- Department of Neurosurgery, Lisie Hospital, Ernakulam, Kerala, India,
- Department of Ophthalmology, Lisie Hospital, Ernakulam, Kerala, India.
Correspondence Address:
Biji Bahuleyan, Department of Neurosurgery, Lisie Hospital, Ernakulam, Kerala, India.
DOI:10.25259/SNI_580_2022
Copyright: © 2022 Surgical Neurology International This is an open-access article distributed under the terms of the Creative Commons Attribution-Non Commercial-Share Alike 4.0 License, which allows others to remix, transform, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Mariette Anto1, Adarsh Manuel1, Akarsh Jayachandran1, Santhosh George Thomas1, Anu Joseph1,2, Anjitha Thankachan1, Biji Bahuleyan1. Horner’s syndrome secondary to T1-T2 intervertebral disc prolapse. 09-Sep-2022;13:412
How to cite this URL: Mariette Anto1, Adarsh Manuel1, Akarsh Jayachandran1, Santhosh George Thomas1, Anu Joseph1,2, Anjitha Thankachan1, Biji Bahuleyan1. Horner’s syndrome secondary to T1-T2 intervertebral disc prolapse. 09-Sep-2022;13:412. Available from: https://surgicalneurologyint.com/surgicalint-articles/11854/
Abstract
Background: T1-T2 intervertebral disc prolapse (IVDP) is a rare clinical condition. Horner’s syndrome is an extremely rare clinical finding in these patients.
Case Description: A 56-year-old man presented with the left C8 T1 radiculopathy, left hand grip weakness, and ipsilateral Horner’s syndrome. Magnetic resonance imaging of the spine showed a contrast-enhancing lesion in the left T1 foramen compressing the left T1 nerve root. He underwent left T1 hemilaminectomy, upper half of left