- Department of Neurosurgery, Sasebo Chuo Hospital, 15 Yamatocho, Sasebo City, Nagasaki, 857-1195, Japan
- Department of Neurosurgery, Faculty of Medicine, Fukuoka University, Nanakuma 7-45-1, Jonan Ward, Fukuoka, Japan
Correspondence Address:
Yoshinobu Horio
Department of Neurosurgery, Faculty of Medicine, Fukuoka University, Nanakuma 7-45-1, Jonan Ward, Fukuoka, Japan
DOI:10.4103/sni.sni_431_17
Copyright: © 2018 Surgical Neurology International This is an open access journal, and articles are distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as appropriate credit is given and the new creations are licensed under the identical terms.How to cite this article: Yoshinobu Horio, Koichiro Takemoto, Seisaburo Sakamoto, Tooru Inoue. Endovascular reconstruction for a kinked internal carotid artery after carotid endarterectomy. 13-Jul-2018;9:135
How to cite this URL: Yoshinobu Horio, Koichiro Takemoto, Seisaburo Sakamoto, Tooru Inoue. Endovascular reconstruction for a kinked internal carotid artery after carotid endarterectomy. 13-Jul-2018;9:135. Available from: http://surgicalneurologyint.com/surgicalint-articles/endovascular-reconstruction-for-a-kinked-internal-carotid-artery-after-carotid-endarterectomy/
Abstract
Background:Although different surgical techniques have been reported for repairing kinked stenosis of the internal carotid artery (ICA) after carotid endarterectomy (CEA), there are no reports using endovascular reconstruction. We present the details of a case successfully treated by carotid artery stenting (CAS).
Case Description:A 73-year-old female was referred to our department with fugacious amaurosis and transient sensory disturbance in the left upper limb due to moderate stenosis of the right ICA that was treated by CEA 28 days after admission. However, postoperative angiography and carotid Doppler revealed a kinked ICA with a high-flow velocity that was not present intraoperatively. After 3 months, she had suffered transient ischemic attacks that were refractory to medical treatment, so we performed CAS to prevent future events. The kinked ICA was immediately resolved by stenting and there was no restenosis at a follow-up angiogram 19 months later.
Conclusions:This case shows that CAS could be a potential therapeutic option for the management of symptomatic kinking stenosis of the ICA after CEA.
Keywords: Carotid artery stenting, carotid endarterectomy, kinking
INTRODUCTION
There are no clearly defined management options for kinking stenosis of the internal carotid artery (ICA) after carotid endarterectomy (CEA). Reports to date have indicated that patch angioplasty,[
CASE DESCRIPTION
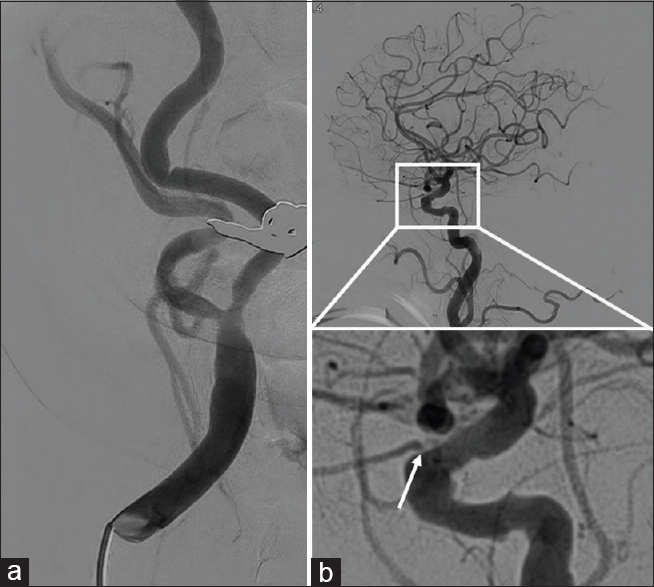
