- Department of Neurosurgery, Hospital de Clínicas “José de San Martín”, Buenos Aires, Argentina.
- Ángel H. Roffo Institute of Oncology, University of Buenos Aires, School of Medicine, Buenos Aires, Argentina.
- Department of Neurosurgery, FLENI, Buenos Aires, Argentina.
Correspondence Address:
Federico E. Minghinelli, Department of Neurosurgery, Hospital de Clínicas “José de San Martín”, University of Buenos Aires, School of Medicine, Buenos Aires, Argentina.
DOI:10.25259/SNI_713_2022
Copyright: © 2022 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Federico E. Minghinelli1, Rodolfo José Recalde1, Diego Martín Prost1, Hernán Javier Cutuli2, Sebastián Juan María Giovannini3, Roberto Steven Zaninovich1. Which biological pathways are responsible for the late appearance of brain metastases in renal cell carcinoma? Analysis of eight cases. 14-Oct-2022;13:466
How to cite this URL: Federico E. Minghinelli1, Rodolfo José Recalde1, Diego Martín Prost1, Hernán Javier Cutuli2, Sebastián Juan María Giovannini3, Roberto Steven Zaninovich1. Which biological pathways are responsible for the late appearance of brain metastases in renal cell carcinoma? Analysis of eight cases. 14-Oct-2022;13:466. Available from: https://surgicalneurologyint.com/surgicalint-articles/11933/
Abstract
Background: Renal cell carcinoma (RCC) represents 1% of all cancers and its brain metastases amount to 8.1% of all metastatic tumors. Late brain metastases are defined as tumors that appear 10 years after diagnosis of the primary lesion. The objective of this work is to discuss which biological pathways are responsible for the late appearance of these metastases analyzing eight cases.