T2 hemilaminectomy and removal of the left foraminal lesion. A biopsy of the lesion was sent for histopathological diagnosis which revealed tissue consistent with disc material. Postoperatively, he had near-complete recovery with residual minimal Horner’s syndrome.
Conclusion: T1-T2 IVDP should be considered in the differential diagnosis when a patient presents with C8 T1 radiculopathy and Horner’s syndrome.
Keywords: Horner’s syndrome, Radiculopathy, Spine, Thoracic disc prolapse
INTRODUCTION
The incidence of thoracic disc prolapse is reported to be around 1 in 1000–1 in 1,000,000.[
CASE REPORT
A 56-year-old man with no previously known comorbidities presented with a 3-week history of radicular pain along the medial aspect of the left arm and forearm, numbness along the medial aspect of the left arm and forearm, and progressive left-hand grip weakness.
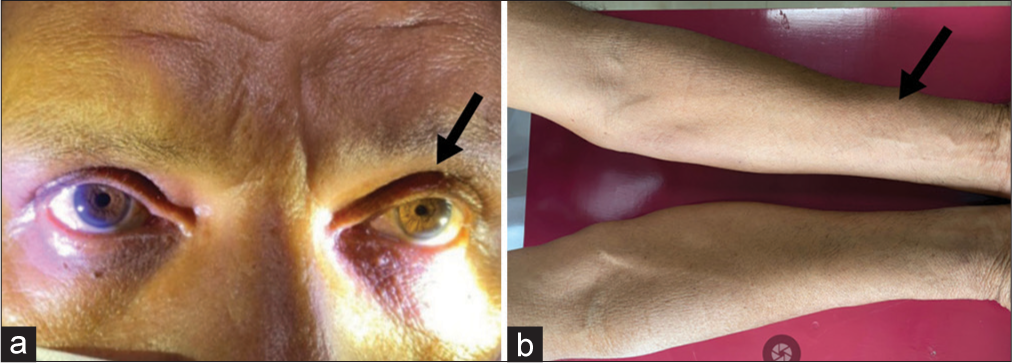
On examination, he had left-sided partial ptosis and miosis [
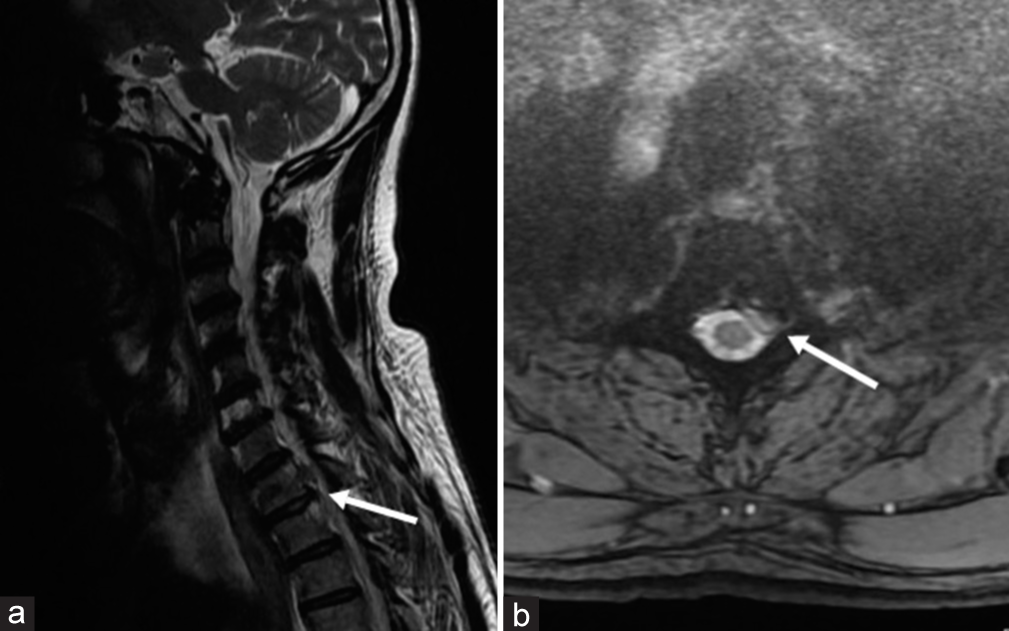
Blood counts and inflammatory markers were normal. Chest X-ray and computed tomography (CT) thorax were normal. A magnetic resonance imaging (MRI) of the cervicothoracic spine revealed T1-T2 disc prolapse on T2-weighted images [
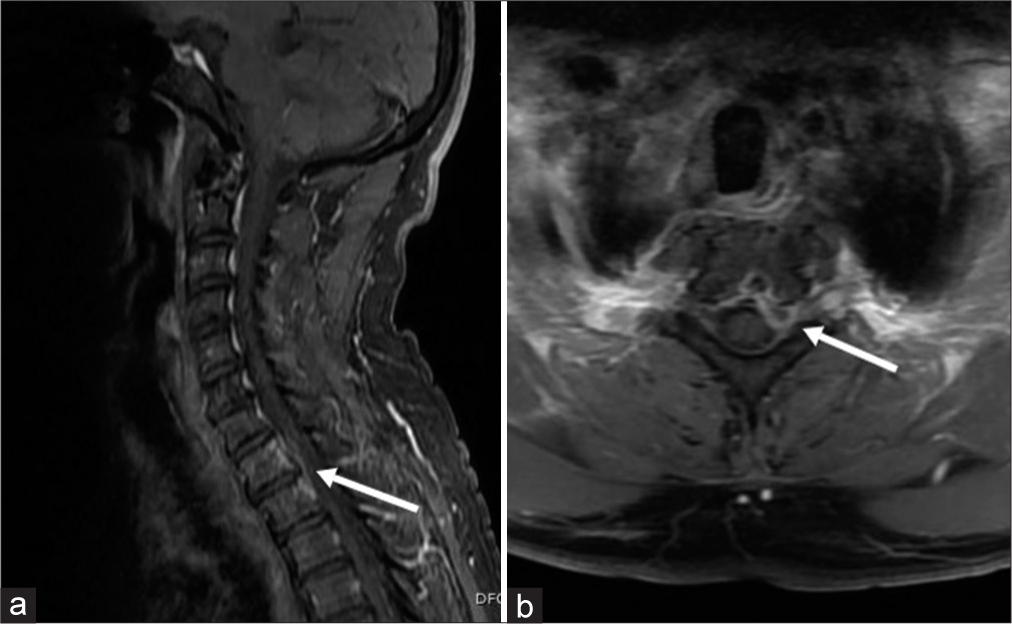
Figure 3:
(a) Gadolinium-enhanced T1-weighted sagittal magnetic resonance imaging (MRI) image of the spine showing contrast-enhancement around T1-T2 disc space (white arrow), (b) Gadolinium-enhanced T1-weighted axial MRI image of the spine showing a contrast-enhancing lesion at the left T1 foramen (white arrow).
