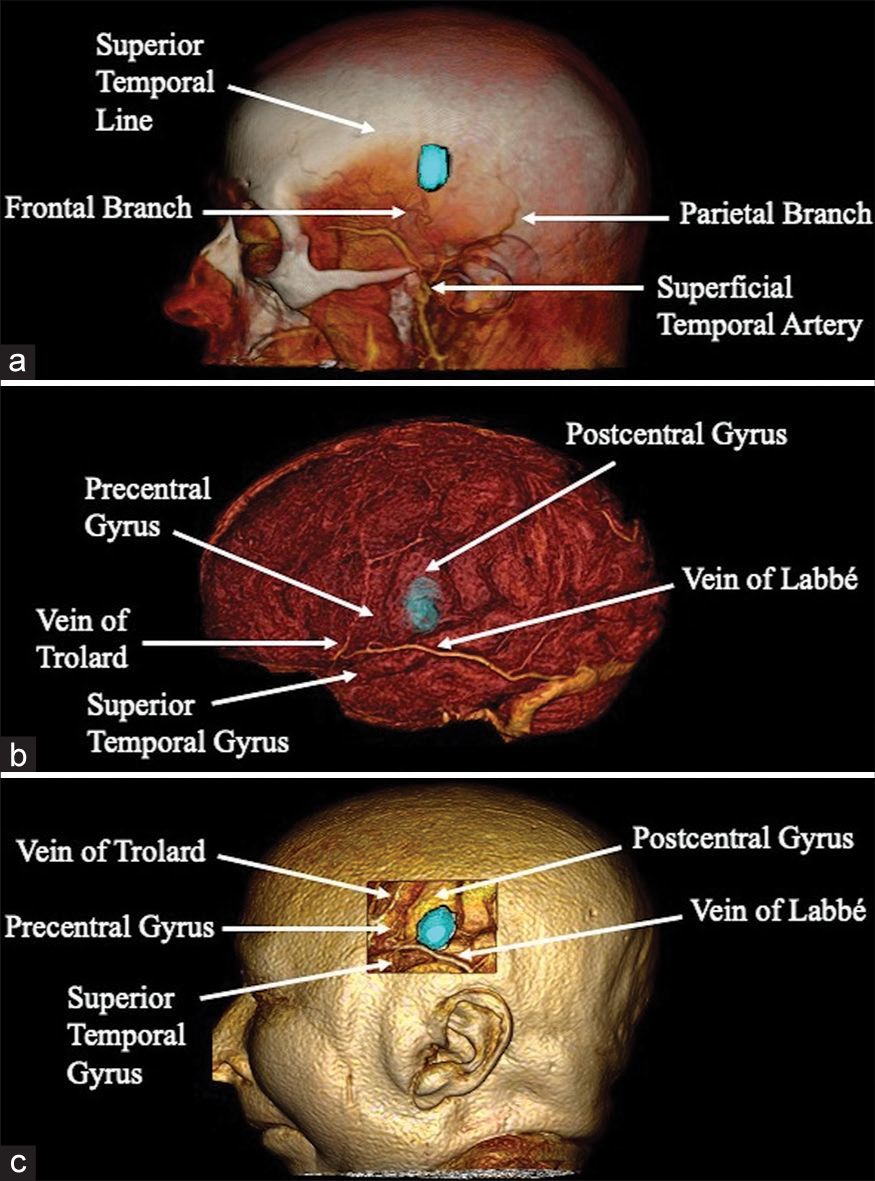
During the extracranial stage, the frontal and parietal branches of the superficial temporal artery were identified as relevant structures, with our lesion located between these and below the superior temporal line [
Determination of surgical approach and simulation
One of the potential advantages of virtual planning is an examination of the selected approach in surgical position, possible alternatives, and the identification of adjacent structures to be recognized during the procedure. In our case, we positioned the patient supine with the head lateralized to the right. A left frontal-parietal linear incision in a cephalo-caudal direction of approximately 6 cm was determined. The frontal branch of the superficial temporal artery was in close proximity to the caudal end of the incision in the subcutaneous and muscular planes; attention to this detail will help ensure its preservation and thus achieve optimal healing. Subsequently, we performed a left temporoparietal craniotomy measuring approximately 3 × 4 cm, immediately inferior to the superior temporal line. Posterior to the dural opening, the vein of Labbé and Sylvian vessels was utilized as the caudal limit of the lesion, establishing a corticotomy over the postcentral gyrus 1 cm rostral to these vessels [
Video 2
The surgical procedure was performed with accurate localization of the lesion by identification of the previously described cortical structures and achieving total removal of the lesion [
DISCUSSION
3D reconstructions for presurgical planning are obtained from volumetric sequences of MRI, CT, and their submodalities (angio-MR and angio-CT, etc.). Numerous free DICOM viewers with 3D reconstruction capacity currently exist, however, the most widely used are Horos® and Radiant viewer®. These provide several tools with neurosurgical applicability, allocating spatial orientation of lesions and their anatomical relationship with adjacent neurovascular and osseous structures.[
3D anatomical orientation and comprehension of cerebral structures in unusual surgical positions can be challenging for the neurosurgeon in training. Through virtual reconstructions, manipulation of anatomical viewpoints and acceleration of the learning curve for spatial orientation can be achieved.[
Several authors have reported successful utilization of “virtual surgeries” for various cerebral pathologies, simulating incisions and craniotomies along with identification of relevant adjacent structures within the layers of the head.[
Intraoperative recognition of vascular and gyral patterns of the cerebral surface are useful for both intra and extra-axial tumoral pathology, allowing the determination of its boundaries, relationship with neighboring structures, and localization of those without cortical expression for less invasive corticotomies. Piece of literature evidence reported similar results with an emphasis on superficial cerebral vascularization as intraoperative reference points.[
In the emergency setting, correct interpretation of the lesion, subsequent surgical approach selection, and prompt action can define the prognosis of a patient. Virtual planning by these means generally requires <30 min, supporting its use in urgent situations.[
Limitations