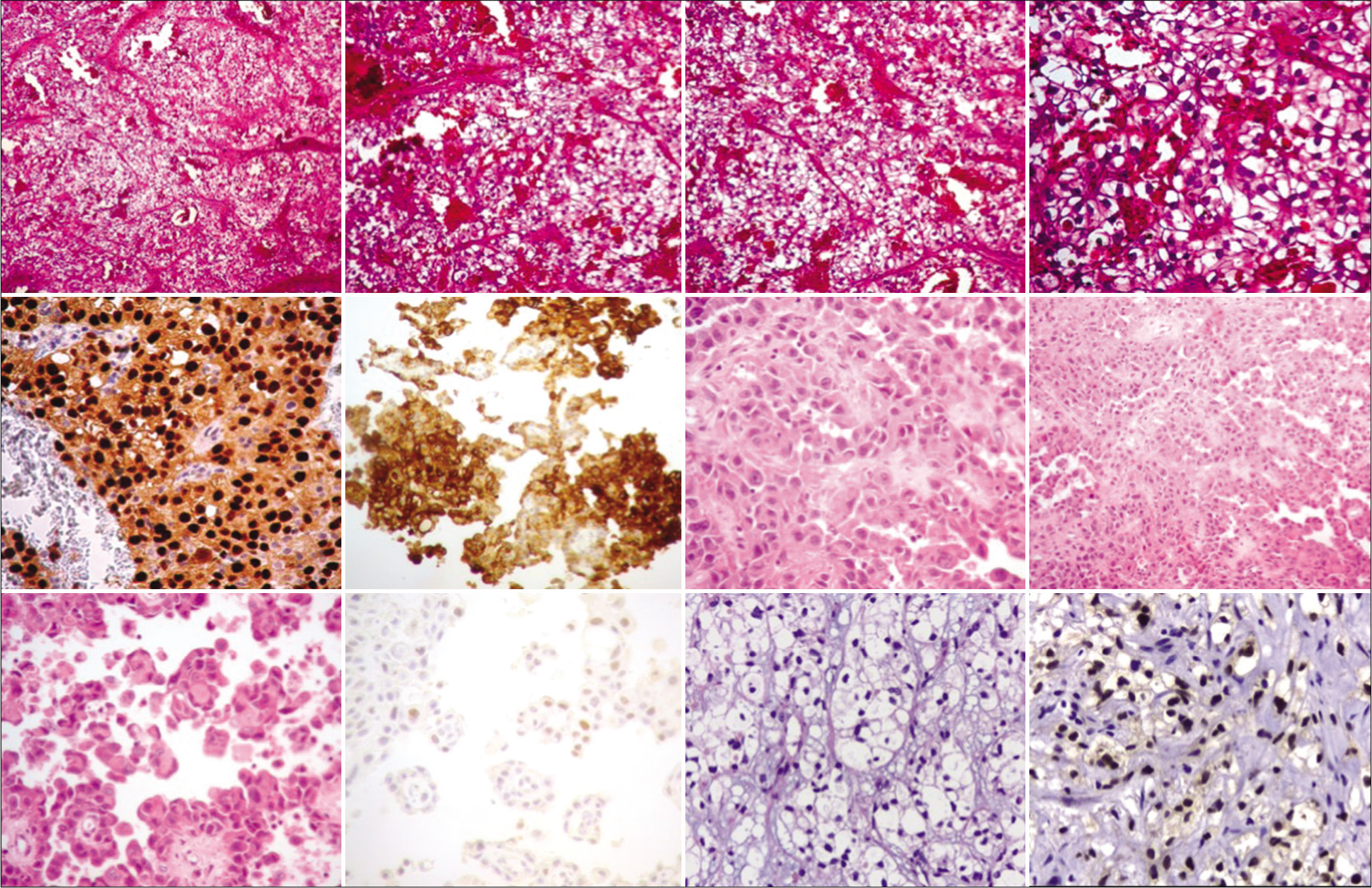
Case Description: We report here eight cases of late brain metastases of RCC treated between 2018 and 2021. Patients consulted for different clinical complaints. Brain magnetic resonance imaging and computed tomography scan were performed on all patients. They were treated by complete surgical resection plus radiosurgery or by radiosurgery alone. The histology of most metastases showed clear cell RCC.
Conclusion: In the presence of a patient with an intracranial tumor and a history of RCC with more than 10 years of evolution, the presence of late metastasis should always be considered. There are many theories described in the literature that try to explain the late appearance of brain metastases from RCC (low mitotic index, impaired immune system, cross talk, self-seeding, and among others).
Keywords: Brain, Late metastasis, Metastases, Pathways, Renal cell carcinoma
INTRODUCTION
Renal cell carcinoma (RCC) is the most common kidney cancer with an incidence estimated of 7.5/100,000 people.[
Many cases of late brain metastases from RCC have been described in the literature;[
CASE REPORT
Eight patients with a 10 years history at least of nephrectomy for RCC were studied. As background, non-robotic-assisted open radical nephrectomy was performed, without adjuvant treatment (radiotherapy or antineoplastic drugs). The patients were free of disease until new symptoms appeared. The symptomatology was related to the affected brain area and due lesion mass effect (headaches, nausea, vomiting, gait disturbance, etc.). Every patient had a brain magnetic resonance imaging (MRI) with and without contrast plus total body tomography.