Under general anesthesia, he underwent left T1 hemilaminectomy, upper half of left T2 hemilaminectomy, and removal of the lesion through a posterior approach in prone position with electrophysiological monitoring. Intraoperatively, the foraminal lesion was seen filling the left T1 foramen and pushing the T1 nerve root superiorly and the dural tube medially. The lesion appeared fibrous and was seen extending into the T1-T2 disc space. The lesion was removed and the nerve root and dural tube were made lax. The dura and the nerve roots were not infiltrated by the lesion. There was no purulent material. Biopsy of the lesion revealed tissue consistent with disc material with no evidence of granuloma, atypical, or malignant cells. The tissue culture from the lesion was reported as sterile. Immediately after surgery, his left upper limb radicular pain improved significantly. At 3-month follow-up, there was a total improvement in his left upper limb radicular pain, numbness, and weakness. Left forearm wasting was gradually improving, and there was residual minimal Horner’s syndrome.
DISCUSSION
T1-T2 IVDP is an extremely rare clinical entity.[
The oculosympathetic pathway consists of central, preganglionic, and postganglionic neurons with two relay centers, the ciliospinal center of Budge and the superior cervical ganglion.[
In T1-T2 IVDP, the disc can impinge on the sympathetic chain within the T1 root.[
There are multiple causes for the upper limb radiculopathy associated with Horner’s syndrome. Pancoast tumor is reported to be the most common cause of T1 nerve root compression presenting with Horner’s syndrome.[
Careful evaluation of the history and proper clinical examination can help localize the lesion.[
With technological advancement, intraoperative ultrasound and neuronavigation are being increasingly used for image-guided spine surgery.[
In 2019, Rahimizadeh et al.[
[
CONCLUSION
T1-T2 IVDP should be considered in the differential diagnosis when a patient presents with C8 T1 radiculopathy and ipsilateral Horner’s syndrome. A careful neurological examination needs to be done to look for Horner’s syndrome in these patients. A contrast-enhanced MRI spine should be the investigation of choice in these patients as it helps to rule out inflammatory and neoplastic causes of C8 T1 radiculopathy. The MRI should always extend to the upper thoracic vertebral levels in these cases. The majority of these patients require a discectomy. A small subgroup of patients with T1-T2 IVDP improves with conservative treatment. These patients generally have good outcomes.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Alstadhaug K. Ervervet Horners syndrom. Tidsskr Nor Legeforen. 2011. 131: 950-4
2. Amonoo-Kuofi HS. Horner’s syndrome revisited: With an update of the central pathway. Clin Anat. 1999. 12: 345-61
3. Araki Y, Ootani M, Furukawa T, Yamamoto T, Tsukaguchi I, Mitomo M. Pseudoenhancement of intervertebral disc herniation. Neuroradiology. 1992. 34: 271-2
4. Barrero MC, Parras N, Martín E, Marsal C, Carbonell MA, Pinto JM. Horner’s syndrome and brachial radiculopathy related to vertebral metastases. Rev Neurol. 1999. 29: 614-6
5. Bhandutia AK, Zuzek Z, Schessler MJ, Tomycz ND, Altman DT. High thoracic disc herniation causing horner syndrome with the intraoperative finding of conjoined nerve root compression: A case report. JBJS Case Connect. 2017. 7: e4
6. Caner H, Kilinçoglu BF, Benli S, Altinörs N, Bavbek M. Magnetic resonance image findings and surgical considerations in T1-2 disc herniation. Can J Neurol Sci J Can Sci Neurol. 2001. 30: 152-4
