- Department of Neurosurgery, National Defense Medical College, Tokorozawa, Saitama, Japan
Correspondence Address:
Naoki Otani
Department of Neurosurgery, National Defense Medical College, Tokorozawa, Saitama, Japan
DOI:10.4103/sni.sni_377_16
Copyright: © 2017 Surgical Neurology International This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.How to cite this article: Naoki Otani, Terushige Toyooka, Kojiro Wada, Kentaro Mori. Modified extradural temporopolar approach with suction decompression for clipping of large paraclinoid aneurysm: Technical note. 18-Jul-2017;8:148
How to cite this URL: Naoki Otani, Terushige Toyooka, Kojiro Wada, Kentaro Mori. Modified extradural temporopolar approach with suction decompression for clipping of large paraclinoid aneurysm: Technical note. 18-Jul-2017;8:148. Available from: http://surgicalneurologyint.com/surgicalint-articles/modified-extradural-temporopolar-approach-with-suction-decompression-for-clipping-of-large-paraclinoid-aneurysm-technical-note/
Abstract
Background:Surgical clipping of complicated large paraclinoid aneurysms are still challenging because strong adhesion of aneurysm itself may hinder the dissection of the perforators and the surrounding anatomical structures from the aneurysm dome.
Methods:We describe 13 consecutive patients and the clipping of complicated, large-sized paraclinoid aneurysms using a modified extradural temporopolar approach combined with retrograde suction decompression and discuss its advantages and pitfalls.
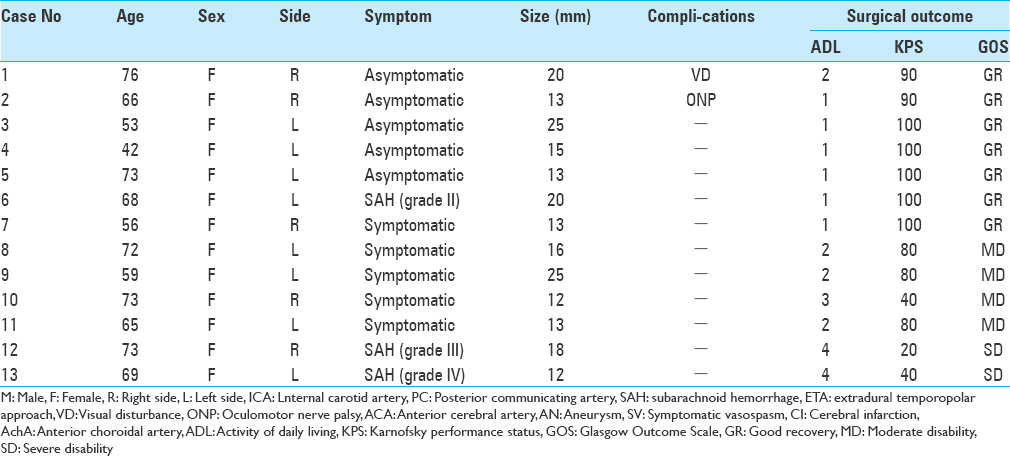
Results:Modified extradural temporopolar approach with suction decompression (SD) assistance was performed in all patients. There was no complication related to the surgical procedure. Postoperative outcome was good recovery in 7 patients, moderate disability in 4, and severe disability in 2 caused by severe subarachnoid hemorrhage. Favorable outcomes were achieved in 10 patients (84.6%).
Conclusion:We recommend its less invasive, safe, and useful combined technique in the treatment of symptomatic paraclinoid aneurysms, which carry the risk of neurovascular injury caused by dissection from the aneurysm dome.
Keywords: Extradural anterior clinoidectomy, extradural temporopolar approach, paraclinoid aneurysm, suction decompression
BACKGROUND
There are considerable difficulties in the complete clipping of large paraclinoid aneurysms for achieving proximal control of the parent artery and obtaining adequate visualization of the aneurysm neck due to their large sizes and broad necks and because of the complicated anatomical location close to the optic and surrounding bony structures.[
Microsurgical treatment of these aneurysms may often require the use of the retrograde suction decompression (RSD) technique to facilitate safe and complete clipping.[
PATIENTS AND METHODS
This retrospective study included 13 consecutive patients with complicated large-sized paraclinoid aneurysms treated by direct clipping via a modified ETA with RSD assistance between March 2004 and March 2016.