Although many advantages of this resource have been documented, certain disadvantages must be considered. Infratentorial reconstructions were found to be of little use by the authors for topographic location. In addition, precise surface marking and measurement of certain injuries can be difficult due to the cranial curvatures and lack of tools for correction; the exact distances between the lesions and multiple reference points may require corroboration by means of traditional measurement with 2D images. Furthermore, despite simple and intuitive user interfaces, utilization of these programs requires a basic notion of informatics.
CONCLUSION
The neurosurgical field is found within a digital paradigm aimed at a greater interaction between medical personnel and digital programming for the treatment of pathologies. Despite a large number of freely accessible technological resources worldwide, the educational and illustrative potential of 3D reconstruction with open-sourced software is not fully exploited in our field, either due to access to more sophisticated devices or unawareness of their existence. 3D interpretation of cranial structures is essential for developing an effective and safe surgical approach. Digital manipulation of the various cerebral structures and visualization through multiple angles favors the anatomical comprehension of each individual patient. The described technique is a viable and accessible option for presurgical planning, useful for both the resident in training and experienced neurosurgeon.
Institutional review board statement
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Institutional Review Board.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Videos available online at
Disclaimer
The views and opinions expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Journal or its management. The information contained in this article should not be considered to be medical advice; patients should consult their own physicians for advice as to their specific medical needs.
References
1. Aydin K, Cokluk C, Kuruoglu E, Gelmez S, Diren B, Rakunt C. Using the magnetic resonance three-dimensional volume rendering for tissues technique in the planning of craniotomy flaps with linear scalp incision. Minim Invasive Neurosurg. 2006. 49: 189-93
2. Beyer J, Hadwiger M, Wolfsberger S, Bühler K. High-quality multimodal volume rendering for preoperative planning of neurosurgical interventions. IEEE Trans Vis Comput Graph. 2007. 13: 1696-703
3. Campero A, Baldoncini M, Villalonga JF, Umana GE, Luzzi S, Pipolo DO. Single-stage complete removal of dumbbell-shaped trigeminal schwannoma: 3-Dimensional operative video. World Neurosurg. 2022. 168: 51
4. Canzi P, Avato I, Marconi S, Del Maestro M, Lucifero AG, Magnetto M. A 3D printed custom-made mask model for frameless neuronavigation during retrosigmoid craniotomy. A preclinical cadaveric feasibility study. Ann Ital Chir. 2020. 9: S2239253X20032843
5. da Silva EB, Leal AG, Milano JB, da Silva LF, Clemente RS, Ramina R. Image-guided surgical planning using anatomical landmarks in the retrosigmoid approach. Acta Neurochir (Wien). 2010. 152: 905-10
6. de Oliveira Santos BF, da Costa MD, Centeno RS, Cavalheiro S, de Paiva Neto MA, Lawton MT. Clinical application of an open-source 3D volume rendering software to neurosurgical approaches. World Neurosurg. 2018. 110: e864-72
7. Ferroli P, Tringali G, Acerbi F, Schiariti M, Broggi M, Aquino D. Advanced 3-dimensional planning in neurosurgery. Neurosurgery. 2013. 72: 54-62
8. Giotta Lucifero A, Luzzi S, Brambilla I, Trabatti C, Mosconi M, Savasta S. Innovative therapies for malignant brain tumors: The road to a tailored cure. Acta Biomed. 2020. 91: 5-17
9. Gong XY, Higano S, Mugikura S, Umetsu A, Murata T, Kumabe T. Virtually peeling off the skull and scalp: A simple way of mapping the superficial cerebral veins on the brain surface. Stereotact Funct Neurosurg. 2008. 86: 345-50