7. Carlstedt T, Hultgren T, Nyman T, Hansson T. Cortical activity and hand function restoration in a patient after spinal cord surgery. Nat Rev Neurol. 2009. 5: 571-4
8. Carson J, Gumpert J, Jefferson A. Diagnosis and treatment of thoracic intervertebral disc protrusions. J Neurol Neurosurg Psychiatry. 1971. 34: 68-77
9. Davies PR, Kaar G. High thoracic disc prolapse in a rugby player: The first reported case. Br J Sports Med. 1993. 27: 177-8
10. Ellenberg M, Ilechukwu I. Gadolinium-enhanced disc lesion. Am J Phys Med Rehabil. 2004. 83: 550
11. Foss-Skiftesvik J, Hougaard MG, Larsen VA, Hansen K. Clinical reasoning: Partial horner syndrome and upper right limb symptoms following chiropractic manipulation. Neurology. 2015. 84: e175-80
12. Ganau M, Syrmos N, Martin AR, Jiang F, Fehlings MG. Intraoperative ultrasound in spine surgery: History, current applications, future developments. Quant Imaging Med Surg. 2018. 8: 261-7
13. Gelch MM. Herniated thoracic disc at T1-2 level associated with Horner’s syndrome. Case report. J Neurosurg. 1978. 48: 128-30
14. Hammon WM. Extruded upper thoracic disc causing Horner’s syndrome: Report of a case. Med Ann Dist Columbia. 1968. 37: 541-2
15. Heckmann JG, Pauli S. Horner syndrome and thoracic disc herniation. Neurohospitalist. 2016. 6: 42
16. Kahraman A, Yildirim I, Kiliç MA, Okur E, Demirpolat G. Horner’s syndrome from giant schwannoma of the cervical sympathetic chain: Case report. B ENT. 2009. 5: 111-4
17. Kumar R, Buckley TF. First thoracic disc protrusion. Spine. 1986. 11: 499-501
18. Larkin SS, Gray AJ. An unusual cause of Horner syndrome. J Gen Intern Med. 2018. 33: 774-5
19. Lee JH, Jeen YM, Kang SG, Tark MS, Kim CH. Horner syndrome in neurofibromatosis Type 1. J Craniofac Surg. 2015. 26: 165-6
20. Lloyd T, Johnson J, Paul D, Hunt W. Horner’s syndrome secondary to herniated disc at T1--T2. Am J Roentgenol. 1980. 134: 184-5
21. Ma H, Kim I. Horner syndrome associated with a herniated cervical disc: A case report. Korean J Spine. 2012. 9: 108-10
22. Nehete LS, Konar SK, Nandeesh BN, Shukla D, Devi BI. Atypical presentation of extraspinal neurofibroma presenting with acute-onset monoparesis and Horner’s syndrome: Case report and review of literature. J Craniovertebral Junction Spine. 2019. 10: 188-91
23. Possley D, Luczak SB, Angus A, Montgomery D. T1-T2 disk herniation presenting with Horner syndrome: A case report with literary review. J Am Acad Orthop Surg Glob Res Rev. 2018. 2: e016
24. Rahimizadeh A, Zohrevand AH, Kabir NM, Asgari N. T1-T2 disc herniation: Report of four cases and review of the literature. Surg Neurol Int. 2019. 10: 56
25. Simper LB, Jørgensen AK. Intervertebral disk prolapse as an unusual cause of Horner’s syndrome. Ugeskr Laeger. 1989. 151: 949-50
26. Son ES, Lee SH, Park SY, Kim KT, Kang CH, Cho SW. Surgical treatment of t1-2 disc herniation with t1 radiculopathy: A case report with review of the literature. Asian Spine J. 2012. 6: 199-202
27. Spacey K, Dannawi Z, Khazim R, Dannawi Z. Horner’s syndrome secondary to intervertebral disc herniation at the level of T1-2. Case Rep. 2014. 2014: 204820
28. Teixeira JC, Santos MM, Melancia JL. T1-T2 Herniated disk presenting with Horner syndrome. World Neurosurg. 2017. 107: 1050.e13-5
29. Whittaker CK, Bernhardt M. Magnetic resonance imaging shows gadolinium enhancement of intradural herniated disc. Spine (Phila Pa 1976). 1994. 19: 1505-7
30. Yokota H, Yokoyama K, Noguchi H, Uchiyama Y. Spinal cord herniation into associated pseudomeningocele after brachial plexus avulsion injury: Case report. Neurosurgery. 2007. 60: E205