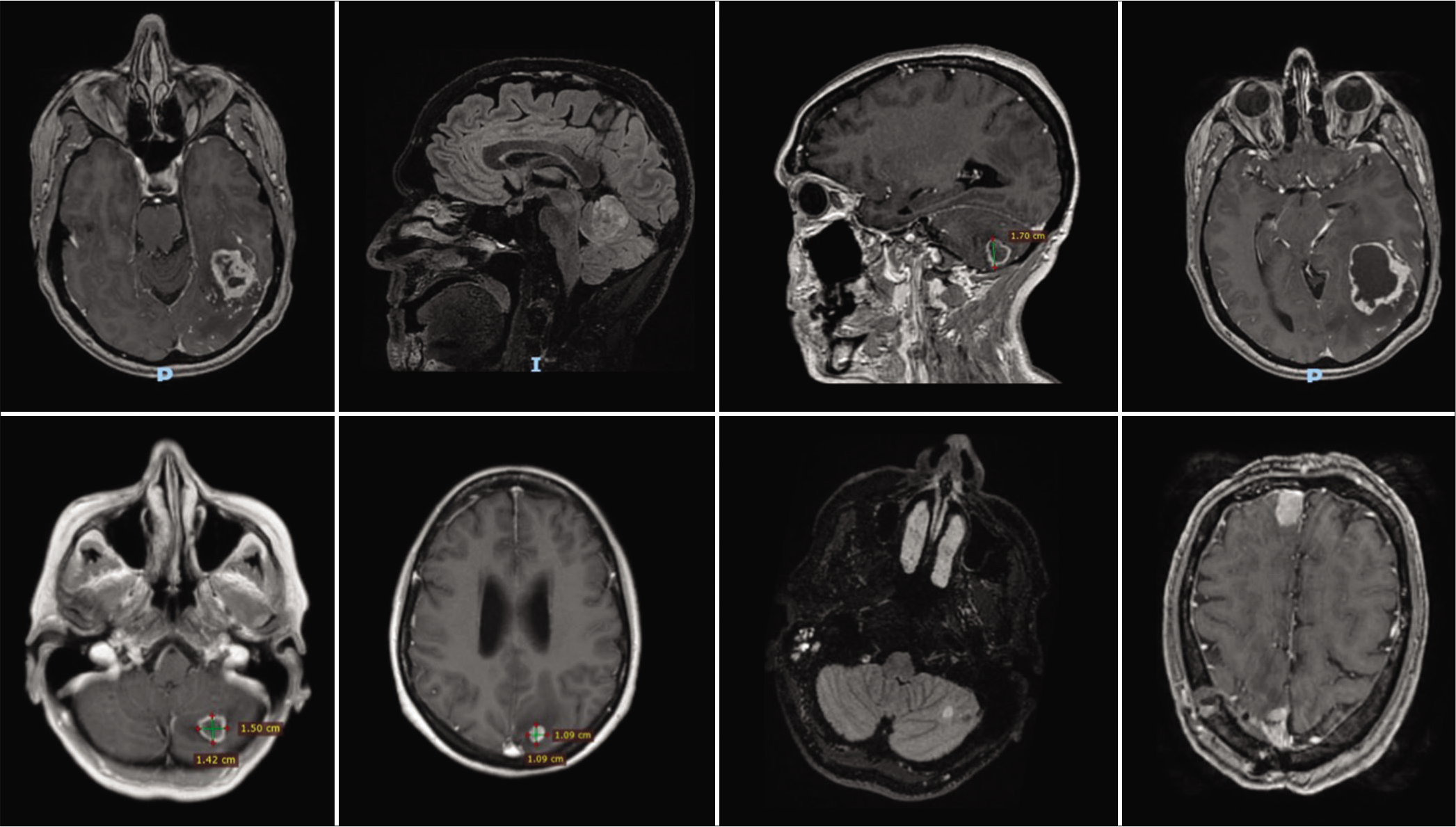
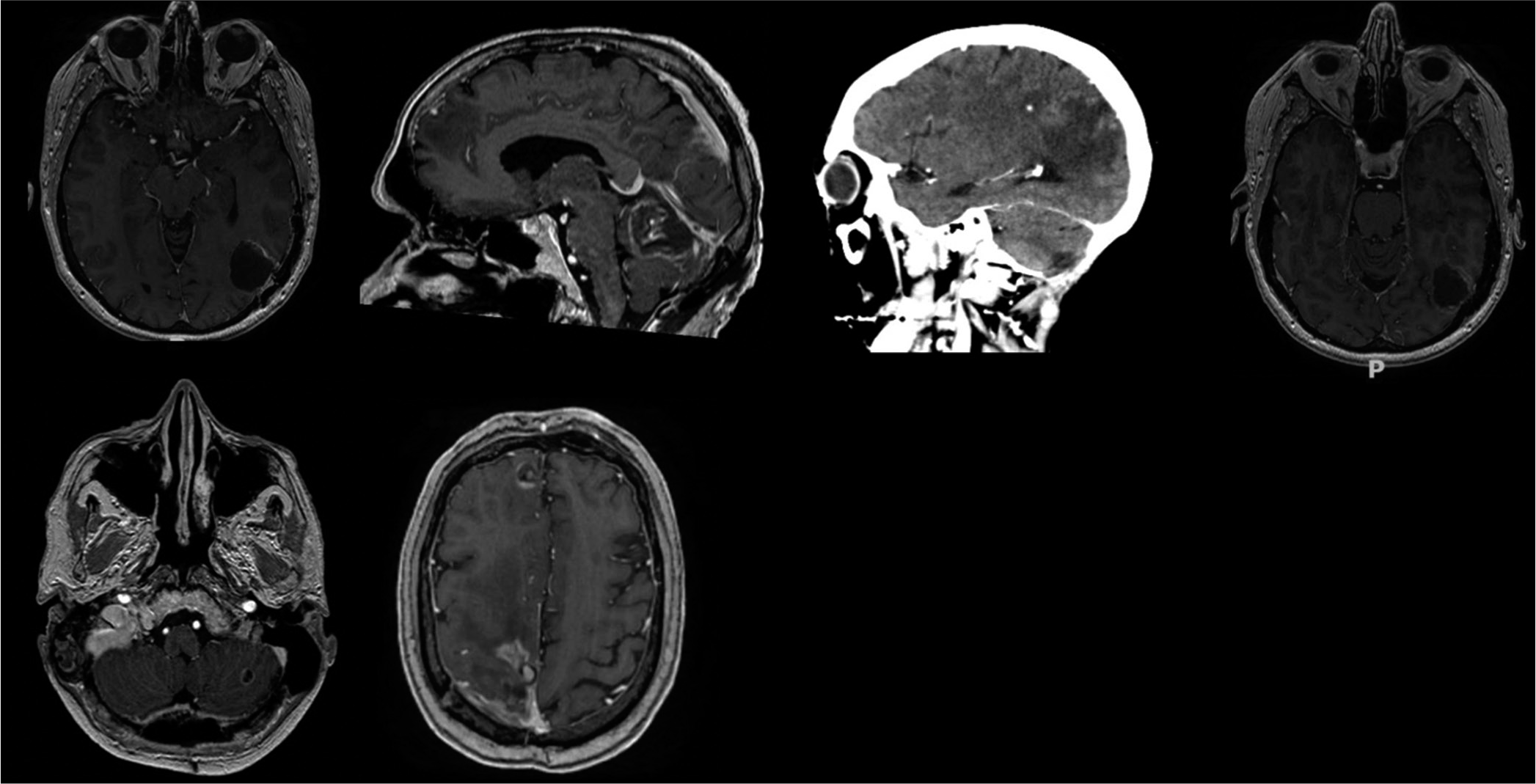
All MRI images were enhanced following contrast administration. Tomography showed no lesions in the rest of the body in the eight patients. Five patients had a single metastasis and three patients suffer from multiple metastases. The areas more affected were the cerebellum, parietal, frontal, and occipital lobes [
Surgical removal of metastases was carried out in seven patients, whereas in the remaining patient, stereotactic biopsy was performed [
Up to now, none of the eight patients had evidence of recurrence.