A 73-year-old female was transferred to our hospital with a history of fugacious amaurosis and transient sensory disturbance of the left upper limb, but was symptom free at admission. Magnetic resonance imaging showed an acute spotty cerebral infarction lateral to the caudate head, and magnetic resonance angiography revealed moderate stenosis in the right ICA (North American Symptomatic Carotid Endarterectomy Trial [NASCET], 58%). The right ICA was noted to be tortious, following a curved course medially. Plaque was present and markedly calcified. Also, the origin of the right ophthalmic artery was stenotic and had delayed blood flow on assessment by digital subtraction angiography (DSA) [
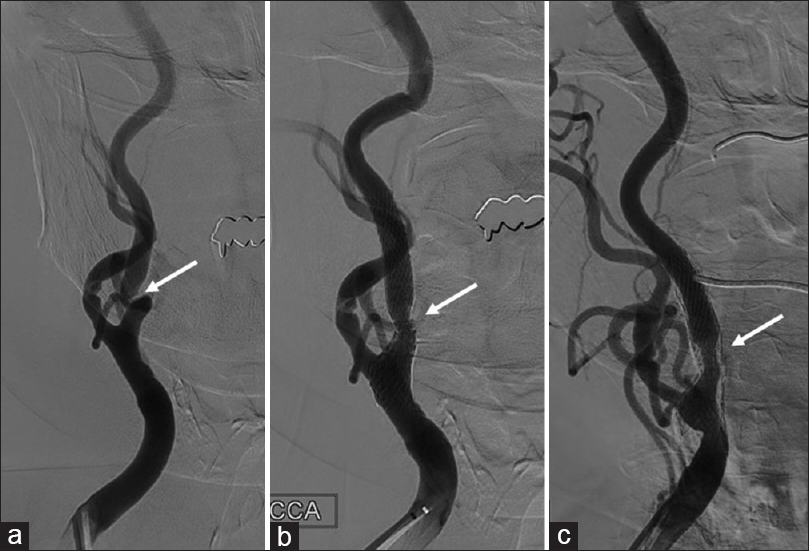
Based on the presentation, we performed CEA 21 days after onset to reduce the risk of recurrence. During surgery, no kinking stenosis was observed and video angiography with indocyanine green showed no delay in ICA blood flow. Postoperatively, clopidogrel 75 mg daily was started. Routine DSA revealed severe kinking stenosis of the right ICA. Ophthalmic symptoms appeared 20 days postoperatively. Fundus copy revealed cotton-wool spots in the right eye. Therefore, supplemental aspirin was added at a dose of 100 mg daily. Despite dual therapy, however, her symptoms recurred frequently and were refractory to further medical treatment. Moreover, no improvement of the stenotic lesion was seen on DSA or carotid Doppler ultrasound (peak systolic velocity [PSV], 226 cm/s) after 3 months. Therefore, CAS was performed under filter protection.
During the CAS procedure, the kinking lesion was easily crossed with a micro guidewire and an open cell stent was deployed at the appropriate site. However, navigation of the stent delivery system beyond the kink was difficult. Postoperatively, both the ICA stenosis and the patient's symptoms improved. Fundus copy showed that there had been resolution of the cotton-wool spots in her right eye after CAS. No restenosis was seen by DSA performed 19 months after the procedure [
DISCUSSION
Yuan et al. reported that kinking stenosis detected by intraoperative ultrasonography during CEA was seen in approximately 27 of 285 patients (9.5%). They asserted that these cases were caused by differences in the arterial wall thicknesses of the proximal and distal ICA, where newly patent vessels with thin walls after CEA were unable to support the acute changes in flow rate and velocity.[
The accurate timing of kinking is unclear. We assume that most of the kinking may occur intraoperatively or immediately after CEA, when the rotated head is returned to its actual position. Intraoperative DSA may be useful for detecting kinking at that particular time.