Surgical procedures
Just before positioning in the surgical posture, lumbar spinal drainage was instituted to ensure adequate brain relaxation, to obtain full exposure of the epidural space in the extradural surgical procedure, and to avoid postoperative cerebrospinal fluid leakage. Intraoperative monitoring of the motor evoked potential was routinely monitored for a safe neck clipping. Indocyanine green videoangiography and/or microvascular Doppler ultrasonography were regularly used to assess blood flow of the parent and branch vessels.
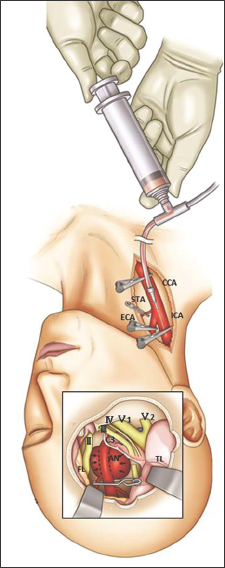
The surgical procedure is shown in
Figure 1
Surgical procedure for the suction decompression with modified extradural temporopolar approach. Just before dissection of the aneurysm, the CCA was punctured with a 20 gauge plastic needle and connected with the pressurized bag filled with heparinized saline (heparin 5000 U/500 ml saline). The aneurysm was trapped by clamping of the CCA and ECA followed by temporary clipping of the intracranial ICA distal to the aneurysm neck. Blood was then aspirated through a catheter, resulting in shrinkage of the aneurysm dome and allowing complete dissection and clipping of the aneurysm
Simultaneously, the cervical CCA, ICA, and external carotid artery (ECA) are routinely exposed for proximal control, suction decompression, intraoperative angiography, and high-flow bypass, if necessary. The CCA is punctured using a 20-gauge plastic needle just before suction decompression. After 3000 ~ 5000 U heparinization, the CCA and ECA are clamped. Subsequently, the aneurysm is temporary trapped by putting a temporal clip on the intracranial ICA distal to the aneurysm neck with special attention to spare the AchoA. Blood is aspirated through the catheter introduced into the cervical ICA, resulting in collapse of the aneurysm, and therefore, enabling the surgeon to complete dissection and neck clipping. The occlusion time can be limited to within 5 min even if there are no obvious changes occur in electrophysiological monitoring. The RSD were repeated after declamping the arteries for duration longer than each occlusion time. These procedures can be repeated until confirming the complete clipping. Intraoperative DSA through the catheter placed in the cervical ICA confirmed the complete clipping without stenosis of the parent artery. Complete hemostasis at the puncture site is achieved by suturing.