DISCUSSION
RCC is characterized by slow growth and resistance to cytostatics. This tumor derives from the cells of the proximal convoluted tubule and suffers almost entirely a conducting mutation in the VHL gene, which provides two distinct features such as great vascularization and growth dependent on the balance with the immune system and with angiogenesis. These characteristics determine that the renal cell tumor follows a different development than other solid tumors, because it depends on molecules such as transforming growth factor alpha, vascular endothelial growth factor, and platelet-derived growth factor receptor that allow or stimulate growth.[
Brain metastases from RCC can appear early or late in the course of the disease. Late recurrence is defined as that developing patients who had a disease-free interval of 10 years after the initial surgery.[
The eight cases in the present report had late metastases. In all these patients, nephrectomy was the initial treatment. This sequence of events was central to the understanding of the pathophysiology of this process, since there is a clear relationship between the immune system and RCC. It would be possible that the tumor can suppress the immune system or make it its partner, as suggested by Vuong et al.[
The preceding observations are lead us to suggest that the slow growth of metastases following the resection of the primary tumor may have been caused by the suppression of the crossed-talk between the primary tumor and its metastases. Another view is that of a self-seeding, consisting of infiltration of aggressive circulating tumor cells into the primary tumor.[
These pathways would support the hypothesis proposed here, that these patients presented synchronous metastases, and it would be the elimination of the primitive tumor that generates orphan metastases and a state of quiescence exceeding 10 years.[
CONCLUSION
In a patient with an intracranial tumor and a history of RCC of more than 10-year duration, the possible presence of late metastasis should be considered. There are many theories described in the literature that try to explain the late appearance of brain metastases from RCC (low mitotic index, impaired immune system, cross-talk, self-seeding, and among others). More cases of late brain metastases in RCC need to be investigated to recognize the biological and genetic pathways underlying the presence of metastases after a long period of time. The series reported in the literature showed that the proper management can control the disease, improve the quality of life, and prolong survival.
Declaration of patient consent
Patients’ consent not required as patients’ identities were not disclosed or compromised
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Ammirati M, Samii M, Skaf G, Sephernia A. Solitary brain metastasis 13 years after removal of renal adenocarcinoma. J Neurooncol. 1993. 15: 87-90
2. Bademci G, Bozdogan O, Berdan F, Evliyaoglu C. Extremely delayed renal cell carcinoma metastasis mimicking convexity meningioma. Neurocirugia (Astur). 2008. 19: 562-4
3. Bianchi M, Sun M, Jeldres C, Shariat SF, Trinh QD, Briganti A. Distribution of metastatic sites in renal cell carcinoma: A population-based analysis. Ann Oncol. 2012. 23: 973-80
4. Cannady SB, Cavanaugh KA, Lee SY, Bukowski RM, Olencki TE, Stevens GH. Results of whole brain radiotherapy and recursive partitioning analysis in patients with brain metastases from renal cell carcinoma: A retrospective study. Int J Radiat Oncol Biol Phys. 2004. 58: 253-8
5. Cervoni L, Salvati M, Delfini R. Late solitary cerebral metastasis from renal carcinoma. J Neurosurg Sci. 1993. 37: 247-9
6. Choi WH, Koh YC, Song SW, Roh HG, Lim SD. Extremely delayed brain metastasis from renal cell carcinoma. Brain Tumor Res Treat. 2013. 1: 99-102
7. Choueiri TK, Xie W, Kollmannsberger C, North S, Knox JJ, Lampard JG. The impact of cytoreductive nephrectomy on survival of patients with metastatic renal cell carcinoma receiving vascular endothelial growth factor targeted therapy. J Urol. 2011. 185: 60-6