It was also reported by Yuan et al. that a kinking stenosis with hemodynamically significant PSV (>120 cm/s) on intraoperative duplex ultrasonography should be repaired.[
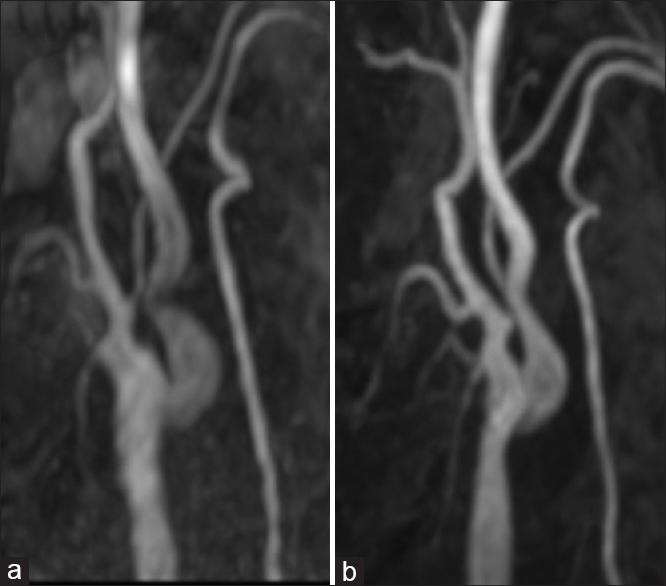
Figure 3
Representative imaging in a 60-year-old male with asymptomatic right ICA stenosis. CEA was performed, and the right ICA was kinked but asymptomatic after the procedure. The stenotic lesion subsequently improved without intervention, and the patient remained free of strokes. The images are taken after 2 months (a) and after 39 months (b)
Regarding the surgical options for repair of kinking stenosis after CEA, various procedures have been reported. However, we selected CAS because redo surgery has been reported to cause temporary cranial nerve injury in 13% of patients with restenosis after CEA.[
CONCLUSION
Kinking stenosis of the ICA after CEA can improve naturally over time, so in asymptomatic patients, conservative treatment may be the best option. However, revascularization is recommended when patients are symptomatic, and given the risk of cranial nerve injury with other procedures, we believe that CAS represents a promising alternative to current treatment strategies. Further research is required to elucidate the safety of the procedure.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Ahmadi RA, Schillinger M, Haumer M, Willfort A, Minar E. Carotid stenting in a case of combined kinking and stenosis. CardiovascIntervent Radiol. 2001. 24: 197-9
2. Harrison JH, Davalos PA. Cerebral ischemia. Surgical procedure in cases due to tortuosity and buckling of the cervical vessels. Arch Surg (Chicago, Ill: 1960). 1962. 84: 85-93
3. Illuminati G, Ricco JB, Calio FG, D’Urso A, Ceccanei G, Vietri F. Results in a consecutive series of 83 surgical corrections of symptomatic stenotic kinking of the internal carotid artery. Surgery. 2008. 143: 134-9
4. Kubota H, Sanada Y, Tanikawa R, Kato A. The use of fibrin and gelatin fixation to repair a kinked internal carotid artery in carotid endarterectomy. Surg NeurolInt. 2016. 7: S434-6
5. Marques de Marino P, Martinez Lopez I, Hernandez Mateo MM, Cernuda Artero I, Cabrero Fernandez M, Reina Gutierrez MT. Open versus endovascular treatment for patients with post-carotid endarterectomy restenosis: Early and long-term results. AnnVasc Surg. 2016. 36: 159-65
6. Milic DJ, Jovanovic MM, Zivic SS, Jankovic RJ. Coiling of the left common carotid artery as a cause of transient ischemic attacks. JVasc Surg. 2007. 45: 411-3
7. Paulsen F, Tillmann B, Christofides C, Richter W, Koebke J. Curving and looping of the internal carotid artery in relation to the pharynx: Frequency, embryology and clinical implications. JAnat. 2000. 197: 373-81
8. Poorthuis MH, Brand EC, Toorop RJ, Moll FL, de Borst GJ. Posterior transverse plication of the internal carotid artery to correct for kinking. JVasc Surg. 2014. 59: 968-77
9. Tetik O, Yurekli I, Yilik L, Akhan G, Gurbuz A. Surgical treatment of symptomatic coiling or kinking internal carotid artery. Vascular. 2010. 18: 294-6
10. Yuan JY, Durward QJ, Pary JK, Vasgaard JE, Coggins PK. Use of intraoperative duplex ultrasonography for identification and patch repair of kinking stenosis after carotid endarterectomy: Asingle-surgeon retrospective experience. World Neurosurg. 2014. 81: 334-43