10. Harput MV, Gonzalez-Lopez P, Türe U. Three-dimensional reconstruction of the topographical cerebral surface anatomy for presurgical planning with free OsiriX Software. Neurosurgery. 2014. 10: 426-35 discussion 435
11. Hu XP, Tan KK, Levin DN, Galhotra S, Mullan JF, Hekmatpanah J. Three-dimensional magnetic resonance images of the brain: Application to neurosurgical planning. J Neurosurg. 1990. 72: 433-40
12. Jaimovich SG, Guevara M, Pampin S, Jaimovich R, Gardella JL. Neurosurgical planning using osirix software. Surg Neurol Int. 2014. 5: S267-71
13. Jha DK, Khera P, Bhaskar S, Garg M. Three-dimensional volume rendering: An underutilized tool in neurosurgery. World Neurosurg. 2019. 130: 485-92
14. Lovato RM, Araujo JL, Paiva AL, Pesente FS, Yaltirik CK, Harput MV. The use of osirix for surgical planning using cranial measures and region of interest tools: Technical note. Asian J Neurosurg. 2019. 14: 762-6
15. Luzzi S, Lucifero AG. Microscope-based augmented reality with diffusion tensor imaging and fluorescein in insular glioma resection. Neurosurg Focus Video. 2022. 6: V10
16. Luzzi S, Lucifero AG, Baldoncini M, Del Maestro M, Galzio R. Postcentral gyrus high-grade glioma: Maximal safe anatomic resection guided by augmented reality with fiber tractography and fluorescein. World Neurosurg. 2022. 159: 108
17. Luzzi S, Lucifero AG, Martinelli A, Maestro MD, Savioli G, Simoncelli A. Supratentorial high-grade gliomas: Maximal safe anatomical resection guided by augmented reality high-definition fiber tractography and fluorescein. Neurosurg Focus. 2021. 51: E5
18. Luzzi S, Maestro MD, Elia A, Vincitorio F, Perna GD, Zenga F. Morphometric and radiomorphometric study of the correlation between the foramen magnum region and the anterior and posterolateral approaches to ventral intradural lesions. Turk Neurosurg. 2019. 29: 875-86
19. Mandel M, Amorim R, Paiva W, Prudente M, Teixeira MJ, Andrade AF. 3D preoperative planning in the ER with OsiriX®: When there is no time for neuronavigation. Sensors (Basel). 2013. 13: 6477-91
20. Mert A, Buehler K, Sutherland GR, Tomanek B, Widhalm G, Kasprian G. Brain tumor surgery with 3-dimensional surface navigation. Neurosurgery. 2012. 71: ons286-94 discussion ons 294-85
21. Miller J, Acar F, Hamilton B, Burchiel K. Preoperative visualization of neurovascular anatomy in trigeminal neuralgia. J Neurosurg. 2008. 108: 477-82
22. Rotariu DI, Ziyad F, Budu A, Poeata I. The role of osirix based virtual endoscopy in planning endoscopic transsphenoidal surgery for pituitary adenoma. Turk Neurosurg. 2017. 27: 339-45
23. Sinha P, Poggio T. Role of learning in three-dimensional form perception. Nature. 1996. 384: 460-3
24. Spiriev T, Nakov V, Laleva L, Tzekov C. OsiriX software as a preoperative planning tool in cranial neurosurgery: A step-by-step guide for neurosurgical residents. Surg Neurol Int. 2017. 8: 241
25. Wang YC, Liu YC, Hsieh TC, Lee ST, Li ML. Aneurysmal subarachnoid hemorrhage diagnosis with computed tomographic angiography and OsiriX. Acta Neurochir (Wien). 2010. 152: 263-9 discussion 269
26. Xia L, Zhang M, Qu Y, Ren M, Wang H, Zhang H. Localization of transverse-sigmoid sinus junction using preoperative 3D computed tomography: Application in retrosigmoid craniotomy. Neurosurg Rev. 2012. 35: 593-8 discussion 598-9
27. Zele T, Matos B, Knific J, Bajrović FF, Prestor B. Use of 3D visualisation of medical images for planning and intraoperative localisation of superficial brain tumours: Our experience. Br J Neurosurg. 2010. 24: 555-60