8. Fukushima Y, Yoshikawa G, Takasago M, Shimizu S, Tsutsumi K. Extremely delayed multiple brain metastases from renal cell carcinoma: Remission achieved with total surgical removal: Case report and literature review. World Neurosurg. 2016. 92: 583.e13-7
9. Ishikawa J, Umezu K, Yamashita H, Maeda S. Solitary brain metastasis from renal cell carcinoma 14 years after nephrectomy: A case report. Hinyokika Kiyo. 1990. 36: 1439-41
10. Jubelirer SJ. Late solitary cerebral metastasis from renal cell carcinoma: A case report and review of the literature. W V Med J. 1996. 92: 26-7
11. Karasawa H, Naito H, Sugiyama K, Ueno J, Kin H, Nagayama T. A brain metastasis developed eleven years after a renal cell carcinoma removal: A case report. No Shinkei Geka J. 1994. 3: 446-50
12. Killebrew K, Krigman M, Mahaley MS, Scatliff JH. Metastatic renal cell carcinoma mimicking a meningioma. Neurosurgery. 1983. 13: 430-4
13. Kim MY, Oskarsson T, Acharyya S, Nguyen DX, Zhang XH, Norton L. Tumor self-seeding by circulating cancer cells. Cell. 2009. 139: 1315-26
14. Kim YH, Kim JW, Chung HT, Paek SH, Kim DG, Jung HW. Brain metastasis from renal cell carcinoma. Prog Neurol Surg. 2012. 25: 163-75
15. Kolsi F, Mechergui H, Kammoun B, Mellouli M, Khrifech M, Boudawara MZ. Delayed brain metastasis from renal cell carcinoma. Urol Case Rep. 2019. 22: 54-6
16. Kuroki K, Taguchi H, Sumida M, Daimaru Y, Onda J. Cerebral metastasis from a renal cell carcinoma more than 10 years after nephrectomy: Report of two cases. No Shinkei Geka. 1999. 27: 89-93
17. McIntosh AG, Ristau BT, Ruth K, Jennings R, Ross E, Smaldone MC. Active surveillance for localized renal masses: Tumor growth, delayed intervention rates, and >5-yr clinical outcomes. Eur Urol. 2018. 74: 157-64
18. McNichols DW, Segura JW, DeWeerd JH. Renal cell carcinoma: Long-term survival and late recurrence. J Urol. 1981. 126: 17-23
19. Middleton R. Surgery for metastatatic renal cell carcinomas. J Urol. 1967. 97: 973-7
20. Miyao N, Naito S, Ozono S, Shinohara N, Masumori N, Igarashi T. Late recurrence of renal cell carcinoma: Retrospective and collaborative study of the Japanese society of renal cancer. Urology. 2011. 77: 379-84
21. Montano N, Puca A, Pierconti F, Larocca LM. Extremely delayed falx metastasis from renal cell carcinoma. Neurology. 2007. 68: 1541-2
22. Nakano E, Fujioka H, Matsuda M, Osafune M, Takaha M, Sonoda T. Late recurrence of renal cell carcinoma after nephrectomy. Eur Urol. 1984. 10: 347-9
23. Nieder C, Spanne O, Nordøy T, Dalhaug A. Treatment of brain metastases from renal cell cancer. Urol Oncol. 2011. 29: 405-10
24. Pulido EG, Centeno AM, Rey PM, Narbón ES. Molecular biology of the clear cell renal cell carcinoma: Principles for a selective treatment. Actas Urol Esp. 2007. 31: 233-43
25. Radley MG, McDonald JV, Pilcher WH, Wilbur DC. Late solitary cerebral metastases from renal cell carcinoma: Report of two cases. Surg Neurol. 1993. 39: 230-4
26. Rosenberg SA, Mulé JJ, Spiess PJ, Reichert CM, Schwarz SL. Regression of established pulmonary metastases and subcutaneous tumor mediated by the systemic administration of high-dose recombinant interleukin 2. J Exp Med. 1985. 161: 1169-88
27. Roser F, Rosahl SK, Samii M. Single cerebral metastasis 3 and 19 years after primary renal cell carcinoma: Case report and review of the literature. J Neurol Neurosurg Psychiatry. 2002. 72: 257-8
28. Sadatomo T, Yuki K, Migita K, Taniguchi E, Kodama Y, Kurisu K. Solitary brain metastasis from renal cell carcinoma 15 years after nephrectomy: Case report. Neurol Med Chir (Tokyo). 2005. 45: 423-7
29. Satomi Y. A clinical study of the prognosis of renal carcinoma with reference to factors on the part of host. Nippon Hinyokika Gakkai Zasshi. 1973. 64: 195-216
30. Vuong L, Kotecha RR, Voss MH, Hakimi AA. Tumor microenvironment dynamics in clear-cell renal cell carcinoma. Cancer Discov. 2019. 9: 1349-57