RESULTS
All patient characteristics are summarized in
DISCUSSION
Removal of the ACP is one of the essential skull base techniques to treat large paraclinoid lesions,[
The RSD through an angiocatheter inserted into the cervical ICA was reported in 1990 and 1991.[
On the other hand, endovascular techniques have gained widespread application in the treatment for paraclinoid aneurysms. However, high aneurysmal recurrent risk has been the main limitation of endovascular treatment of intracranial aneurysms.[
CONCLUSION
Our experience of direct clipping of complicated paraclinoid aneurysms using RSD through direct puncture of the CCA combined with modified ETA is a less invasive technique. The RSD technique, which can be easily and rapidly prepared, provides several benefits such as proximal flow control of the ICA, opportunity to safely repeat deflation of the aneurysm, and unlimited possibilities to perform intraoperative DSA and/or extracranial–intracranial bypass without limitations of the operative field. We recommend this less invasive, safe, and useful combined technique in the treatment of symptomatic paraclinoid aneurysms which carry the risk of neurovascular injury caused by dissection from the aneurysm dome.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
1. Batjer HH, Samson DS. Retrograde suction decompression of giant paraclinoidal aneurysms. Technical note. J Neurosurg. 1990. 73: 305-6
2. Coscarella E, Bakaya MK, Morcos JJ. An alternative extradural exposure to the anterior clinoid process: The superior orbital fissure as a surgical corridor. Neurosurgery. 2003. 53: 162-6
3. Crobeddu E, Lanzino G, Kallmes DF, Cloft HJ. Review of 2 decades of aneurysm recurrence literature, Part 1: Reducing recurrence after endovascular coiling. AJNR Am J Neuroradiol. 2013. 34: 266-70
4. Day AL. Aneurysms of the ophthalmic segment. A clinical and anatomical analysis. J Neurosurg. 1990. 72: 677-91
5. Day JD, Giannotta SL, Fukushima T. Extradural temporopolar approach to lesions of the upper basilar artery and infrachiasmatic region. J Neurosurg. 1994. 81: 230-5
6. Day JD, Fukushima T, Giannotta SL. Cranial base approaches to posterior circulation aneurysms. J Neurosurg. 1997. 87: 544-54
7. Fan YW, Chan KH, Lui WM, Hung KN. Retrograde suction decompression of paraclinoid aneurysm-a revised technique. Surg Neurol. 1999. 51: 129-31
8. Fulkerson DH, Horner TG, Payner TD, Leipzig TJ, Scott JA, DeNardo AJ. Results, outcomes, and follow-up of remnants in the treatment of ophthalmic aneurysms: A 16-year experience of a combined neurosurgical and endovascular team. Neurosurgery. 2009. 64: 218-29
9. Giannotta SL. Ophthalmic segment aneurysm surgery. Neurosurgery. 2002. 50: 558-62
10. Hauck EF, Wohlfeld B, Welch BG, White JA, Samson D. Clipping of very large or giant unruptured intracranial aneurysms in the anterior circulation: An outcome study. J Neurosurg. 2008. 109: 1012-8
11. Lanzino G, Crobeddu E, Cloft HJ, Hanel R, Kallmes DF. Efficacy and safety of flow diversion for paraclinoid aneurysms: A matched-pair analysis compared with standard endovascular approaches. AJNR Am J Neuroradiol. 2012. 33: 2158-61
12. Mori K, Yamamoto T, Nakao Y, Esaki T. Surgical Simulation of Extradural Anterior Clinoidectomy through the Trans-superior Orbital Fissure Approach Using a Dissectable Three-dimensional Skull Base Model with Artificial Cavernous Sinus. Skull Base. 2010. 20: 229-35
13. Noguchi A, Balasingam V, Shiokawa Y, McMenomey SO, Delashaw JB. Extradural anterior clinoidectomy. Technical note. J Neurosurg. 2005. 102: 945-50
14. Nutik SL. Removal of the anterior clinoid process for exposure of the proximal intracranial carotid artery. J Neurosurg. 1988. 69: 529-34
15. Otani N, Wada K, Toyooka T, Fujii K, Kobayashi Y, Mori K. Operative surgical nuances of modified extradural temporopolar approach with mini-peeling of dura propria based on cadaveric anatomical study of lateral cavernous structures. Surgical neurology Int. 2016. 7: S454-8
16. Puffer RC, Kallmes DF, Cloft HJ, Lanzino G. Patency of the ophthalmic artery after flow diversion treatment of paraclinoid aneurysms. J Neurosurg. 2012. 116: 892-6
17. Tamaki N, Kim S, Ehara K, Asada M, Fujita K, Taomoto K. Giant carotid-ophthalmic artery aneurysms: Direct clipping utilizing the “trapping-evacuation“ technique. J Neurosurg. 1991. 74: 567-72
18. Tripathi M, Deo RC, Damodaran N, Suri A, Srivastav V, Baby B. Quantitative analysis of variable extent of anterior clinoidectomy with intradural and extradural approaches: 3-dimensional analysis and cadaver dissection. Neurosurgery. 2015. 11: 147-60